
 2CuO,CuO+2HNO3="=" Cu(NO3)2 + H2O
2CuO,CuO+2HNO3="=" Cu(NO3)2 + H2O 3Cu(NO3)2��2NO����4H2O
3Cu(NO3)2��2NO����4H2O| A�����ַ��������漰�ķ�Ӧ������������ԭ��Ӧ |
| B�����ַ���������������ʣ���>��>�� |
| C����ȡ��ͬ��������ͭ���ڲ������к�����Ȣ۶� |
| D�����ַ����У��ٷ������� |
 �����ÿ�ʱѵ��ϵ�д�
�����ÿ�ʱѵ��ϵ�д� ��Ԫȫ��������ϵ�д�
��Ԫȫ��������ϵ�д� �»ƸԱ����ܾ�ϵ�д�
�»ƸԱ����ܾ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����������(����)����NaOH��Һ����Һ |
| B����������(����)����Na2CO3��Һ����Һ |
| C�������飨�Ҵ�������ˮ����Һ |
| D����(����)������ˮ������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A�������ʵ��������������ڹ��������·�Ӧ���������������ȡһ�����顣 |
| B��ʵ��������ϩ����ʱ���¶�Ӧ������������ֹ����̼���� |
| C��ʵ��������Ȳ�ռ��������������д̼�����ζ������������Ȳ�л�������H2S�����塣 |
| D��ʯ�ͷ���ʱ���¶ȼƲ������ȵ�Һ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
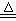 Cl2����MnCl2��2H2O�Իش��������⣺
Cl2����MnCl2��2H2O�Իش��������⣺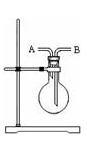
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
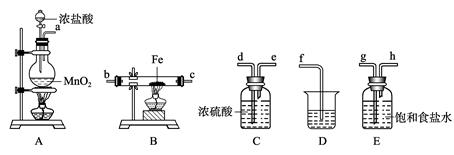
 _________________________________��
_________________________________�� _________________________��
_________________________�� ��
��
 ͼ��ʾ����װ���ɸ���ܡ��齺�ܺ�50 mL�ζ��ܸ������װ���ɣ��˴����õζ�����________�����ʽ����ʽ�����ζ��ܡ�
ͼ��ʾ����װ���ɸ���ܡ��齺�ܺ�50 mL�ζ��ܸ������װ���ɣ��˴����õζ�����________�����ʽ����ʽ�����ζ��ܡ� �ᵼ��������������__________���ƫ����ƫС������Ӱ�족����
�ᵼ��������������__________���ƫ����ƫС������Ӱ�족�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A������5%NaCl��Һʱ����������NaCl�����ձ��мӼ�����ˮ�����ܽ� |
| B������1mol��L��1NaOH��Һʱ�����ܽ���NaOH��Һ����ע������ƿ |
| C������0��1mol/L��H2SO4��Һʱ������ȡ��ŨH2SO4��������ƿ�м�ˮϡ�� |
| D����Һ����ʱ���Ƚ���Һ©���е��²�Һ��ų���Ȼ���ٽ��ϲ�Һ��ų� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
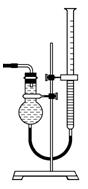
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
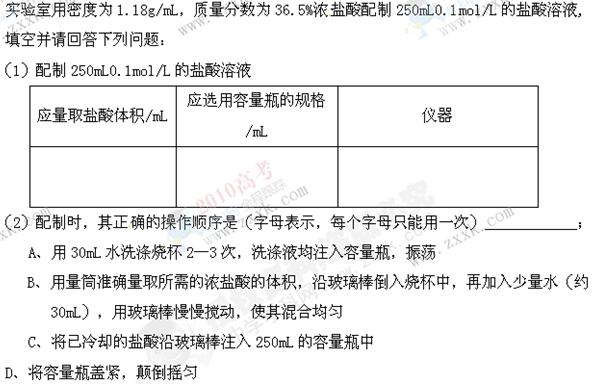
 ��������ƿ��С�ļ�ˮ��ֱ��Һ��ӽ��̶�1��2cm��
��������ƿ��С�ļ�ˮ��ֱ��Һ��ӽ��̶�1��2cm�� ��
���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
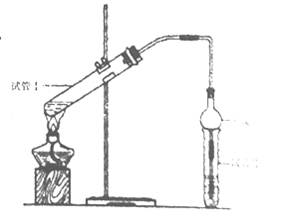
| ʵ���� | �Թܢ��е��Լ� | �Թܢ��е��Լ� | ����л���ĺ��/cm |
| A | 2mL�Ҵ���2 mL���ᡢ1 mL 18mol/LŨ���� | ����̼������Һ | 5.0 |
| B | 3 mL�Ҵ���2 mL���� | 0.1 | |
| C | 3 mL�Ҵ���2 mL���ᡢ6 mL 3mol/L���� | 1.2 | |
| D | 3 mL�Ҵ���2 mL���ᡢ���� | 1.2 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com