
��12�֣���1����25�������½�pH=11�İ�ˮϡ��100������Һ��pHΪ__ ������ţ���
A��9 B��13 C��11��13֮�� D��9��11֮��
(2) pH��3�Ĵ����pH��11������������Һ�������Ϻ����Һ��
c(Na��)_______c(CH3COO��) �� �>������������<������
(3) �����HCl��CH3COONa ��Һ��Ũ�ȡ��������ϣ�������Һ�и�����Ũ�ȵĴ�С��ϵΪ ��
(4) �����£���NaOH��Һ�е�c(OH�D)��CH3COOH ��Һ�е�c(H��)��ͬ���ֽ�NaOH��CH3COOH����Һ�ֱ�ϡ��10����ϡ�ͺ�NaOH �� CH3COOH��Һ��pH�ֱ���pH1��pH2��ʾ����pH1+pH2 14�� ��>������������<����
��5����Һ���������ȵ�ϡ����Ͱ�ˮ��϶��ɣ���ǡ�ó����ԣ�����ǰc(HCl)______c(NH3��H2O)�������С�ڻ���ڣ���ͬ��,���ǰ����c(H+)�ͼ���c(OH-)�Ĺ�ϵc(H+)_____c(OH-).

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
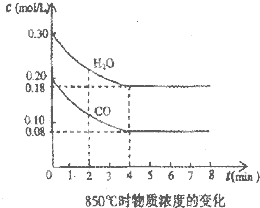 ��1����25�������½�pH=3�Ĵ���ϡ��100������Һ��pHΪ
��1����25�������½�pH=3�Ĵ���ϡ��100������Һ��pHΪ�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
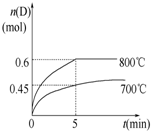 ����ѧ��Ӧԭ����
����ѧ��Ӧԭ�����鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com