
�о����������������ڴ����к������ӵ������ʱ���漰���·�Ӧ��
2NO2��g��+NaCl��s��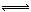 NaNO3��s��+ClNO��g�� K1 ∆H < 0 ��I��
NaNO3��s��+ClNO��g�� K1 ∆H < 0 ��I��
2NO�� g��+Cl2��g��
g��+Cl2��g��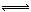 2ClNO��g�� K2 ∆H < 0 ��II��
2ClNO��g�� K2 ∆H < 0 ��II��
��1��4NO2��g��+2NaCl��s��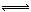 2NaNO3��s��+2NO��g��+Cl2��g����ƽ�ⳣ��K=
2NaNO3��s��+2NO��g��+Cl2��g����ƽ�ⳣ��K=  ����K1��K2��ʾ����
����K1��K2��ʾ����
��2��Ϊ�о���ͬ�����Է�Ӧ��II����Ӱ�죬�ں��������£���2L�����ܱ������м���0.2mol NO��0.1mol Cl2��10minʱ��Ӧ��II���ﵽƽ�⡣���10min ��v��ClNO��=7.5��10-3mol•L-1•min-1����ƽ���n��Cl2��= mol��NO��ת���ʧ�1= �������������ֲ��䣬��Ӧ��II���ں�ѹ�����½��У�ƽ��ʱNO��ת���ʧ�2 ��1���>����<����=������ƽ�ⳣ��K2 ���������С�����䡱����ҪʹK2��С���ɲ��õĴ�ʩ�� ��
��v��ClNO��=7.5��10-3mol•L-1•min-1����ƽ���n��Cl2��= mol��NO��ת���ʧ�1= �������������ֲ��䣬��Ӧ��II���ں�ѹ�����½��У�ƽ��ʱNO��ת���ʧ�2 ��1���>����<����=������ƽ�ⳣ��K2 ���������С�����䡱����ҪʹK2��С���ɲ��õĴ�ʩ�� ��
��3��ʵ���ҿ���NaOH��Һ����NO2����ӦΪ2NO2+2NaOH=NaNO3+NaNO2+H2O����0.2mol NaOH��ˮ��Һ��0.2mol NO2ǡ����ȫ��Ӧ��1L�� ҺA����ҺBΪ0.1mol•L‾1��CH3COONa��Һ��������Һ��c��NO3‾����c��NO2-����c��CH3COO‾���ɴ�С��˳��Ϊ ������֪HNO2�ĵ��볣��Ka=7.1��10-4mol•L‾1��CH3COOH�ĵ��볣��K a=1.7��10-5mol•L‾1����ʹ��ҺA����ҺB��pH��ȵķ����� ��
ҺA����ҺBΪ0.1mol•L‾1��CH3COONa��Һ��������Һ��c��NO3‾����c��NO2-����c��CH3COO‾���ɴ�С��˳��Ϊ ������֪HNO2�ĵ��볣��Ka=7.1��10-4mol•L‾1��CH3COOH�ĵ��볣��K a=1.7��10-5mol•L‾1����ʹ��ҺA����ҺB��pH��ȵķ����� ��
a������ҺA�м�����ˮ b.����ҺA�м�����NaOH
c������ҺB�м�����ˮ d..����ҺB�м�����NaOH
 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij������Һ�����ܺ���NH4����Mg2+��Al3+��Na����Cu2����OH����Cl����I����
NO3����SO42���������еļ��֣��ֽ�������ʵ�飺
�����Թ���ȡ������Һ���μӼ���ʯ����Һ����Һ�ʺ�ɫ��
����ȡԭ��Һ��������������CCl4���ڲ����������¼���������ˮ�����ã��ϲ�ӽ���ɫ���²���Ϻ�ɫ��
���������ˮ��Һ�м���AgNO3��Һ���а�ɫ�������ɡ�
����ȡԭ��Һ���μ�NaOH��Һ���ð�ɫ��������������NaOH��Һ�����������������ܽ⣬���˺����Һ���ȣ��д̼�����ζ�����������
��1���ɴ��ж�ԭ��Һ��һ���д����� ���ӡ�
��2������ڷ�Ӧ�����ӷ���ʽ��________________________________
��3��������г����ܽ�����ӷ���ʽ��
��4��������ȷ������������ ����μ��� ��д��ʵ�����Ƽ��ж����ݵ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�о����������������ڴ����к������ӵ������ʱ���漰���·�Ӧ��
2NO2(g)��NaCl(s) NaNO3(s)��ClNO(g)��K1����H1<0(��)
NaNO3(s)��ClNO(g)��K1����H1<0(��)
2NO(g)��Cl2(g) 2ClNO(g)��K2����H2<0(��)
2ClNO(g)��K2����H2<0(��)
(1)4NO2(g)��2NaCl(s) 2NaNO3(s)��2NO(g)��Cl2(g)��ƽ�ⳣ��K��________(��K1��K2��ʾ)��
2NaNO3(s)��2NO(g)��Cl2(g)��ƽ�ⳣ��K��________(��K1��K2��ʾ)��
(2)Ϊ�о���ͬ�����Է�Ӧ(��)��Ӱ�죬�ں��������£���2 L�����ܱ������м���0.2 mol NO��0.1 mol Cl2��10 m inʱ��Ӧ(��)�ﵽƽ�⡣���10 min��v(ClNO)��7.5��10��3 mol·L��1·min��1����ƽ���n(Cl2)��________mol��NO��ת������1��________�������������ֲ��䣬��Ӧ(��)�ں�ѹ�����½��У�ƽ��ʱNO��ת���ʦ�2________��1(�>����<������)��ƽ�ⳣ��K2______(�������С�����䡱)����ҪʹK2��С���ɲ�ȡ�Ĵ�ʩ��________��
inʱ��Ӧ(��)�ﵽƽ�⡣���10 min��v(ClNO)��7.5��10��3 mol·L��1·min��1����ƽ���n(Cl2)��________mol��NO��ת������1��________�������������ֲ��䣬��Ӧ(��)�ں�ѹ�����½��У�ƽ��ʱNO��ת���ʦ�2________��1(�>����<������)��ƽ�ⳣ��K2______(�������С�����䡱)����ҪʹK2��С���ɲ�ȡ�Ĵ�ʩ��________��
(3)ʵ���ҿ���NaOH��Һ����NO2����ӦΪ2NO2��2NaOH===NaNO3��NaNO2��H2O����0.2 mol NaOH��ˮ��Һ��0. 2 mol NO2ǡ����ȫ��Ӧ��1 L��ҺA����ҺBΪ0.1 mol·L��1��CH3COONa��Һ��������Һ��c(NO
2 mol NO2ǡ����ȫ��Ӧ��1 L��ҺA����ҺBΪ0.1 mol·L��1��CH3COONa��Һ��������Һ��c(NO )��c(NO
)��c(NO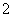 )��c(CH3COO��)�ɴ�С��˳��Ϊ____________________________________________��(��֪HNO2���볣��Ka��7.1��10��4 mol·L��1��CH3COOH�ĵ��볣��Ka��1.7��10��5 mol·L��1)
)��c(CH3COO��)�ɴ�С��˳��Ϊ____________________________________________��(��֪HNO2���볣��Ka��7.1��10��4 mol·L��1��CH3COOH�ĵ��볣��Ka��1.7��10��5 mol·L��1)
��ʹ��ҺA����ҺB��pH��ȵķ�����________��
a������ҺA�м�����ˮ
b������ҺA�м�����NaOH
c������ҺB�м�����ˮ
d������ҺB�м�����NaOH
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������������ʼ��仯�����ڹ�ҵ���й㷺Ӧ�á�
��1��ͬ��ʯ�ڸ������Ʊ������Ȼ�ѧ����ʽΪ��
4Ca5(PO4)3F(s)+21SiO2(s)+30C(s)=3P4(g)+20CaSiO3(s)+30CO(g)+SiF4(g) H
H
��֪��ͬ�����£�
4Ca3(PO4)2F(s)+3SiO2(s)=6Ca3(PO4)2(s)+2CaSiO3(s)+SiF4(g) ��H1
2Ca3(PO4)2(s)+10C(s)=P4(g)+6CaO(s)+10CO(g) ��H2
SiO2(s)+CaO(s)=CaSiO3(s) ��H3
�á�H1����H2�͡�H3��ʾ H����
H���� H=____________________��
H=____________________��
��2�������������Ϊ����������ӣ�����ṹʽ��ͼ��֮����ȥ����ˮ���Ӳ����ṹʽΪ________________________________�����������ƣ��׳ơ����ơ����dz��õ�ˮ���������仯ѧʽΪ____________��
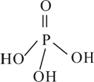
��3���������ƣ�NaH2PO2�������ڹ�ҵ�ϵĻ�ѧ������
�ٻ�ѧ��������Һ�к���Ni2+��H2PO2���������Ե������·���������Ӧ��
��a��_____ Ni2+ + ____ H2PO2��+ _____ �� ___Ni++______ H2PO3��+ ____
��b��6H2PO-2 +2H+ =2P+4H2PO3+3H2��
���������д������ƽ��Ӧʽ��a����
�����â��з�Ӧ�������϶Ƽ����������—�Ͻ𣬴Ӷ��ﵽ��ѧ������Ŀ�ģ�����һ�ֳ����Ļ�ѧ�ơ�������·���Ƚϻ�ѧ�����ơ�
�����ϵIJ�ͬ�㣺______________________________________________________��
ԭ���ϵIJ�ͬ�㣺______________________________________________________��
��ѧ�Ƶ��ŵ㣺________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
һ���¶��£���ij���ݵ��ܱ������м���0.2 mol CO��0.2 mol H2O���������·�Ӧ��CO(g)��H2O(g)  CO2(g)��H2(g)��4 minʱ��Ӧ�ﵽƽ��״̬�����n(CO)��n(CO2)��3��2������˵����ȷ����(����)
CO2(g)��H2(g)��4 minʱ��Ӧ�ﵽƽ��״̬�����n(CO)��n(CO2)��3��2������˵����ȷ����(����)
A����С�����������������ܶȲ���
B��v��(CO)��v��(CO2)ʱ��������Ӧ�ﵽƽ��״̬
C��ƽ��ʱCO��H2O��ת�������
D��������������·�Ӧ��ƽ�ⳣ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
һ�������´��ڷ�Ӧ��CO(g)��H2O(g)CO2(g)��H2(g)��������Ӧ���ȡ�����������ͬ��2 L���ݾ���(�����û����������)�ܱ��������ڢ��г���1 mol CO��1 mol H2O���ڢ��г���1 mol CO2��1 mol H2���ڢ��г���2 mol CO��2 mol H2O,700 �������¿�ʼ��Ӧ���ﵽƽ��ʱ������˵����ȷ����(����)
A��������������Ӧ������ͬ
B���������з�Ӧ��ƽ�ⳣ����ͬ
C����������CO�����ʵ������������еĶ�
D����������CO��ת��������������CO2��ת����֮��С��1
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����������������Դ������Ҫ�Ļ���ԭ�ϣ��ü�����������һ�����۵����ⷽ�����йص��Ȼ�ѧ����ʽ���£�
��CH4(g)�� O2(g)
O2(g)  CO(g)��2H2(g)����H<0
CO(g)��2H2(g)����H<0
��CH4(g)��H2O(g)  CO(g)��3H2(g)����H>0
CO(g)��3H2(g)����H>0
����˵������ȷ����(����)
A������Ӧ����v(CH4)����v(CO)��ʱ�������÷�Ӧ��ƽ��״̬
B��������������ʱ��������ϵѹǿ����Ӧ�١����м����ת���ʾ���С
C��ʹ�ø�Ч�������Լӿ췴Ӧ���ʣ�ͬʱ���CH4��ת����
D��ͬһ������ͬʱ������Ӧ�����ʱ���������¶ȿ��ܱ��ֲ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����£�����������Һ�У���ˮ�������������Ũ��֮��Ϊ(����)
��1 mol/L �����ᡡ��0.1 mol·L��1������
��0.01 mol·L��1��NaOH
A��1��10��100 B��0��1��12
C��14��13��12 D��14��13��2
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com