
 4NO +6H2O£Ø2·Ö£©
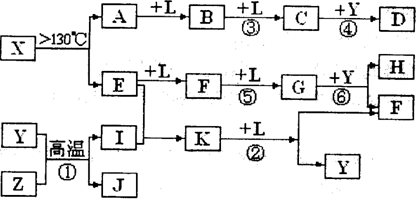
4NO +6H2O£Ø2·Ö£© Fe3O4+4H2£¬ZŹĒFe£¬IŹĒH2”£H”¢K¾łŹĒÖŲŅŖµÄ¹¤Ņµ²śĘ·£¬“ÓE”¢F”¢GÖ®¼äµÄ×Ŗ»ÆĢŲÕ÷£¬æÉÖŖEŹĒN2£¬N2ÓėH2·“Ӧɜ³ÉK£¬KŹĒNH3£¬·“Ó¦¢ŚŹĒNH3ÓėO2µÄ“ß»ÆŃõ»Æ£¬Éś³ÉNOŗĶH2O£¬·ūŗĻæņĶ¼£¬FŹĒNO£¬NOÓėO2¼ĢŠų·“Ӧɜ³ÉNO2£¬GŹĒNO2£¬NO2ÓėĖ®·“Ӧɜ³ÉĻõĖįŗĶNO£¬HŹĒĻõĖį”£×īŗóÓÉAŗĶE£¬ŅŌ¼°XŹĒ½į¹¹ÓŠ“żĢ½¾æµÄŅ»ÖÖŠĀŠĶ·Ö×Ó£¬Ęä×é³ÉŌŖĖŲµÄĻą¶ŌŌ×ÓÖŹĮæĻą²ī18,æɵĆXŹĒS4N4”£
Fe3O4+4H2£¬ZŹĒFe£¬IŹĒH2”£H”¢K¾łŹĒÖŲŅŖµÄ¹¤Ņµ²śĘ·£¬“ÓE”¢F”¢GÖ®¼äµÄ×Ŗ»ÆĢŲÕ÷£¬æÉÖŖEŹĒN2£¬N2ÓėH2·“Ӧɜ³ÉK£¬KŹĒNH3£¬·“Ó¦¢ŚŹĒNH3ÓėO2µÄ“ß»ÆŃõ»Æ£¬Éś³ÉNOŗĶH2O£¬·ūŗĻæņĶ¼£¬FŹĒNO£¬NOÓėO2¼ĢŠų·“Ӧɜ³ÉNO2£¬GŹĒNO2£¬NO2ÓėĖ®·“Ӧɜ³ÉĻõĖįŗĶNO£¬HŹĒĻõĖį”£×īŗóÓÉAŗĶE£¬ŅŌ¼°XŹĒ½į¹¹ÓŠ“żĢ½¾æµÄŅ»ÖÖŠĀŠĶ·Ö×Ó£¬Ęä×é³ÉŌŖĖŲµÄĻą¶ŌŌ×ÓÖŹĮæĻą²ī18,æɵĆXŹĒS4N4”£ 4NO +6H2O”££Ø3£©NO2”¢NO»ģŗĻĘųĢåĶØČė×ćĮæÉÕ¼īČÜŅŗ£¬ĘųĢåČ«²æ±»ĪüŹÕ£¬×Ŗ»ÆĪŖĻõĖįŃĪŗĶŃĒĻõĖįŃĪ£¬Ö»ŅŖNO2µÄĪļÖŹµÄĮæ“óÓŚNOµÄĪļÖŹµÄĮæ¾ĶÄÜČ«²æĪüŹÕ£¬¼“n(NO2)£ŗn(NO)”Ż1£ŗ1£¬·¢ÉśµÄ·“Ó¦ĪŖ2NO2+2NaOH=NaNO2+NaNO3+H2O”¢NO2+NO+2NaOH=2NaNO2+H2O”££Ø4£©Fe3O4Óė¹żĮæµÄĻ”ĻõĖį·“Ӧɜ³ÉĻõĖįĢś”¢NOŗĶĖ®£¬Ąė×Ó·½³ĢŹ½ĪŖ3Fe3O4+28H++NO3-=9Fe3++ NO”ü+14H2O”££Ø5£©ŗĻ³É°±Õż·“Ó¦ŹĒŅ»øöĢå»żĖõŠ”µÄ·“Ó¦£¬Ģå»ż¼õ°ė£¬Ļąµ±ÓŚŃ¹ĒæŌö“óŅ»±¶£¬Ń¹ĒæŌö“ó£¬Ę½ŗāĻņÕż·“Ó¦·½ĻņŅĘ¶Æ£¬NH3µÄĢå»ż·ÖŹżŌö“ó”£
4NO +6H2O”££Ø3£©NO2”¢NO»ģŗĻĘųĢåĶØČė×ćĮæÉÕ¼īČÜŅŗ£¬ĘųĢåČ«²æ±»ĪüŹÕ£¬×Ŗ»ÆĪŖĻõĖįŃĪŗĶŃĒĻõĖįŃĪ£¬Ö»ŅŖNO2µÄĪļÖŹµÄĮæ“óÓŚNOµÄĪļÖŹµÄĮæ¾ĶÄÜČ«²æĪüŹÕ£¬¼“n(NO2)£ŗn(NO)”Ż1£ŗ1£¬·¢ÉśµÄ·“Ó¦ĪŖ2NO2+2NaOH=NaNO2+NaNO3+H2O”¢NO2+NO+2NaOH=2NaNO2+H2O”££Ø4£©Fe3O4Óė¹żĮæµÄĻ”ĻõĖį·“Ӧɜ³ÉĻõĖįĢś”¢NOŗĶĖ®£¬Ąė×Ó·½³ĢŹ½ĪŖ3Fe3O4+28H++NO3-=9Fe3++ NO”ü+14H2O”££Ø5£©ŗĻ³É°±Õż·“Ó¦ŹĒŅ»øöĢå»żĖõŠ”µÄ·“Ó¦£¬Ģå»ż¼õ°ė£¬Ļąµ±ÓŚŃ¹ĒæŌö“óŅ»±¶£¬Ń¹ĒæŌö“ó£¬Ę½ŗāĻņÕż·“Ó¦·½ĻņŅĘ¶Æ£¬NH3µÄĢå»ż·ÖŹżŌö“ó”£


| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
 £»
£»²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
A£® | B£®4.7g | C£®7.4g | D£® g g |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com