�⣺��1�������µ���A��ij�ֳ���һԪǿ����Һ��Ӧ���ɵõ�B��C����A��B��C�ж�����X��˵��A�м��з������绯��Ӧ���������������Һ�����绯��Ӧ������ѧ��ѧ����Ҫ��
Cl
2��Br
2�ȣ�������D���ȴ��ֽ⣬���Ƶ�Ԫ��Y�ĵ��ʣ���֪ӦΪKClO
3���ɴ˿�֪XΪO��YΪCl��ZΪK��AΪCl
2��DΪKClO
3���ʴ�Ϊ��Cl�����ȣ���K����أ���
��2��Cl
2�ڼ�����Һ�з�������������ԭ��Ӧ������KCl��KClO���ʴ�Ϊ��Cl
2+2KOH�TKCl+KClO+H
2O��
��3��KClO
3�ڴ����������ȷֽ�����O
2�����������غ㶨�ɿ�д����ѧ����ʽ���ʴ�Ϊ��2KClO
3
2KCl+3O
2����
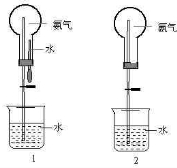
��1��ʵ�����ư������Ȼ�狀���ʯ���ڼ��������·�Ӧ���ɰ�����ˮ�����������غ㶨�ɿ�д����ѧ����ʽ���ʴ�Ϊ��2NH
4Cl+Ca��OH��
2�T2NH
3��+CaCl
2+2H
2O��
��2�������ܶȱȿ���С����������ˮ��ֻ���������ſշ��ռ�������Ϊ�������壬Ӧ�ü��Ը��������ʴ�Ϊ�������ſ����� ��ʯ�ң�
��3�����ݰ�����������ˮ����ֹˮ�У�������ͷ�ι��е�ˮʱ����ƿ��ѹǿѸ�ټ�С��������ѹ�ɽ��ձ��е�ˮѹ����ƿ���γ���Ȫ���ʴ�Ϊ����ֹˮ�У�������ͷ�ι��е�ˮ�� ���������ܽ���ˮ����ʹ��ƿ������ѹǿѸ�ټ�С��
��4��������֣�����ë���ȣ�����ƿ���ȣ������������ͣ��ϳ����������ڵĿ�����������ˮ�Ӵ�����ƿ��ѹǿѸ�ټ�С��������ѹ�ɽ��ձ��е�ˮѹ����ƿ���γ���Ȫ���ʴ�Ϊ�����ӣ����֣�����ë���ȣ�����ƿ���ȣ������������ͣ��ϳ����������ڵĿ�����������ˮ�Ӵ�����������Ȫ��
�����������ͻ�ƿ��ǣ������µ���A��ij�ֳ���һԪǿ����Һ��Ӧ���ɵõ�B��C����A��B��C�ж�����X��˵��A�м��з������绯��Ӧ���������������Һ�����绯��Ӧ������ѧ��ѧ����Ҫ��Cl
2��Br
2�ȣ������С��˼���ķ�Χ���ٽ�����������Ӷ��ҳ��𰸣�
��ʵ�����ư������Ȼ�狀���ʯ���ڼ��������·�Ӧ�����ݰ��������ʣ��ܶȱȿ���С����������ˮ��Ϊ��������ȿ��ҳ��𰸣�����ʽ��Ȫʵ���ԭ�����ǣ�Ҫ�γ���Ȫ������ʹԲ����ƿ�ڵ�ѹǿ��������ѹС�ſ��ԣ��ݴ˻ش�
����������ѧ����Ԫ�ػ�����֪ʶ����Ϥ�̶ȣ�����Ŀ1��ѧ���ۺ�������Ϣ����϶��������ʵ��˽⣬�²��һЩ����C1
2��KC1O
3�ȣ���˳�ơ����ƺ������֤�����ȷ����ȫ���𰸣����ѧ�������е��������ʲ���Ϥ��һ���ӱ�A��B��C��D��X��Y��Z������ţ�����Ҳ�һͨ��

 2KCl+3O2����
2KCl+3O2����



 ��֪��A��B��C��D��E��F��XΪ���ڱ���ǰ�����ڵ�����Ԫ�أ����ǵ�ԭ��������������A�����ڱ���ԭ�Ӱ뾶��С��Ԫ�أ�B�Ļ�̬ԭ����3����ͬ���ܼ������ܼ��е�������ȣ�D�Ļ�̬ԭ��2p�ܼ��ϵ�δ�ɶԵ�������Bԭ�ӵ���ͬ��D2-������E2+���Ӿ�����ͬ���ȶ����Ӳ�ṹ��F�С����������֮�ƣ�F4+���Ӻ��ԭ�ӵĺ�������Ų���ͬ��X�Ļ�̬ԭ�ӵļ۵����Ų�ʽΪ3d84s2��
��֪��A��B��C��D��E��F��XΪ���ڱ���ǰ�����ڵ�����Ԫ�أ����ǵ�ԭ��������������A�����ڱ���ԭ�Ӱ뾶��С��Ԫ�أ�B�Ļ�̬ԭ����3����ͬ���ܼ������ܼ��е�������ȣ�D�Ļ�̬ԭ��2p�ܼ��ϵ�δ�ɶԵ�������Bԭ�ӵ���ͬ��D2-������E2+���Ӿ�����ͬ���ȶ����Ӳ�ṹ��F�С����������֮�ƣ�F4+���Ӻ��ԭ�ӵĺ�������Ų���ͬ��X�Ļ�̬ԭ�ӵļ۵����Ų�ʽΪ3d84s2��

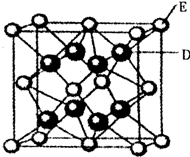 ��֪��A��B��C��D��E��F�����ڱ���ǰ36��Ԫ�أ�A��ԭ�Ӱ뾶��С��Ԫ�أ�BԪ�ػ�̬ԭ�ӵ�2P�����ֻ���������ӣ�CԪ�صĻ�̬ԭ��L��ֻ��2�ԳɶԵ��ӣ�D��Ԫ�����ڱ��е縺������Ԫ�أ�E2+�ĺ�������Ų���Arԭ����ͬ��F�ĺ˵������D��E�ĺ˵����֮�ͣ�
��֪��A��B��C��D��E��F�����ڱ���ǰ36��Ԫ�أ�A��ԭ�Ӱ뾶��С��Ԫ�أ�BԪ�ػ�̬ԭ�ӵ�2P�����ֻ���������ӣ�CԪ�صĻ�̬ԭ��L��ֻ��2�ԳɶԵ��ӣ�D��Ԫ�����ڱ��е縺������Ԫ�أ�E2+�ĺ�������Ų���Arԭ����ͬ��F�ĺ˵������D��E�ĺ˵����֮�ͣ�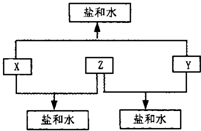
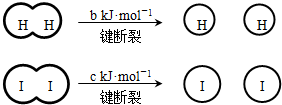 ��a��b��c�������㣩
��a��b��c�������㣩