
��ҵ����ij����(����Cu2O��Al2O3��Fe2O3��SiO2)��ȡͭ�IJ����������£�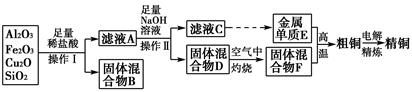
��֪��Cu2O��2H��=Cu��Cu2����H2O
(1)ʵ������������Ϊ________���ڿ��������չ�������Dʱ���õ����ֹ������ʵ������������������ƾ��ơ��������⣬����________(����������)��
(2)��ҺA����Ԫ�صĴ�����ʽΪ________(�����ӷ���)�����ɸ����ӵ����ӷ���ʽΪ____________________________________________��������ҺA�д��ڸ����ӵ��Լ�Ϊ________(���Լ�����)��
(3)��������E���������F������ijһ��Ӧ�����ں��Ӹֹ죬�÷�Ӧ�Ļ�ѧ����ʽΪ__________________________________________________��
(4)�����£���pH��NaAlO2��NaOH������Һ�У���ˮ�������c(OH��)ǰ��Ϊ���ߵ�108������������Һ��pH��________��
(5)��Ũ���ᡢŨ���ᡢ����ˮ��ѡ�ú��ʵ��Լ����ⶨ��ͭ��Ʒ�н���ͭ�������������漰����Ҫ���裺��ȡһ����������Ʒ��________________�����ˡ�ϴ�ӡ����������ʣ�����ͭ��������(��ȱ�ٵIJ������裬���������������̵�ϸ��)

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����������������������������������������������ȣ�Ϊԭ���Ʊ�����{�ɱ�ʾΪFe2(OH)n(SO4)3-n/2�����̷�(FeSO4.7H2O)���гɱ��͡���Ӧ�졢��Ʒ�����ߵ��ŵ㡣�Ʊ���������ͼ��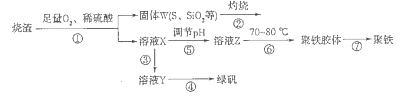
��1������ͼ����ҺXΪFe(SO4)3����Һ���ݴ�д��������������������ԭ��Ӧ�����ӷ���ʽ��____________��
��2�������ڵĴ������ڱˣ��ñ���_____________��������W��KOH��Һ���ϼ��ȣ�����һ����Ӧ�Ļ�ѧ����ʽΪ3S+6KOH 2K2S+K2SO3+3H2O���÷�Ӧ���������뻹ԭ������֮��Ϊ___________��
2K2S+K2SO3+3H2O���÷�Ӧ���������뻹ԭ������֮��Ϊ___________��
��3����������Ҫ��������ʣ��Լ�����_________��Ŀ����___________________________��
��4����������ʹ��ҺpH___________�������С������
��5�����������¶�Ϊ70 -80���Ŀ����_________________________________________ ��
��6��˫��ˮ�����Ի��������ᣩ����һ��ǿ�����������Խ������������������������Ƶ�Fe2(OH)n(SO4)3-n/2����Ӧ�Ļ�ѧ����ʽΪ__________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij�о�С�齫һ����������·�徭Ũ�����ϡ���ᴦ����õ�һ�����Һ,���к���Cu2+��Fe2+��Fe3+��Al3+�Ƚ�������,��������������������Էֱ���ȡCuSO4��5H2O�����AlCl3
��Һ: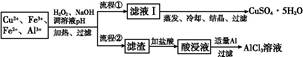
��֪:��ؽ������ӿ�ʼ��������ȫ����ʱ��pH��ΧΪ:
| ���� | Fe3+ | Fe2+ | Al3+ | Cu2+ |
| pH��Χ | 2.2��3.2 | 5.5��9.0 | 4.1��5.0 | 5.3��6.6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�Ի�ͭ��(��Ҫ�ɷ���CuFeS2,����������SiO2)Ϊԭ����ͭ�ķ�����Ϊ������ͭ��ʪ����ͭ���֡�������,ʪ����ͭ�����½�չ,��ѧ�ҷ�����һ��ϸ��������ˮ��Һ������������,���Խ���ͭ��������������:4CuFeS2+2H2SO4+17O2 4CuSO4+2Fe2(SO4)3+2H2O��ij�������ø�ԭ������ͭ���̷��Ĺ�������:
4CuSO4+2Fe2(SO4)3+2H2O��ij�������ø�ԭ������ͭ���̷��Ĺ�������: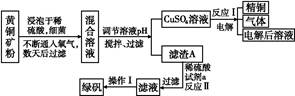
�ش���������:
(1)���������п����������������е�����ҺpH������������(�����)��
| A��Cu�� | B��Cu2(OH)2CO3�� | C��H2SO4�� | D��Fe��E.CuO |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���ҵĻ������÷����ܶ࣬���ú���Al2O3��SiO2������FeO xFe2O3�������Ʊ�Al2(S04)3
xFe2O3�������Ʊ�Al2(S04)3 18H2O�������������£�
18H2O�������������£�
��ش��������⣺
��1���������ϡH2SO4�ܽ�Al2O3�����ӷ���ʽ��______________��
��2�������м��˵�KMnO4Ҳ����H2O2���棬����H2O2������Ӧ�Ļ�ѧ����ʽΪ_______________��
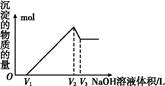
��3����֪��Ũ�Ⱦ�ΪO.1mol/L�Ľ��������ӣ������������������pH���±���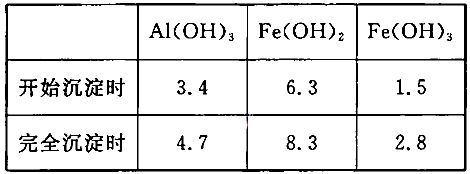
����۵�Ŀ����__________________________________________________________�����ڸ�Ũ���³�ȥ���Ļ��������pH�����Χ��___________��
��4����֪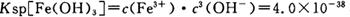 ��pH=2ʱ��Fe3����ʼ������Ũ��Ϊ_______________��
��pH=2ʱ��Fe3����ʼ������Ũ��Ϊ_______________��
��5�������ܷ�����Ӧ�����ӷ���ʽΪ__________________________________________��Ϊ����֤�ò������ù�����ȷʵ����MnO2����ѡ�õ��Լ���_________��_________��
��6�������ݡ�һϵ�в���"�����������в����õ���___________������ţ���
| A�������� | B������ | C�������� | D���ƾ���E��©�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij�������Թ�ҵ��ˮ�к���һ������Na����Al3���� Fe3����Cu2����Cl�����ó�������ͼ��ʾ�Ĺ�������ͼ�����ó���������������ᡢ���ҵ�����еķ���м���ӷ�ˮ�����������Ȼ�������������NaCl����ͽ���ͭ�������˺ܺõ���ᾭ��Ч�档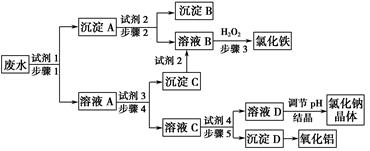
����д���пհף�
(1)ͼ���Լ�1��________���Լ�2��________��
(2)����1�Ͳ���2���õ��IJ���������________��
(3)����1��Ӧ�����ӷ���ʽΪ_______________________________________��
(4)����3��Ӧ�����ӷ���ʽΪ_______________________________________��
(5)�ӽ�ԼҩƷ�ͻ������濼�ǣ�����5��������Ӧ�����ӷ���ʽӦΪ__________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������ͭ�Ƚ������仯�������ճ�������Ӧ�ù㷺,���������ʵ��ش�����:
(1)�����к���һ����̼������X(Fe3C)��X�������Ŀ����и�������,�����д��ԵĹ���Y,��Y���ڹ����������Һ�к��еĴ�������������������
(2)ij��Һ����Mg2+��Fe2+��Al3+��Cu2+����������,�����м��������NaOH��Һ��,����,�������������ղ������պ�Ĺ���Ͷ�������ϡ������,������Һ��ԭ��Һ���,��Һ�д������ٵ�����������������
A.Mg2+ B.Fe2+ C.Al3+ D.Cu2+
(3)����������Ҫ��ҵ����,�÷���м�Ʊ�������������:
�ش���������:
�ٲ������������������,���������������������
��д���ڿ���������FeCO3�Ļ�ѧ����ʽ������������������������������
(4)��Щͬѧ��ΪKMnO4��Һ�ζ�Ҳ�ܽ�����Ԫ�غ����IJⶨ��
a.��ȡ2.850 g�̷�(FeSO4��7H2O)��Ʒ,�ܽ�,��250 mL����ƿ�ж���;
b.��ȡ25.00 mL������Һ������ƿ��;
c.�������ữ��0.010 00 mol��L-1 KMnO4��Һ�ζ����յ�,����KMnO4��Һ�����ƽ��ֵΪ20.00 mL��
��ʵ��ǰ,����Ҫ��ȷ����һ�����ʵ���Ũ�ȵ�KMnO4��Һ250 mL,����ʱ��Ҫ����������ƽ�����������ձ�����ͷ�ι���,������������
��ijͬѧ��Ƶ����еζ���ʽ,�����������������(�гֲ�����ȥ)(����ĸ���) 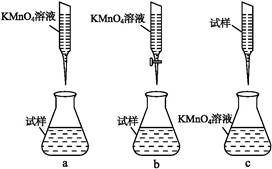
��д���ζ������з�Ӧ�����ӷ���ʽ:��������������������������������������
�ܼ���������Ʒ��FeSO4��7H2O����������Ϊ����������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij�����ĵ�������к���ͭ�����Ƚ��������Ϊʵ����Դ�Ļ������ò���Ч��ֹ������Ⱦ��������¹������̣�
��1����������H2O2��Ŀ���� ����pH�����м�����Լ������ ���ѧʽ����ʵ���ҽ��й��˲������õ��IJ��������� ��
��2�����CuSO4��Һ��ԭ���� ����CuSO4��Һ�м���һ������NaCl��Na2SO3���������ɰ�ɫ��CuCl������д���÷�Ӧ�Ļ�ѧ����ʽ ��
��3����ȡ���Ʊ���CuCl��Ʒ0.2500g����һ������0.5mol��L-1FeCl3��Һ�У�����Ʒ��ȫ�ܽ��ˮ20mL����0.1000mol��L-1��Ce��SO4��2��Һ�ζ��������յ�ʱ����Ce��SO4��2��Һ25.00mL���йصĻ�ѧ��ӦΪ��Fe3����CuCl��Fe2����Cu2����Cl����Ce4����Fe2����Fe3����Ce3���������CuCl��Ʒ���������� ��
��4��Fe3+����ˮ�ⷴӦFe3����3H2O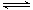 Fe(OH)3��3H�����÷�Ӧ��ƽ�ⳣ������ʽΪ ��
Fe(OH)3��3H�����÷�Ӧ��ƽ�ⳣ������ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ú���Al2O3������Fe2O3��SiO2���������Ʊ���ˮ������Һ��ۺ����������������������£����ֲ����������ԣ���
I�����������м������H2SO4���ȡ����衢���ˡ�
II������Һ�м���һ������FeSO4��7H2O��˫��ˮ��
III������Һ�м���Ca(OH)2���壬������Һ��pHԼΪ1�����ˡ�
IV�������ȶ��������ȣ��õ���Ʒ��
��1��Fe2O3��H2SO4��Ӧ�����ӷ���ʽ��___________��
��2������I�й��˵õ��������ɷ���________���ѧʽ����
��3������I ��H2SO4��Ũ���뷴Ӧ�¶Ȼ�Ӱ���������Ľ����ʡ�������ͼ����������I ��H2SO4Ũ�ȵ����˷�Χ��__________����Ӧ�������¶���_________��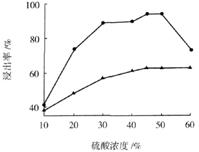

��4������II������n(Fe3+)�����ӷ���ʽ��_________��
��5������III�õ���ʽ��������[AlFe(OH)n(SO4)m]����Һ������II��Ӧ����n(Fe3+)��
n(Al3+)�sn(Fe3+)= ��
��6���о�������Һ��ۺ����������Ĵ���Խ�ߣ���ˮЧ��Խ�á���֪��
һЩ������20��ʱ���ܽ��
| ���� | Ca(OH)2 | CaSO4 | Na2SO4 |
| �ܽ��/g | 0.153 | 0.258 | 19.5 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com