
����Ŀ��2 L�ܱ������н��з�Ӧ��pZ(g)��qQ(g) ![]() mX(g)��nY(g)��ʽ��m��n��p��qΪ��ѧ����������0��3 min�ڣ������ʵ����ʵ����ı仯���±���ʾ��
mX(g)��nY(g)��ʽ��m��n��p��qΪ��ѧ����������0��3 min�ڣ������ʵ����ʵ����ı仯���±���ʾ��
���� | X | Y | Z | Q |
��ʼ/mol | 0.7 | 1 | ||
2 minĩ/mol | 0.8 | 2.7 | 0.8 | 2.7 |
3 minĩ/mol | 0.8 |
��֪��2 min��v(Q)��0.075 mol��L��1��min��1��v(Z)��v(Y)��1��2��
��ش��������⣺
(1)2 min��X�ķ�Ӧ����v(X)��__________��
(2)��ʼʱn(Y)��__________��
(3) 3 minĩ�Ƿ�ﵽƽ��_______(���ǻ��)��
(4)���ڸ÷�Ӧ������������Ӧ���ʵĴ�ʩ��________(����ţ���ͬ)��
A����С������� B�����߲���Q
C��ͨ�����He�� D�������¶�
���𰸡�0.025 mol��L��1��min��1 2.3 mol �� A D
��������
����ͼ�����ݼ���Ӧ����ʽ���㷴Ӧ���ʣ��������ݷ�����Ӧ�Ƿ�ﵽƽ�⡣
2 L�ܱ������н��з�Ӧ��pZ(g)��qQ(g) ![]() mX(g)��nY(g)��ʽ��m��n��p��qΪ��ѧ����������0��3 min�ڣ������ʵ����ʵ����ı仯���±���ʾ��
mX(g)��nY(g)��ʽ��m��n��p��qΪ��ѧ����������0��3 min�ڣ������ʵ����ʵ����ı仯���±���ʾ��
���� | X | Y | Z | Q |
��ʼ/mol | 0.7 | 1 | ||
2 minĩ/mol | 0.8 | 2.7 | 0.8 | 2.7 |
3 minĩ/mol | 0.8 |
��֪��2 min��v(Q)��0.075 mol��L��1��min��1��v(Z)��v(Y)��1��2��
��ش��������⣺
(1)2 min��X��Ũ��0.35 mol��L��1��Ϊ0.40 mol��L��1������v(X)��![]() =0.025 mol��L��1��min��1��
=0.025 mol��L��1��min��1��
�ʴ�Ϊ��0.025 mol��L��1��min��1��
(2)v(Z)��v(Y)��1��2����p��n=1��2��2min��Z��Ӧ��1mol-0.8mol=0.2mol����ͬʱ����YΪ0.2mol��2=0.4mol������ʼʱn(Y)��2.7mol-0.4mol=2.3 mol ��
�ʴ�Ϊ��2.3 mol ��
(3) 2minĩ��3 minĩZ�����ʵ�����ȣ���˵���Ѿ��ﵽƽ�⣬
�ʴ�Ϊ���ǣ�
(4)A����С�����������Ӧ��Ũ����������Ӧ���ʼӿ죬��A��ȷ��
B�����߲���Q����Ӧ��Ũ�ȼ�С������Ӧ���ʼ�С����B����
C��ͨ�����He������Ӧ��Ũ�Ȳ��䣬����Ӧ���ʲ��䣬��C����
D�������¶ȣ�����Ӧ���ʼӿ죬��D��ȷ��
�ʴ�Ϊ��AD��


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ijʵ��С�����������װ�öԽ�̿��ԭ���������������ijɷֽ���̽����
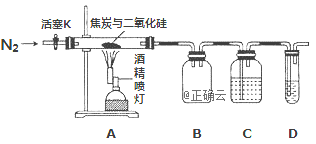
��֪��PdCl2��Һ�����ڼ���CO����Ӧ�Ļ�ѧ����ʽΪ�� CO+PdCl2 + H2O = CO2 +2HCl + Pd����������ɫ�����ٷ�ĩ��ʹ��Һ����ǣ���
��1��ʵ��ʱҪͨ���㹻��ʱ���N2����ԭ����________
��2��װ��B��������________
��3��װ��C����ʢ�Լ�ӦΪ___________
��4����װ�õ�ȱ����________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����������Ϊ����ɫ��ij��������һ����ѩ��������ɫ�����ѩ���������£��������ѷ����ܱ�״̬���õ��������ڣ����һ��֧��С�������ڷ�����ǣ�����ʹ����ۻ���ȼ�գ��������ң���ѩ�������Ƴ��ˣ���ѩ�����������ã��������к���������ѩ�������õ���
A. ��Ļ�ԭ��
B. ���Ư����
C. ��������Ļ�ԭ��
D. ���������Ư����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��X��Y��Z��Q��W��R���ֶ�����Ԫ��ԭ��������������������׳ƿ����ƣ���X��Z��Q����Ԫ����ɡ�ҽѧ�г���QR��Һ����������ˮ������������X��R����Ԫ����ɡ���ش��������⣺
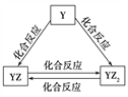
(1)Q�����ӽṹʾ��ͼΪ____________________��
(2)YԪ�صĵ��ʼ��仯�����ܷ�������ͼ��ʾ��ת������YԪ��Ϊ___(��Ԫ�ط���)���ڼ���Һ��ͨ������YZ2���壬д���÷�Ӧ�����ӷ���ʽ��________________________��
(3)W�ĵ��ʼ��������Һ��Ӧ������������Һ��Ӧ�������£���W�ĵ��ʺͼ���Һ��ϣ�������Ӧ�����ӷ���ʽΪ___________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����500 mL��BaCl2��KCl�Ļ����Һ�ֳ�5�ȷݣ�ȡһ�ݼ��뺬a mol�����Ƶ���Һ��ǡ��ʹ��������ȫ��������ȡһ�ݼ��뺬b mol����������Һ��ǡ��ʹ��������ȫ��������ԭ�����Һ�м��������ʵ���Ũ��Ϊ�� ��
A. 0.1��b-2a��mol/L B. 10��2a-b��mol/L
C. 10��b-a��mol/L D. 10��b-2a��mol/L
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���±�ΪԪ�����ڱ���һ���֡�
̼ | �� | Y | |
X | �� | Z |
�ش���������:
��1��ZԪ�������ڱ��е�λ��Ϊ_______________��
��2��������ʵ��˵��YԪ�صķǽ����Ա���Ԫ�صķǽ�����ǿ����___________��
a.Y������H2S��Һ��Ӧ����Һ�����
b.��������ԭ��Ӧ�У�1molY���ʱ�1mol��õ��Ӷ�
c.Y������Ԫ�صļ��⻯�����ȷֽ���ǰ�ߵķֽ��¶ȸ�
��3��X��Z��Ԫ�صĵ��ʷ�Ӧ����1molX����ۻ�����ָ������£�����687kJ����֪�û�������ۡ��е�ֱ�Ϊ-69����58����д���÷�Ӧ���Ȼ�ѧ����ʽ:_____________��
��4��1mol̼��þ�γɵĻ�����Q��4moˮ��Ӧ������2molMg(OH)2��1mol��������������̼��������Ϊ9:1�����ĵ���ʽΪ____________��
��5��Q��ˮ��Ӧ�Ļ�ѧ����ʽΪ_____________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ѡ��1���Լ�һ�μ����Ȼ�����ƫ�����ơ���������̼����������ɫ��Һ�������Լ���(����)
A.����B.ϡ����C.����������ҺD.�Ȼ�����Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����й���������������������ڷ�Ӧ�������������( )
A. HCl + NaOH = NaCl+H2O B. CaO+H2O=Ca(OH)2
C. 2Na + 2H2O = 2NaOH+H2�� D. H��Br �� H + Br
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����1����5.1gþ���Ͻ�ķ�ĩ��������������У��õ�5.6 LH2����״���£���
�ٺϽ���þ�����ʵ���_________��
��д���úϽ���������NaOH��Һ�Ļ�ѧ����ʽ_________________��ͬʱ����H2���������״���£�Ϊ____________ ��
��2����ͬ�����£�ijCl2��O2�������100 mLǡ����150 mL H2��������HCl��H2O������������Cl2��O2�������Ϊ__________����������ƽ����Է�������Ϊ____________��
��3��������ͬ�ݻ����ܱ�����X��Y����25 ���£�X�г���a g A���壬Y�г���a g CH4���壬X��Y�ڵ�ѹǿ֮����4��11����A��Ħ������Ϊ____________��
��4����ͬ�¡�ͬѹ�£�ʵ����CO��N2��O2��������Ļ��������ܶ���H2��14.5��������O2����������Ϊ____________ (����������1λС��) ��������CO��N2�����ʵ���֮��Ϊ1��1��������������Ԫ�ص���������Ϊ_______________ (����������1λС��)��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com