
 �����Ծ�ϵ�д�
�����Ծ�ϵ�д� �ο�����������100��ϵ�д�
�ο�����������100��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
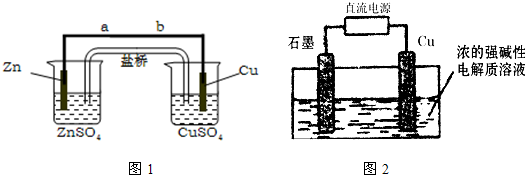
| ʵ�� ��� |
��Ӧ�¶� ������ˮԡ���ȣ� |
ϡ���� ��Һ |
MnSO4���� | 0.10mol/L KMnO4��Һ |
��ɫ ʱ�� |
| 1 | 750�� | ---- | --- | 1mL | 1OOs |
| 2 | 750�� | 10�� | ---- | 1mL | 40s |
| 3 | 750�� | 10�� | �������� | 1mL | 3s |
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1�� |
|
| ����2�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ��ɽ��ʡΫ���и߶�������ҵ��ѧ�������Ծ� ���ͣ������
����ѧƽ���ƶ�ԭ����ͬ��Ҳ����������ƽ��
��1����֪�ڰ�ˮ�д�������ƽ�⣺NH3��H2O NH3��H2O
NH3��H2O  NH��OH��
NH��OH��
��ˮ�м���MgCl2����ʱ��ƽ������ �ƶ���OH����Ũ��
��Ũ��ˮ�м�������NaOH���壬ƽ���� �ƶ�����ʱ�����������ǣߣߣߣߣߣߣߣߣߣߣߡ�
��2���Ȼ���ˮ������ӷ���ʽΪ�ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣ����Ȼ�����Һ�м���̼��Ʒ�ĩ������̼������ܽ⣬��������ɫ���壬�����ӷ���ʽΪ�ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣ�ͬʱ�к��ɫ�������ɣ���ԭ����
��3����Mg(OH)2������Һ�м���NH4Cl��Һ������ ��ԭ
��Ϊ
����ij��Ԫ�� H2A �ĵ��뷽��ʽ�ǣ�H2A=H++HA��HA- A2-+H+���ش��������⣺
A2-+H+���ش��������⣺
��1��H2A�� ���ǿ����ʡ���������ʡ��ǵ���ʡ���
��2��Na2A ��Һ�� ������ԡ��������ԡ������ԡ����������ǣ������ӷ���ʽ��ʾ�� ��
��3��NaHA ��Һ�� ������ԡ��������ԡ������ԡ����������ǣ������ӷ���ʽ��ʾ�� ��
��4���� 0��1mol��L-1NaHA ��Һ�� pH=2���� 0��1mol��L-1 H2A��Һ�������ӵ����ʵ���Ũ�ȿ��� 0.11mol��L����������������������������ǣ� ��
��5��0��1mol��L NaHA��Һ�и�����Ũ���ɴ�С��˳���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��㶫ʡ������ֱ����У�����ڶ���������2�£����ۻ�ѧ�Ծ��������棩 ���ͣ������
���������ھ�������������ˮ��������������ҵ�������������.
��1���������������������ҿ������������е��ʷ�Ӧ����:6Ag(s)+O3(g)=3Ag2O(s)? ��H=-235.8kJ/mol.��֪2Ag2O(s)=4Ag(s)+O2(g)? ��H=+62.2kJ/mol,�����·�Ӧ: 2O3(g)=3O2(g)����H=?????????????????????? .
��2����ѧ��P.Tatapudi��������ʹ�������������µ��ˮ�ķ����Ƶó�����������������Χ��ˮ�в���,�缫��ӦʽΪ3H2O-6e-=O3��+6H+,���������ܽ���ˮ�е��������ɹ�������,��缫��ӦʽΪ???????????????????? ��
��3��O3�ڼ��������¿ɽ�Na2SO4������Na2S2O8��д���÷�Ӧ�Ļ�ѧ����ʽΪ��????????????????????????
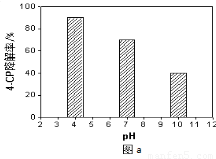 ???????????
??????????? 
��4�����õ�Na2S2O8��Һ�ɽ����л���Ⱦ��4-CP��ԭ����Na2S2O8��Һ��һ�������¿ɲ���ǿ���������ɻ���SO4-������ͨ���ⶨ4-CP�����ʿ��ж�Na2S2O8��Һ����SO4-��������ij�о�С�����ʵ��̽������Һ����ԡ�Fe2+��Ũ�ȶԲ���SO4-����Ӱ�졣
����Һ����Ե�Ӱ�죺����������ͬ����4-CP���뵽��ͬpH��Na2S2O8��Һ�У������ͼa��ʾ���ɴ˿�֪����Һ������ǿ��???????????????? ���� ������������������������Na2S2O8����SO4-����
��Fe2+Ũ�ȵ�Ӱ�죺��ͬ�����£�����ͬŨ�ȵ�FeSO4��Һ�ֱ����c(4-CP)=1.56��10-4 mol��L��1��c(Na2S2O8)=3.12��10-3 mol��L��1�Ļ����Һ�С���Ӧ240 min����ʵ������ͼb��ʾ����֪ S2O82- + Fe2+= SO4-��+ SO42- + Fe3+������ͼʾ��֪����˵����ȷ���ǣ�_________________������ţ�
A.��Ӧ��ʼһ��ʱ���ڣ� 4-CP��������Fe2+Ũ�ȵ������������ԭ����Fe2+��ʹNa2S2O8���������SO4-����
B.Fe2+��4-CP���ⷴӦ�Ĵ���
C.��c(Fe2+)����ʱ��4-CP�����ʷ����½���ԭ�������Fe2+����SO4����������Ӧ�����IJ���SO4������
D.4-CP�����ʷ����½���ԭ����������ɵ�Fe3+ˮ��ʹ��Һ��������ǿ�������ڽ��ⷴӦ�Ľ��С�
����c(Fe2+)��3.2 ��10-3 mol��L��1ʱ��4-CP�����ƽ����Ӧ���ʵļ������ʽΪ??????????????? ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com