
| װ�� | ���� | ���ۼ����� |
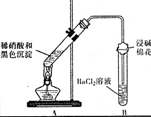 | ��A�Թ��к�ɫ�������ܽ� ��A�Թ��Ϸ����ֺ���ɫ���� ��B�Թ��г��ְ�ɫ���� | a�������˵����ɫ�������� ��ԭ�ԣ� b���Թ�B�в�����ɫ�������ܷ�Ӧ�����ӷ���ʽΪ NO2+SO2+Ba2++H2O=BaSO4��+NO+2H+ |
���� ��1������ͭ��Ũ���ᷴӦ���ɶ�������������н��
��2������������Ϣ�м���ͭ���ӵķ����Ԣڽ��з�����Ȼ��ó���ȷ���ۣ�
��3��a������ɫ����Ϊ����������˵��ϡ���ᱻ��ԭ����һ����������ɫ������л�ԭ�ԣ�
b�����ݷ�Ӧ����ۿ�֪��ɫ������ϡ���ᷴӦ�����˶�������֤����ɫ�����к�����Ԫ�أ�������������������Ļ�������ܹ����Ȼ�����Ӧ�������ᱵ�������ݴ�д����Ӧ�����ӷ���ʽ��
��4��CuS����Һ�д��ڳ����ܽ�ƽ�⣬����ƽ���ƶ�������
��5�����ݵζ�ʵ�����ݼ���ʣ�����������ʵ������õ�������ͭ����ͭ��Ӧ�ĸ���������ʵ��������ݷ�Ӧ�����ӷ���ʽ��ʽ����õ�����
��� �⣺��1��Cu��Ũ���ᷴӦ��������ͭ�����������ˮ����ӦΪCu+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+2SO2��+2H2O������������������SO2��
�ʴ�Ϊ��SO2��
��2������Һ�еμ�K4[Fe��CN��6]��Һ�����������ɫ������֤����Cu2+�����ݢڽ���ɫ��������ϡ�����У�һ��ʱ��μ�K4[Fe��CN��6]��Һ��δ�����ɫ������֪����ɫ������һ������CuO��
�ʴ�Ϊ����ɫ�����в�����CuO��
��3��a��A�Թ����Ϸ����ֺ���ɫ���壬˵����Ӧ����һ���������ɣ�֤���˺�ɫ������л�ԭ�ԣ��ڷ�Ӧ�б�������
�ʴ�Ϊ����ԭ�ԣ�
b�����ݷ�Ӧ�����B�Թ��г��ְ�ɫ������֪����ɫ����Ϊ���ᱵ��˵����ɫ�����к�����Ԫ�أ�������Ӧ�����ӷ���ʽΪ��NO2+SO2+Ba2++H2O�TBaSO4��+NO��+2H+��
�ʴ�Ϊ��B�Թ��г��ְ�ɫ������NO2+SO2+Ba2++H2O�TBaSO4��+NO��+2H+��
��4��CuS������ˮ����ˮ��Һ�л��к�������Cu�ܽ⣬��Һ�д��ڳ����ܽ�ƽ�⣬CuS��s��?Cu2+��aq��+S2-��aq�����ȵ�Ũ���ὫS2-������ʹS2-Ũ�ȼ�С���ٽ�����ƽ���������ƶ���ʹCuS�ܽ⣻
�ʴ�Ϊ��CuS�����ܽ�ƽ��CuS��s��?Cu2+��aq��+S2-��aq�����ȵ�Ũ���ὫS2-������ʹS2-Ũ�ȼ�С���ٽ�����ƽ���������ƶ���ʹCuS�ܽ⣻
��5�������ķ�ӦΪ��
8MnO4-+5Cu2S+44H+�T10Cu2++5SO2��+8Mn2++22H2O
6MnO4-+5CuS+28H+�T5Cu2++5SO2��+6Mn2++14H2O
MnO4-+5Fe2++8H+�TMn2++5Fe3++4H2O
��Cu2S��CuS�����ʵ����ֱ�Ϊx��y��
��Cu2S��CuS��Ӧ��ʣ��KMnO4�����ʵ�����0.035L��0.1mol/L��$\frac{1}{5}$=0.0007mol��
160x+96y=0.2
$\frac{8x}{5}$+$\frac{6y}{5}$=0.04��0.075-0.0007
���x=0.0005mol��
Cu2S������������$\frac{0.0005mol��160g/mol}{0.2g}$��100%=40%��
�ʴ�Ϊ��40%��
���� ���⿼����Ũ����Ļ�ѧ���ʡ�����ʵ�鷽������ƣ���Ŀ�Ѷ��еȣ������漰�������Դ�֪ʶ��϶࣬����������Ϣ�ǽ���ؼ�������������ѧ���ķ���������������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
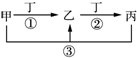 �ס��ҡ�����������ѧ���������ʣ����мס��ҡ���������ͬһ��Ԫ�أ���һ�������µ�ת����ϵ��ͼ������˵����ȷ���ǣ�������
�ס��ҡ�����������ѧ���������ʣ����мס��ҡ���������ͬһ��Ԫ�أ���һ�������µ�ת����ϵ��ͼ������˵����ȷ���ǣ�������| A�� | ����Ϊ���������;���Ľ������ʣ��ҵ���Һһ��ΪFeCl3 | |
| B�� | ��ͨ������¼ס��ҡ��������������壬���ҺͶ�Ϊ��������Ҫ�ɷ֣���Ӧ�ٵĻ�ѧ����ʽΪ4NH3+5O2$\frac{\underline{\;����\;}}{��}$4NO+6H2O | |
| C�� | ���ס��ҡ�������Һ�Լ��ԣ�������ܿ�����Ϊҽ��������θ�����֢��ҩ�� | |
| D�� | ����Ϊ�������Ϊ�ȼҵ����Ҫ��Ʒ�����һ��Ϊ��Al3+���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| Ԫ�ش��� | L | M | Q | R | T |
| ԭ�Ӱ뾶/nm | 0.160 | 0.143 | 0.089 | 0.104 | 0.066 |
| ��Ҫ���ϼ� | +2 | +3 | +2 | +6��-2 | -2 |
| A�� | �⻯��ķе�ΪH2T��H2R | B�� | ������ϡ���ᷴӦ�Ŀ���ΪL��Q | ||
| C�� | M��T�γɵĻ����������ӻ����� | D�� | L2+��R2-�ĺ����������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
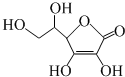
| A�� | ά����C�����Ĺ��������ǻ����Ȼ���̼̼˫�� | |
| B�� | ά����C�ܺ���ˮ�������ظ������Һ��Ӧ | |
| C�� | ά����C�ķ���ʽΪC6H6O6 | |
| D�� | ά����C�ܷ����ӳɷ�Ӧ��������Ӧ�����ܷ���ȡ����Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
| A�� | 3.0 L 0.1 mol•L-1 NaOH��Һ�л���ͨ��CO2����Һ����8.8 gʱ����Һ�У�c��Na+����c��CO32-����c��HCO3-����c��OH-����c��H+�� | |
| B�� | �����£���CH3COONa��Һ��ϡ����������ҺpH=7�� c��Na+����c��CH3COO-����c��Cl-��=c��CH3COOH����c��H+��=c��OH-�� | |
| C�� | �����£�pH=3�Ĵ�����Һ��pH=13������������Һ�������Ϻ����ǻ�Ϻ���Һ����ı仯����ǡ����ȫ��Ӧ����ԭ������Һ����ĵ����Ϊ1% | |
| D�� | ���ʵ���Ũ��֮��Ϊ1��2��NaClO��NaHCO3�����Һ�У�c��HClO��+c��ClO-��=2c��HCO3-��+2c��H2CO3��+2c��CO32-�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
| ���� | K+ | Na+ | NH4+ | SO42- | NO3- | Cl- |
| Ũ��/mol•L-1 | 4��10-6 | 6��10-6 | 2��10-5 | 4��10-5 | 3��10-5 | 2��10-5 |
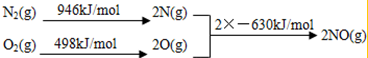
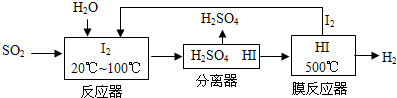
| �¶�/�� | 0 | 100 | 200 | 300 | 400 |
| ƽ�ⳣ�� | 667 | 13 | 1.9��10-2 | 2.4��10-4 | 1��10-5 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ����ϩʳƷ��װ����ʳ�ﱣ��Ĥ�������ĸ߷��Ӳ��� | |
| B�� | ������ɱ�����в�������Ϊ���ɲ����ĵ��������ȱ��� | |
| C�� | ̫���ܵ�ذ��еĹ��ǹ赥�ʣ����ά����Ҫ�ɷ��Ƕ������� | |
| D�� | ����[KAl��SO4��2•12H2O]����������ˮ�ľ�����ɱ������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| ���� | ���� | �Լ� | �ᴿ���� | |
| A | �� | ���� | ��ˮ | ���� |
| B | ������̼ | �������� | ����̼������Һ | ϴ�� |
| C | �������� | ���� | ϡ����������Һ | ��������÷�Һ |
| D | ������ | ���� | �����������Һ | ���������ˡ�ϴ�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com