
����ʹ�õ�ȼ�ϣ��ִ��Ϊú����Һ��ʯ������ú������Ҫ�ɷ�ΪCO�� �Ļ�����壬����ú̿��ˮ�����ڸ����·�Ӧ�Ƶã����ֳ�ˮú�����Խ���������⣺
�Ļ�����壬����ú̿��ˮ�����ڸ����·�Ӧ�Ƶã����ֳ�ˮú�����Խ���������⣺
(1)д����ȡˮú������Ҫ��ѧ����ʽ________���÷�Ӧ��________��Ӧ��(����ȡ����ȡ�)
(2)��Һ��ʯ��������Ҫ�ɷ�Ϊ���� ������ȼ�պ����Ϊ
������ȼ�պ����Ϊ ��
�� ���ԱȽ���ȫȼ�յ�������
���ԱȽ���ȫȼ�յ������� ��CO�����������������Ϊ________��
��CO�����������������Ϊ________��
(3)������δ������Դ��ȼ��ʱ�������������⣬�����е��ŵ���_________��
(4)ʹ�ùܵ�ú�������ֹ��ú��й©��������ж��ͱ�ը��ijס����ú��й©��������ը�¹ʣ����鷢��ú�����ڴﵽ���ڿ���������6��ʱ������ը�ģ���ס��ú��й©�ٶ�Ϊ0.6g��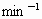 ����ըʱú����й©Լ________h(����������Ϊ25
����ըʱú����й©Լ________h(����������Ϊ25 �������ܶ�ԼΪ1.29g��
�������ܶ�ԼΪ1.29g�� )
)

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��8�֣�����ʹ�õ�ȼ�ϣ��ִ����ú����Һ��ʯ������ú������Ҫ�ɷ���CO��H2�Ļ�����壬����ú̿��ˮ������Ӧ�Ƶã����ֳ�ˮú����
��1����д����ȡˮú������Ҫ��ѧ����ʽ____________________________.
��2��Һ��ʯ��������Ҫ�ɷ��DZ��飬����ȼ�յ��Ȼ�ѧ����ʽΪ
C3H8(g) +5O2(g)== 3CO2(g) + 4H2O(l)����H=�C2220.0 kJ��![]() ,
,
��֪CO����ȼ�յ��Ȼ�ѧ����ʽΪ��
CO(g) +1/2O2(g) == CO2(g) ����H=�C282.57kJ��![]() ,
,
�ԱȽ�ͬ������C3H8��COȼ�գ�������������ֵԼΪ_________��1��
��3����֪����ȼ�յ��Ȼ�ѧ����ʽΪ2H2(g)+ O2(g) == 2H2O(l)����H=�C571.6 kJ��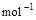 ,�ԱȽ�ͬ������H2��C3H8ȼ�գ�������������ֵԼΪ____________��1��
,�ԱȽ�ͬ������H2��C3H8ȼ�գ�������������ֵԼΪ____________��1��
��4��������δ������Դ������Դ�ḻ֮�⣬�����е��ŵ���____________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010������ʡ��Ϫ�ظ��и߶����ڲ��νο��Ի�ѧ���� ���ͣ������
��8�֣�����ʹ�õ�ȼ�ϣ��ִ����ú����Һ��ʯ������ú������Ҫ�ɷ���CO��H2�Ļ�����壬����ú̿��ˮ������Ӧ�Ƶã����ֳ�ˮú����
��1����д����ȡˮú������Ҫ��ѧ����ʽ____________________________.
��2��Һ��ʯ��������Ҫ�ɷ��DZ��飬����ȼ�յ��Ȼ�ѧ����ʽΪ
C3H8(g) +5O2(g) == 3CO2(g) + 4H2O(l)����H=�C2220.0 kJ�� ,
,
��֪CO����ȼ�յ��Ȼ�ѧ����ʽΪ��
CO(g) +1/2O2(g) == CO2(g) ����H=�C282.57kJ�� ,
,
�ԱȽ�ͬ������C3H8��COȼ�գ�������������ֵԼΪ_________��1��
��3����֪����ȼ�յ��Ȼ�ѧ����ʽΪ2H2(g) + O2(g) == 2H2O(l)����H=�C571.6 kJ�� ,�ԱȽ�ͬ������H2��C3H8ȼ�գ�������������ֵԼΪ____________��1��
,�ԱȽ�ͬ������H2��C3H8ȼ�գ�������������ֵԼΪ____________��1��
��4��������δ������Դ������Դ�ḻ֮�⣬�����е��ŵ���____________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010������ʡ�߶����ڲ��νο��Ի�ѧ���� ���ͣ������
��8�֣�����ʹ�õ�ȼ�ϣ��ִ����ú����Һ��ʯ������ú������Ҫ�ɷ���CO��H2�Ļ�����壬����ú̿��ˮ������Ӧ�Ƶã����ֳ�ˮú����
��1����д����ȡˮú������Ҫ��ѧ����ʽ____________________________.
��2��Һ��ʯ��������Ҫ�ɷ��DZ��飬����ȼ�յ��Ȼ�ѧ����ʽΪ
C3H8(g) +5O2(g)
== 3CO2(g) + 4H2O(l)����H=�C2220.0 kJ��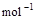 ,
,
��֪CO����ȼ�յ��Ȼ�ѧ����ʽΪ��
CO(g) +1/2O2(g) == CO2(g) ����H=�C282.57kJ��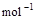 ,
,
�ԱȽ�ͬ������C3H8��COȼ�գ�������������ֵԼΪ_________��1��
��3����֪����ȼ�յ��Ȼ�ѧ����ʽΪ2H2(g)
+ O2(g) == 2H2O(l)����H=�C571.6 kJ��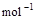 ,�ԱȽ�ͬ������H2��C3H8ȼ�գ�������������ֵԼΪ____________��1��
,�ԱȽ�ͬ������H2��C3H8ȼ�գ�������������ֵԼΪ____________��1��
��4��������δ������Դ������Դ�ḻ֮�⣬�����е��ŵ���____________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��ͬ���� ���ͣ��ж���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����ʹ�õ�ȼ�ϣ��ִ����ú����Һ��ʯ������ú������Ҫ�ɷ���CO��H2�Ļ�����壬����ú̿��ˮ������Ӧ�Ƶã����ֳ�ˮú����
��1����д����ȡˮú������Ҫ��ѧ����ʽ____________________________.
��2��Һ��ʯ��������Ҫ�ɷ��DZ��飬����ȼ�յ��Ȼ�ѧ����ʽΪ
C3H8(g) +5O2(g) == 3CO2(g) + 4H2O(l)����H=�C2220.0 kJ��![]() ,
,
��֪CO����ȼ�յ��Ȼ�ѧ����ʽΪ��
CO(g) +1/2O2(g) == CO2(g) ����H=�C282.57kJ��![]() ,
,
�ԱȽ�ͬ������C3H8��COȼ�գ�������������ֵԼΪ_________��1��
��3����֪����ȼ�յ��Ȼ�ѧ����ʽΪ2H2(g) + O2(g) == 2H2O(l)����H=�C571.6 kJ��![]() ,�ԱȽ�ͬ������H2��C3H8ȼ�գ�������������ֵԼΪ____________��1��
,�ԱȽ�ͬ������H2��C3H8ȼ�գ�������������ֵԼΪ____________��1��
��4��������δ������Դ������Դ�ḻ֮�⣬�����е��ŵ���____________________
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com