
µŚŅ»²½£ŗĮ¬½ÓŗĆ×°ÖĆ£¬¼ģŃéĘųĆÜŠŌ£¬ŌŚŅĒĘ÷ČװČėŅ©Ę·”£
µŚ¶ž²½£ŗĻČČĆŅ»×°ÖĆ·¢Éś·“Ó¦£¬Ö±µ½²śÉśµÄĘųĢå²»ÄÜŌŁŌŚCÖŠČܽāŹ±£¬ŌŁĶØČėĮķŅ»×°ÖĆÖŠ²śÉśµÄĘųĢå£¬Ę¬æĢŗó£¬CÖŠ³öĻÖ¹ĢĢ唣¼ĢŠųĻņCÖŠĶØČėĮ½ÖÖĘųĢ壬ֱµ½²»ŌŁÓŠ¹ĢĢå²śÉś”£
µŚČż²½£ŗ¹żĀĖCÖŠĖłµĆµÄ»ģŗĻĪļ£¬µĆµ½NaHCO3¹ĢĢ唣
µŚĖIJ½£ŗĻņĀĖŅŗÖŠ¼ÓČėŹŹĮæµÄNaCl·ŪÄ©£¬ÓŠNH4Cl¾§ĢåĪö³ö”£””
ĒėĶź³ÉĻĀĮŠĪŹĢā£ŗ
£Ø1£©×°ÖƵÄĮ¬½ÓĖ³ŠņŹĒ£ŗ£Øa£©½Ó£Ø £© £Ø £©½Ó£Ø £©£»£Øb£©½Ó£Ø £©

£Ø2£©AÖŠ³£Ń”ÓĆµÄ¹ĢĢå·“Ó¦ĪļĪŖ____________£»D֊ӦєÓƵÄŅŗĢåĪŖ____________£»BÖŠ·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ_________________________________________________________”£
£Ø3£©µŚ¶ž²½ÖčÖŠ±ŲŠėĻČČĆ_______________×°ÖĆĻČ·¢Éś·“Ó¦”£
£Ø4£©CÖŠÓĆĒņŠĪøÉŌļ¹Ü¶ų²»ÓĆÖ±µ¼¹Ü£¬Ęä×÷ÓĆŹĒ____________________________________£¬CÖŠ¹ćæŚĘæÄŚ²śÉś¹ĢĢåµÄ×Ü»Æѧ·½³ĢŹ½ĪŖ______________________________________”£
£Ø5£©µŚĖIJ½ÖŠ·ÖĄė³öNH4Cl¾§ĢåµÄ²Ł×÷ŹĒ___________________£»ĘäĖłµĆµÄNH4Cl¾§ĢåÖŠ³£ŗ¬ÓŠÉŁĮæµÄNaClŗĶNaHCO3£ØŌ¼Õ¼5%”Ŗ8%£©£¬ĒėÉč¼ĘŅ»øö¼ņµ„µÄŹµŃéÖ¤Ć÷ĖłµĆ¹ĢĢåµÄ³É·Ö“ó²æ·ÖŹĒNH4Cl”£¼ņŅŖŠ“³ö²Ł×÷ŗĶĻÖĻó£ŗ______________________________________”£
£Ø1£©f e d c
£Ø2£©æéדŹÆ»ŅŹÆ ±„ŗĶNaHCO3ČÜŅŗ CaO+NH3”¤H2O====Ca£ØOH£©2+NH3”ü£ØĘäĖūŗĻĄķ“š°ø¾łæÉ£©
£Ø3£©B
£Ø4£©·Ąµ¹Īü CO2+NH3+NaCl+H2O====NaHCO3”ż+NH4Cl
£Ø5£©¹żĀĖ ȔɣĮæ¹ĢĢå·ÅČėŹŌ¹ÜÖŠ£¬¼ÓČČ£¬¹ĢĢå“ó²æ·ÖĻūŹ§£¬ŌŚŹŌ¹ÜæŚÓÖÓŠ½Ļ¶ąµÄ¹ĢĢåÄż½į£ØĘäĖūŗĻĄķ“š°ø¾łæÉ£©
½āĪö£ŗÓÉĢāŅāæÉÖŖ£¬ŠčÖʵĆNH3ŗĶCO2Į½ÖÖĘųĢå£¬Č»ŗó½«Ęä·ÖĻČŗóĶØČė±„ŗĶŹ³ŃĪĖ®ÖŠ£¬¼“æÉ·¢Éś·“Ó¦£¬Éś³ÉĢ¼ĖįĒāÄĘ³Įµķ”£ČōĻČĶضžŃõ»ÆĢ¼ĘųĢ壬ĘäŌŚ±„ŗĶŹ³ŃĪĖ®ÖŠµÄČܽā¶Č²»“ó£¬ŌŁĶØ°±ĘųŗóÉś³ÉµÄĢ¼ĖįĒāÄʵÄĮæÉŁ”£¹ŹÓ¦ĻČĶØ°±Ęų£¬ŗóĶضžŃõ»ÆĢ¼”£
ŌŚÓĆŃĪĖįÓė“óĄķŹÆÖʵƵĶžŃõ»ÆĢ¼ĘųĢåÖŠ£¬»ģÓŠĀČ»ÆĒāĘųĢ壬Ėü»įÓėĢ¼ĖįĒāÄĘ·“Ó¦£¬Ó°ĻģĢ¼ĖįĒāÄĘµÄ²śĮ棬ĖłŅŌÓ¦³żČ„¶žŃõ»ÆĢ¼ÖŠ»ģÓŠµÄĀČ»ÆĒā£¬Ņ²¾ĶŹĒÓ¦½«ĘųĢåĻČĶØČė±„ŗĶĢ¼ĖįĒāÄĘČÜŅŗÖŠ”£
ÓÉÓŚ°±Ęų¼«Ņ×ČÜÓŚĖ®£¬ĪŖ·Ąµ¹Īü£¬Ó¦½«µ¼¹ÜæŚøÕ½Ó“„Ė®Ćę£¬²¢²ÉÓĆøÉŌļ¹Ü½ųŅ»²½¼ÓŅŌŌ¤·Ą”£
ĀČ»Æļ§µÄŹµŃéÖ¤Ć÷³£²ÉÓĆ¼ÓČČ·Ö½ā·Ø£¬æ“Ź£Óą²śĪļµÄ¶ąÉŁ£¬“Ó¶ųĖµĆ÷Ęäŗ¬ĮæµÄ¶ąÉŁ”£



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
| ||

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
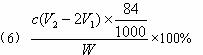
ĖÄÖÖŃĪŌŚ²»Ķ¬ĪĀ¶ČĻĀµÄČܽā¶Č£Øg/100 gĖ®£©±ķ

(ĖµĆ÷£ŗ¢Ł£¾35 ”ę NH4HCO3»įÓŠ·Ö½ā£©
Ēė»Ų“šŅŌĻĀĪŹĢā£ŗ
£Ø1£©×°ÖƵÄĮ¬½ÓĖ³ŠņÓ¦ŹĒ_________________________£ØĢī×ÖÄø£©”£
£Ø2£©A×°ÖĆÖŠŹ¢·ÅµÄŹŌ¼ĮŹĒ_________£¬Ęä×÷ÓĆŹĒ____________________________________”£
£Ø3£©ŌŚŹµŃé¹ż³ĢÖŠ£¬ŠčŅŖæŲÖĘDĪĀ¶ČŌŚ30”Ŗ35 ”ę£¬ŌŅņŹĒ___________________________”£ĪŖĮĖ±ćÓŚæŲÖĘ“ĖĪĀ¶Č·¶Ī§£¬²ÉČ”µÄ¼ÓČČ·½·ØĪŖ____________________________________”£
£Ø4£©·“Ó¦½įŹųŗ󣬽«×¶ŠĪĘ潞ŌŚĄäĖ®ÖŠ£¬Īö³öNaHCO3¾§ĢåµÄŌŅņŹĒ____________”£ÓĆÕōĮóĖ®Ļ“µÓNaHCO3¾§ĢåµÄÄæµÄŹĒ³żČ„___________________________ŌÓÖŹ£ØŅŌ»ÆѧŹ½±ķŹ¾£©”£
£Ø5£©½«×¶ŠĪĘæÖŠµÄ²śĪļ¹żĀĖŗó£¬ĖłµĆµÄÄøŅŗÖŠŗ¬ÓŠ______________£ØŅŌ»ÆѧŹ½±ķŹ¾£©£¬æɼÓČėĀČ»ÆĒā£¬²¢½ųŠŠ_________²Ł×÷£¬Ź¹NaClČÜŅŗŃ»·Ź¹ÓĆ£¬Ķ¬Ź±æÉ»ŲŹÕNH4Cl”£
£Ø6£©²āŹŌ“æ¼ī²śĘ·ÖŠNaHCO3ŗ¬ĮæµÄ·½·ØŹĒ£ŗ×¼Č·³ĘČ”“æ¼īѳʷW g£¬·ÅČė׶ŠĪĘæÖŠ¼ÓÕōĮóĖ®Čܽā£¬¼Ó1”Ŗ2µĪ·ÓĢŖÖøŹ¾¼Į£¬ÓĆĪļÖŹµÄĮæÅضČĪŖc( mol”¤L-1)µÄHClČÜŅŗµĪ¶ØÖĮČÜŅŗÓÉŗģÉ«µ½ĪŽÉ«£ØÖøŹ¾![]() +H+====
+H+====![]() ·“Ó¦µÄÖÕµć£©£¬ĖłÓĆHClČÜŅŗĢå»żĪŖV1 mL£¬ŌŁ¼Ó1”Ŗ2µĪ¼×»ł³ČÖøŹ¾¼Į£¬¼ĢŠųÓĆHClČÜŅŗµĪ¶ØÖĮČÜŅŗÓɻʱä³Č£¬ĖłÓĆHClČÜŅŗ×ÜĢå»żĪŖV2 mL”£Š“³ö“æ¼īѳʷ֊NaHCO3ÖŹĮæ·ÖŹżµÄ¼ĘĖćŹ½£ŗw(NaHCO3)=_________________”£
·“Ó¦µÄÖÕµć£©£¬ĖłÓĆHClČÜŅŗĢå»żĪŖV1 mL£¬ŌŁ¼Ó1”Ŗ2µĪ¼×»ł³ČÖøŹ¾¼Į£¬¼ĢŠųÓĆHClČÜŅŗµĪ¶ØÖĮČÜŅŗÓɻʱä³Č£¬ĖłÓĆHClČÜŅŗ×ÜĢå»żĪŖV2 mL”£Š“³ö“æ¼īѳʷ֊NaHCO3ÖŹĮæ·ÖŹżµÄ¼ĘĖćŹ½£ŗw(NaHCO3)=_________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
µŚŅ»²½£ŗĮ¬½ÓŗĆ×°ÖĆ£¬¼ģŃéĘųĆÜŠŌ£¬ŌŚŅĒĘ÷ČװČėŅ©Ę·”£
µŚ¶ž²½£ŗĻČČĆŅ»×°ÖĆ·¢Éś·“Ó¦£¬Ö±µ½²śÉśµÄĘųĢå²»ÄÜŌŁŌŚCÖŠČܽāŹ±£¬ŌŁĶØČėĮķŅ»×°ÖĆÖŠ²śÉśµÄĘųĢå£¬Ę¬æĢŗó£¬CÖŠ³öĻÖ¹ĢĢ唣¼ĢŠųĻņCÖŠĶØČėĮ½ÖÖĘųĢ壬ֱµ½²»ŌŁÓŠ¹ĢĢå²śÉś”£
µŚČż²½£ŗ¹żĀĖCÖŠĖłµĆµÄ»ģŗĻĪļ£¬µĆµ½NaHCO3¹ĢĢ唣
µŚĖIJ½£ŗĻņĀĖŅŗÖŠ¼ÓČėŹŹĮæµÄNaCl·ŪÄ©£¬ÓÖÓŠ¾§ĢåĪö³ö£¬””
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ

(1)×°ÖƵÄĮ¬½ÓĖ³ŠņŹĒ£ŗ(a)½Ó(_________)![]() (_________)½Ó(_________)£»
(_________)½Ó(_________)£»
(b)½Ó(_________)”£
(2)AÖŠ³£Ń”ÓĆµÄ¹ĢĢå·“Ó¦ĪļĪŖ_________£»D֊ӦєÓƵÄŅŗĢåĪŖ_________”£
(3)B×°ÖĆÖŠ·ÖŅŗĀ©¶·ÄŚĖł¼ÓŹŌ¼ĮĪŖ_________£»µŚ¶ž²½ÖŠ±ŲŠėĻČČĆ_________×°ÖĆ·¢Éś·“Ó¦”£cÖŠÓĆĒņŠĪøÉŌļ¹Ü¶ų²»ÓĆÖ±µ¼¹Ü£¬Ęä×÷ÓĆŹĒ_________”£
(4)µŚĖIJ½·ÖĄėĖłµĆµÄ¾§ĢåÖŠŗ¬ÓŠ¶ąÖֳɷ֣¬ŹŌÖ¤Ć÷ĘäÖŠŗ¬ÓŠNH4CI£¬¼ņŅŖŠ“³ö²Ł×÷¹ż³ĢŗĶĻÖĻó____________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012½ģøßČżÉĻѧʌµ„ŌŖ²āŹŌ£Ø6£©»ÆѧŹŌ¾ķ£ØŠĀČĖ½Ģ°ę£© ĢāŠĶ£ŗŹµŃéĢā
£Ø10·Ö£©ĪŅ¹ś»Æ¹¤×ؼŅŗīµĀ°ńµÄ”°ŗīŹĻÖĘ¼ī·Ø”±ŌųĪŖŹĄ½ēÖĘ¼ī¹¤Ņµ×ö³öĮĖĶ»³ö¹±Ļ×”£ĖūĄūÓĆNaHCO3”¢NaCl”¢N H4C1µČĪļÖŹ
H4C1µČĪļÖŹ Čܽā¶ČµÄ²īŅģ£¬ŅŌŹ³
Čܽā¶ČµÄ²īŅģ£¬ŅŌŹ³ ŃĪ”¢°±Ęų”¢¶žŃõ»ÆĢ¼µČĪŖŌĮĻÖʵĆNaHCO3£¬½ų¶ųÉś²ś³ö“æ¼ī”£ŅŌĻĀA”¢B”¢C”¢DĖÄøö×°ÖĆæÉ×é×°³ÉŹµŃéŹŅÄ£Äā ”°ŗīŹĻÖĘ¼ī·Ø”±ÖĘČ”NaHCO3µÄŹµŃé×°ÖĆ”£×°ÖĆÖŠ·Ö±šŹ¢ÓŠŅŌĻĀŹŌ¼Į£ŗB£ŗĻ”ĮņĖį£»C£ŗŃĪĖį”¢Ģ¼
ŃĪ”¢°±Ęų”¢¶žŃõ»ÆĢ¼µČĪŖŌĮĻÖʵĆNaHCO3£¬½ų¶ųÉś²ś³ö“æ¼ī”£ŅŌĻĀA”¢B”¢C”¢DĖÄøö×°ÖĆæÉ×é×°³ÉŹµŃéŹŅÄ£Äā ”°ŗīŹĻÖĘ¼ī·Ø”±ÖĘČ”NaHCO3µÄŹµŃé×°ÖĆ”£×°ÖĆÖŠ·Ö±šŹ¢ÓŠŅŌĻĀŹŌ¼Į£ŗB£ŗĻ”ĮņĖį£»C£ŗŃĪĖį”¢Ģ¼ ĖįøĘ£»D£ŗŗ¬°±µÄ±„ŗĶŹ³ŃĪĖ®”¢Ė®
ĖįøĘ£»D£ŗŗ¬°±µÄ±„ŗĶŹ³ŃĪĖ®”¢Ė®
ĖÄÖÖŃĪŌŚ²»Ķ¬ĪĀ¶ČĻĀµÄČܽā¶Č£Øg£Æ100gĖ®£©±ķ
| | 0”ę | 10”ę | 20”ę | 30”ę | 40”ę | 50”ę | 60”ę | 100”ę |
| NaCl | 35£®7 | 35£®8 | 36£®0 | 36_3 | 36£®6 | 37£®0 | 37£®3 | 39£®8 |
| NH4HCO3 | 11£®9 | 15£®8 | 21£®0 | 27£®0 | ”Ŗ¢Ł | ”Ŗ | ”Ŗ | ”Ŗ |
| NaHCO3 | 6£®9 | 8£®1 | 9£®6 | 11£®1 | 12£®7 | 14£®5 | 16£®4 | |
| NH4Cl | 29£®4 | 33£®3 | 37£®2[ Ą“Ō“:Z|xx|k.Com] Ą“Ō“:Z|xx|k.Com] | 41£®4 | 45£®8[Ą“Ō“:Z*xx*k.Com] | 50£®4 | 55£®3 | 77£®3 |
 5£©½«×¶ŠĪĘæÖŠµÄ²śĪļ¹żĀĖŗó£¬ĖłµĆµÄÄøŅŗÖŠŗ¬ÓŠ £ØŅŌ»ÆѧŹ½±ķŹ¾£©£¬¼ÓČėĀČ»ÆĒā£¬²¢½ųŠŠ ²Ł×÷£¬Ź¹NaClČÜŅŗŃ»·Ź¹ÓĆ£¬Ķ¬Ź±æÉ»ŲŹÕNH4C1”£
5£©½«×¶ŠĪĘæÖŠµÄ²śĪļ¹żĀĖŗó£¬ĖłµĆµÄÄøŅŗÖŠŗ¬ÓŠ £ØŅŌ»ÆѧŹ½±ķŹ¾£©£¬¼ÓČėĀČ»ÆĒā£¬²¢½ųŠŠ ²Ł×÷£¬Ź¹NaClČÜŅŗŃ»·Ź¹ÓĆ£¬Ķ¬Ź±æÉ»ŲŹÕNH4C1”£²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012½ģĖÄ“ØŹ”׏֊ĻŲøßČżĮćÄ£æ¼ŹŌĄķ×ŪŹŌĢā£Ø»Æѧ²æ·Ö£© ĢāŠĶ£ŗŹµŃéĢā
£Ø14·Ö£©ĪŅ¹ś»Æ¹¤×ؼŅŗīµĀ°ńµÄ”°ŗīŹĻÖĘ¼ī·Ø”±ŌųĪŖŹĄ½ēÖĘ¼ī¹¤Ņµ×ö³öĮĖĶ»³ö¹±Ļ×”£ĖūĄūÓĆNaHCO3”¢NaCl”¢NH4ClµČĪļÖŹČܽā¶ČµÄ²īŅģ£¬ŅŌŹ³ŃĪ”¢°±Ęų”¢¶žŃõ»ÆĢ¼µČĪŖŌĮĻĻČÖʵĆNaHCO3£¬½ų¶ųÉś²ś³ö“æ¼ī”£ĻĀĆęŹĒŌŚŹµŃéŹŅÖŠÄ£Äā”°ŗīŹĻÖĘ¼ī·Ø”±ÖĘČ”NaHCO3µÄŹµŃé²½Öč£ŗ
µŚŅ»²½£ŗĮ¬½ÓŗĆ×°ÖĆ£¬¼ģŃéĘųĆÜŠŌ£¬ŌŚŅĒĘ÷ČװČėŅ©Ę·”£
µŚ¶ž²½£ŗĻČČĆŅ»×°ÖĆ·¢Éś·“Ó¦£¬Ö±µ½²śÉśµÄĘųĢå²»ÄÜŌŁŌŚCÖŠČܽāŹ±£¬ŌŁĶØČėĮķŅ»×°ÖĆÖŠ²śÉśµÄĘųĢå£¬Ę¬æĢŗó£¬CÖŠ³öĻÖ¹ĢĢ唣¼ĢŠųĻņCÖŠĶØČėĮ½ÖÖĘųĢ壬ֱµ½²»ŌŁÓŠ¹ĢĢå²śÉś”£
µŚČż²½£ŗ¹żĀĖCÖŠĖłµĆµÄ»ģŗĻĪļ£¬µĆµ½NaHCO3¹ĢĢ唣
µŚĖIJ½£ŗĻņĀĖŅŗÖŠ¼ÓČėŹŹĮæµÄNaCl·ŪÄ©£¬ÓŠNH4Cl¾§ĢåĪö³ö”£””
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
(1)×°ÖƵÄĮ¬½ÓĖ³ŠņŹĒ£ŗ(a)½Ó( ) ( )½Ó( )£»(b)½Ó( )
(2)AÖŠ³£Ń”ÓĆµÄ¹ĢĢå·“Ó¦ĪļĪŖ____________£»D֊ӦєÓƵÄŅŗĢåĪŖ___________ £ØĢī»ÆѧŹ½£©
(3)µŚ¶ž²½ÖčÖŠ±ŲŠėĻČČĆ________×°ÖĆĻČ·¢Éś·“Ó¦”£
(4)CÖŠÓĆĒņŠĪøÉŌļ¹Ü¶ų²»ÓĆÖ±µ¼¹Ü£¬Ęä×÷ÓĆŹĒ____________________________£¬CÖŠ¹ćæŚĘæÄŚ²śÉś¹ĢĢåµÄ×Ü»Æѧ·½³ĢŹ½ĪŖ ”£
(5)ŌŚµŚĖIJ½ÖŠ·ÖĄėNH4Cl¾§ĢåµÄ²Ł×÷ŹĒ________________£ØĢī²Ł×÷Ćū³Ę£©£»ĘäĖłµĆµÄNH4Cl¾§ĢåÖŠ³£ŗ¬ÓŠÉŁĮæµÄNaClŗĶNaHCO3Ō¼Õ¼5%”Ŗ8%£©£¬ĒėÉč¼ĘŅ»øö¼ņµ„µÄŹµŃéÖ¤Ć÷ĖłµĆ¹ĢĢåµÄ³É·ÖÖŠŗ¬ÓŠNa£«”£¼ņŅŖŠ“³ö²Ł×÷ŗĶĻÖĻó ”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com