
T��ʱ���мס��������ܱ������������������Ϊ1 L�������������Ϊ2 L���ֱ���ס����������м���6 mol A��3 mol B��������Ӧ���£�3A(g)��bB(g) 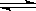 3C(g)��2D(g)����H<0; 4 minʱ�������ڵķ�Ӧǡ�ôﵽƽ�⣬A��Ũ��Ϊ2.4 mol/L��B��Ũ��Ϊ1.8 mol/L; t minʱ�������ڵķ�Ӧ��ƽ�⣬B��Ũ��Ϊ0.8 mol/L.���������Ϣ�ش��������⣺
3C(g)��2D(g)����H<0; 4 minʱ�������ڵķ�Ӧǡ�ôﵽƽ�⣬A��Ũ��Ϊ2.4 mol/L��B��Ũ��Ϊ1.8 mol/L; t minʱ�������ڵķ�Ӧ��ƽ�⣬B��Ũ��Ϊ0.8 mol/L.���������Ϣ�ش��������⣺
(1)�������з�Ӧ��ƽ������v(B)��________����ѧ����ʽ�м�����b��________.
(2)�������з�Ӧ�ﵽƽ��ʱ����ʱ��t________4 min(����ڡ�����С�ڡ����ڡ�)��ԭ����______________________________��
(3)T��ʱ������һ�����������ͬ�ı������У�Ϊ�˴ﵽƽ��ʱB��Ũ����ȻΪ0.8 mol/L����ʼʱ����������м���C��D�����ʵ����ֱ�Ϊ3 mol��2 mol���������A��B�����ʵ����ֱ���________��________.
(4)��Ҫʹ�ס���������B��ƽ��Ũ����ȣ����Բ�ȡ�Ĵ�ʩ��________��
A�������¶Ȳ��䣬����������������2 L
B����������������䣬ʹ�����������¶�
C����������ѹǿ���¶ȶ����䣬����м���һ������A����
D����������ѹǿ���¶ȶ����䣬����м���һ������B����
(5)д��ƽ�ⳣ������ʽK��________����������T��ʱ�Ļ�ѧƽ�ⳣ��K��________.
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
. |
| v |
| t�� | 700 | 800 | 830 | 1000 | 1200 |
| K | 0.6 | 0.9 | 1.0 | 1.7 | 2.6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�Իش��������⣺?
��1������2 min�ڣ���������B�Ļ�ѧ��Ӧ����Ϊ___________����ѧ����ʽ�л�ѧ������b=________��
��2��T ��ʱ������һ�����������ͬ�ı������У���ʼʱ����3 mol C��1 mol D��Ϊʹ�ﵽƽ��ʱB��Ũ����ȻΪ0.6 mol��L -1���������___________mol A��____________mol B��
��3����Ҫʹ�ס���������B��ƽ��Ũ����ȣ����Բ�ȡ�Ĵ�ʩ��____________������ţ���
A.�����¶Ȳ��䣬����������������2 L?
B.��������������䣬ʹ�����������¶�?
C.��������������¶ȶ����䣬��������м���һ������A����?
D.��������������¶ȶ����䣬��������м���һ������B����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010���㽭ʡ���ݶ��и߶��ڶ�ѧ�����п��Ի�ѧ���� ���ͣ������
��6�֣�T��ʱ���мס��������ܱ������������������Ϊ1L�������������Ϊ2L���ֱ���ס����������м���6mol A��3mol B��������Ӧ���£�3A��g����b B��g�� 3C��g����2D��g����
3C��g����2D��g����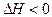 ��4min��������ڵķ�Ӧ�ﵽƽ�⣬A��Ũ��Ϊ2.4mol/L��B��Ũ��Ϊ1.8mol/L��t min���������ڵķ�Ӧ�ﵽƽ�⣬B��Ũ��Ϊ0.8mol/L�������������Ϣ�ش��������⣺
��4min��������ڵķ�Ӧ�ﵽƽ�⣬A��Ũ��Ϊ2.4mol/L��B��Ũ��Ϊ1.8mol/L��t min���������ڵķ�Ӧ�ﵽƽ�⣬B��Ũ��Ϊ0.8mol/L�������������Ϣ�ش��������⣺
��1���������з�Ӧ��ƽ������v��B����_________����ѧ����ʽ�м�����b��________��
��2���������з�Ӧ�ﵽƽ������ʱ��t_________4min������ڡ�����С�ڡ����ڡ�����ԭ����___________________________________________________________��
��3��T��ʱ������һ�����������ͬ�ı������У�Ϊ�˴ﵽƽ��ʱB��Ũ����ȻΪ0.8mol/L����ʼʱ����������м���C��D�����ʵ����ֱ�Ϊ3mol��2mol���������A��B�����ʵ����ֱ���___________��___________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013���½�ũ��ʦ���и߶���ѧ�ڵ�һ�νο��Ի�ѧ�Ծ��������棩 ���ͣ�������
T��ʱ���мס��������ܱ������������������Ϊ1 L�������������Ϊ2 L���ֱ���ס����������м���6 mol A��3 mol B��������Ӧ���£�3A(g)��bB(g) 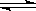 3C(g)��2D(g)����H<0; 4 minʱ�������ڵķ�Ӧǡ�ôﵽƽ�⣬A��Ũ��Ϊ2.4 mol/L��B��Ũ��Ϊ1.8 mol/L; t minʱ�������ڵķ�Ӧ��ƽ�⣬B��Ũ��Ϊ0.8 mol/L.���������Ϣ�ش��������⣺
3C(g)��2D(g)����H<0; 4 minʱ�������ڵķ�Ӧǡ�ôﵽƽ�⣬A��Ũ��Ϊ2.4 mol/L��B��Ũ��Ϊ1.8 mol/L; t minʱ�������ڵķ�Ӧ��ƽ�⣬B��Ũ��Ϊ0.8 mol/L.���������Ϣ�ش��������⣺
(1)�������з�Ӧ��ƽ������v(B)��________����ѧ����ʽ�м�����b��________.
(2)�������з�Ӧ�ﵽƽ��ʱ����ʱ��t________4 min(����ڡ�����С�ڡ����ڡ�)��ԭ����______________________________��
(3)T��ʱ������һ�����������ͬ�ı������У�Ϊ�˴ﵽƽ��ʱB��Ũ����ȻΪ0.8 mol/L����ʼʱ����������м���C��D�����ʵ����ֱ�Ϊ3 mol��2 mol���������A��B�����ʵ����ֱ���________��________.
(4)��Ҫʹ�ס���������B��ƽ��Ũ����ȣ����Բ�ȡ�Ĵ�ʩ��________��
A�������¶Ȳ��䣬����������������2 L
B����������������䣬ʹ�����������¶�
C����������ѹǿ���¶ȶ����䣬����м���һ������A����
D����������ѹǿ���¶ȶ����䣬����м���һ������B����
(5)д��ƽ�ⳣ������ʽK��________����������T��ʱ�Ļ�ѧƽ�ⳣ��K��________.
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010���㽭ʡ�߶��ڶ�ѧ�����п��Ի�ѧ���� ���ͣ������
��6�֣�T��ʱ���мס��������ܱ������������������Ϊ1L�������������Ϊ2L���ֱ���ס����������м���6mol A��3mol
B��������Ӧ���£�3A��g����b B��g�� 3C��g����2D��g����
3C��g����2D��g���� ��4min��������ڵķ�Ӧ�ﵽƽ�⣬A��Ũ��Ϊ2.4mol/L��B��Ũ��Ϊ1.8mol/L��t min���������ڵķ�Ӧ�ﵽƽ�⣬B��Ũ��Ϊ0.8mol/L�������������Ϣ�ش��������⣺
��4min��������ڵķ�Ӧ�ﵽƽ�⣬A��Ũ��Ϊ2.4mol/L��B��Ũ��Ϊ1.8mol/L��t min���������ڵķ�Ӧ�ﵽƽ�⣬B��Ũ��Ϊ0.8mol/L�������������Ϣ�ش��������⣺
��1���������з�Ӧ��ƽ������v��B����_________����ѧ����ʽ�м�����b��________��
��2���������з�Ӧ�ﵽƽ������ʱ��t_________4min������ڡ�����С�ڡ����ڡ�����ԭ����___________________________________________________________��
��3��T��ʱ������һ�����������ͬ�ı������У�Ϊ�˴ﵽƽ��ʱB��Ũ����ȻΪ0.8mol/L����ʼʱ����������м���C��D�����ʵ����ֱ�Ϊ3mol��2mol���������A��B�����ʵ����ֱ���___________��___________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com