
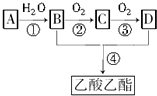
 ��֪����A��ʯ���ѽ�������Ҫ�ɷ֣�A�IJ���ͨ����������һ�����ҵ�ʯ�ͻ���ˮƽ��
��֪����A��ʯ���ѽ�������Ҫ�ɷ֣�A�IJ���ͨ����������һ�����ҵ�ʯ�ͻ���ˮƽ������ A��ʯ���ѽ�������Ҫ�ɷ֣������ͨ����������һ�����ҵ�ʯ�ͻ���ˮƽ����AΪCH2=CH2����ˮ�����ӳɷ�Ӧ�õ�BΪCH3CH2OH��B�������õ�CΪCH3CHO��C�����õ�DΪCH3COOH���Ҵ������ᷢ��������Ӧ�õ�EΪCH3COOCH2CH3���ݴ˽��
��� �⣺A��ʯ���ѽ�������Ҫ�ɷ֣������ͨ����������һ�����ҵ�ʯ�ͻ���ˮƽ����AΪCH2=CH2����ˮ�����ӳɷ�Ӧ�õ�BΪCH3CH2OH��B�������õ�CΪCH3CHO��C�����õ�DΪCH3COOH���Ҵ������ᷢ��������Ӧ�õ�EΪCH3COOCH2CH3��
��1��AΪCH2=CH2���ռ乹�ͣ�ƽ���Σ�
�ʴ�Ϊ��ƽ���Σ�
��2��AΪCH2=CH2�������еĹ�������̼̼˫����CΪCH3CHO�������еĹ�������ȩ����
�ʴ�Ϊ��̼̼˫����ȩ����
��3����Ӧ������ϩ��ˮ�����ӳɷ�Ӧ�����Ҵ�����Ӧ�����Ҵ�����������Ӧ������ȩ��
�ʴ�Ϊ���ӳɷ�Ӧ��������Ӧ��
��4����Ӧ�ٵĻ�ѧ����ʽΪ��CH2=CH2+H2O$\stackrel{����}{��}$CH3CH2OH��
��Ӧ�ڵĻ�ѧ����ʽ��2CH3CH2OH+O2 $��_{��}^{Cu}$2CH3CHO+2H2O��
��Ӧ�����������Ҵ�����������Ӧ����������������Ӧ����ʽΪ��CH3COOH+HOC2H5$��_{��}^{Ũ����}$CH3COOC2H5+H2O��
�ʴ�Ϊ��CH2=CH2+H2O$\stackrel{����}{��}$CH3CH2OH��2CH3CH2OH+O2 $��_{��}^{Cu}$2CH3CHO+2H2O��CH3COOH+HOC2H5$��_{��}^{Ũ����}$CH3COOC2H5+H2O��
���� ���⿼���л�����ƶϣ��漰ϩ��������ȩ�������������ת������Ŀ�ѶȲ����ضԻ���֪ʶ�Ĺ��̣�


 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
 ��
��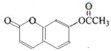 ��
���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��Ӧ����Һ�в������κγ��������Է�Ӧǰ��Cu2+��Ũ�Ȳ��� | |
| B�� | �����ܽ����������ɫ���������[Cu��NH3��4]2+ | |
| C�� | [Cu��NH3��4]2+�Ŀռ乹��Ϊ���������� | |
| D�� | ��[Cu��NH3��4]2+�����У�Cu2+�����¶Ե��ӣ�NH3�ṩ�չ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | CH3CHO��C2H5OH | B�� | C2H5OH��CH2=CH2 | ||
| C�� | 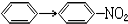 | D�� | CH3COOH��CH3COOC2H5 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
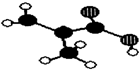 �㽭����������������ij��Ʒֻ��C��H��O����Ԫ�أ������ģ����ͼ��ʾ��ͼ��������֮������ߴ�����ѧ�����絥����˫���ȣ���
�㽭����������������ij��Ʒֻ��C��H��O����Ԫ�أ������ģ����ͼ��ʾ��ͼ��������֮������ߴ�����ѧ�����絥����˫���ȣ���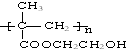 ��
���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��������NaOH����Һ���� | B�� | �ױ���Ũ�����Ũ����ķ�Ӧ | ||
| C�� | ��������NaOHˮ��Һ���� | D�� | �����������ڹ��յ������·�Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | Cl-���ӵĽṹʾ��ͼ�� | B�� | �����ӵı���ģ�ͣ�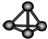 | ||
| C�� | H2O2�Ľṹʽ��H-O-O-H | D�� | CCl4�ĵ���ʽ��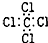 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
 ��ҵ�Ϻ;������ˮú���ڴ��������£���ˮ����������Ӧ��ȡ��������ѧ����ʽΪ��CO��g��+H2O��g��?H2��g��+CO2��g����һ�������£���4molCO��2molH2O��g���������Ϊ2L���ܱ������У���ϵ�и����ʵ������ʱ��仯��ͼ��ʾ��
��ҵ�Ϻ;������ˮú���ڴ��������£���ˮ����������Ӧ��ȡ��������ѧ����ʽΪ��CO��g��+H2O��g��?H2��g��+CO2��g����һ�������£���4molCO��2molH2O��g���������Ϊ2L���ܱ������У���ϵ�и����ʵ������ʱ��仯��ͼ��ʾ���鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com