
��8�֣���������ЧӦ����Դ��ȱ�����⣬��ν��ʹ����е�CO2���������Կ������ã������˸������ձ����ӡ�Ŀǰ��ҵ����һ�ַ�������CO2����ȼ�ϼ״���һ�������·�����Ӧ��CO2(g)+3H2(g) CH3OH(g)+H2O(g)����ͼ��ʾ�÷�Ӧ���й���������(��λΪkJ��mol��1)�ı仯��
CH3OH(g)+H2O(g)����ͼ��ʾ�÷�Ӧ���й���������(��λΪkJ��mol��1)�ı仯��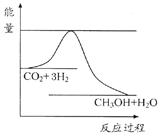
��1�����ڸ÷�Ӧ������˵���У���ȷ���� ��(����ĸ)
A����H>0����S>0 B����H>0����S<0
C����H<0����S<0 D����H<0����S>0
��2���÷�Ӧƽ�ⳣ��K�ı���ʽΪ ��
��3���¶Ƚ��ͣ�ƽ�ⳣ��K (����������䡱��С��)��
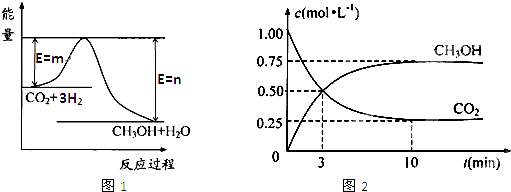
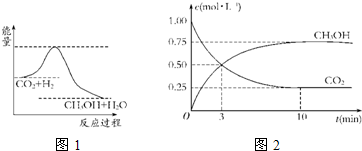
��4��Ϊ̽����Ӧԭ�����ֽ�������ʵ�飺�����Ϊ1L�ĺ����ܱ������У�����1molCO2��3molH2�����CO2��CH3OH(g)��Ũ����ʱ�ʱ仯����ͼ��ʾ���ӷ�Ӧ��ʼ��ƽ�⣬������Ũ�ȱ仯��ʾ��ƽ����Ӧ����v(H2)Ϊ mol��L��1��min��1��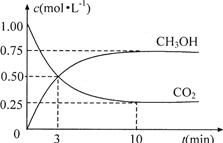
��5�����д�ʩ����ʹ��4����n(CH3OH)/n(CO2)������� ��(����ĸ)
A�������¶�
B���������
C����H2O(g)����ϵ�з���
D���ٳ���1molCO2��3molH2
E������He(g)��ʹ��ϵ��ѹǿ����
��8�֣���1��C ��2�֣���2��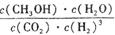 ��1�֣���3������1�֣���4�� 0.225 ��2�֣���5��CD��2�֣�
��1�֣���3������1�֣���4�� 0.225 ��2�֣���5��CD��2�֣�
���������������1�����ݷ�Ӧ�ķ���ʽ��֪������Ӧ�������С�ģ�����S��0������ͼ���֪����Ӧ���������������������������������Ƿ��ȷ�Ӧ����H��0����ѡC��
��2����ѧƽ�ⳣ������һ�������£������淴Ӧ�ﵽƽ��״̬ʱ��������Ũ�ȵ���֮���ͷ�Ӧ��Ũ�ȵ���֮���ı�ֵ�����Ը÷�Ӧ��ƽ�ⳣ������ʽK��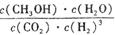 ��
��
��3������Ӧ�Ƿ��ȷ�Ӧ�������¶ȣ�ƽ��������Ӧ�����ƶ���Kֵ����
��4������ͼ���֪��ƽ��ʱ����0.75mol�״��������������������ʵ�����2.25mol����������ƽ����Ӧ������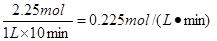 ��
��
��5�������¶ȣ�ƽ�����淴Ӧ�����ƶ���n(CH3OH)/n(CO2)��С�����������ƽ�ⲻ�ƶ���n(CH3OH)/n(CO2)���䣻��H2O(g)����ϵ�з��룬ƽ��������Ӧ�����ƶ���n(CH3OH)/n(CO2)�����ٳ���1molCO2��3molH2���൱���Ǽ�ѹ��ƽ��������Ӧ�����ƶ���n(CH3OH)/n(CO2)����
����He(g)��ʹ��ϵ��ѹǿ����ƽ�ⲻ�ƶ���n(CH3OH)/n(CO2)���䣬��ѡCD��
���㣺���鷴Ӧ�ȡ���ֵ��ƽ�ⳣ������Ӧ�����Լ����������ƽ��״̬��Ӱ��
�����������Ǹ߿��еij������ͣ��ѶȲ������Ի���������Ŀ��飬�����������У������е��Ѷȵ����⡣����ԭ������ѧ���淶���⡢�����������Լ��Ͻ�����˼ά������Ҳ���������ѧ����ѧϰЧ�ʡ�


 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ������CO2����ȼ�ϼ״���һ�������·�����Ӧ��CO2��g��+3H2��g��
������CO2����ȼ�ϼ״���һ�������·�����Ӧ��CO2��g��+3H2��g�� CH3OH��g��+H2O��g����������ͼ��ʾ�÷�Ӧ���й�������������λΪkJ?mol-1���ı仯��
CH3OH��g��+H2O��g����������ͼ��ʾ�÷�Ӧ���й�������������λΪkJ?mol-1���ı仯��
| ��� | NaOH��Һ�����/mL | ��������/mL | ��Һ��pH |
| �� | 20.00 | 0.00 | 8 |
| �� | 20.00 | 20.00 | 6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| c(CH3OH)?c(H2O) |
| c(CO2)?c3(H2) |
| c(CH3OH)?c(H2O) |
| c(CO2)?c3(H2) |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ��� |
| ���� |
| ���� |
| �� |
| ���� |
| �� |
| ���� |
| �� |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com