
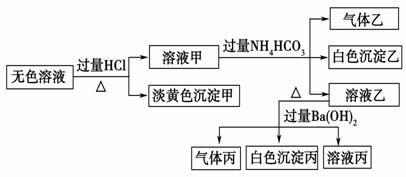
ijͬѧ��̽��ʳƷ���Ӽ������NH4Al��SO4��2��12H2O���·ֽ�������
��1��Ԥ�������й�����������Ԥ�ⲻ�������� ��
A��NH3��N2��SO2��H2O B��NH3��SO3��H2O
C��NH3��SO2��H2O D��NH3��N2��SO3��SO2��H2O
��2�����Լ��飺ȡһ������������������ʵ��̽������ ��
�ٰ�ͼʾ��װ���������ȼ������װ�õ������ԣ�������________��
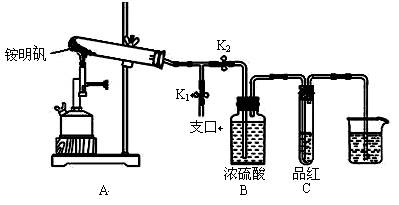
�ڼ�סֹˮ��K1����ֹˮ��K2���þƾ���Ƴ�����ա�ʵ������У�װ��A�͵�����δ������ɫ���壻�Թ�C�е�Ʒ����Һ��ɫ����֧�ڴ��ɼ��鵽NH3�������ǣ���װ��A��B֮���T�͵����г��ְ�ɫ���壬�ð�ɫ���������_________������һ�����ʵĻ�ѧʽ����
�۷����ó�װ��A�Թ��в����İ�ɫ���������������д��������NaOH��Һ�����ӷ���ʽ��
��Ϊ�˷�ֹ������ʵ�����ʱ������____________������ĸ��ţ���Ȼ��Ϩ��ƾ���ơ�
A��ȡ���ձ��еĵ��� B����ֹˮ��K1 C���ر�ֹˮ��K2
��3�������ͽ��ۣ�ʵ��֤����������ǣ�1��D�е�5�����塣��ͬ�����²������N2��SO2��������Ƕ�ֵ��V��N2����V��SO2��=____________��
��1��C��2�֣�
��2���ٹر�֧�ڿ���K1����K2��1�֣��������ĵ���ͨ��ˮ�У��ȴ��Թܣ�1�֣����������ӵ����г������ݣ�1�֣�����ֹͣ���Ⱥ��ڵ���������һ��ˮ������1�֣���֤�������Ժá�����4�֣�
�ڴ�K1����պ��Ũ����IJ���������֧�ڣ������ְ��̣����߲����Լ���������Ҳ���֣���2�֣�������ʪ����ɫʯ����ֽ���鲻�÷֣�����NH4��2SO4���ߣ�NH4��2SO3����SO3����ʽ�μ��������ʵĻ����Ҳ���֣���2�֣�
��Al2O3 +2OH-=2AlO2-+H2O��Al2O3 +3H2O +2OH-=2Al��OH��4-��2�֣���ƽ�����0�֣�
��B��C����BC��2�֣�
��3��1:3��2�֣�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��3Cu��8HNO3(ϡ)��3Cu(NO3)2��2NO����4H2O��Ӧ�У�������_______����������Ԫ���� _______����ԭ������ ���÷�Ӧ��ת�Ƶ��ӵ����ʵ����� mol���õ����ŷ���ʾ�÷�Ӧ _______��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���ӷ���ʽH++OH-=H2O�ɱ�ʾ�Ļ�ѧ����ʽ��
A.������������ͭ�ķ�Ӧ B.���������������ķ�Ӧ
C.�������������Ƶķ�Ӧ D.�����백ˮ�ķ�Ӧ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij��ɫ��Һ�����п��ܴ���Na����Ba2����AlO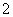 ��S2����SO
��S2����SO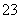 ��SO
��SO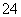 ��ȡ����Һ�����й�ʵ�飬ʵ��������ͼ��ʾ��
��ȡ����Һ�����й�ʵ�飬ʵ��������ͼ��ʾ��
��ش��������⣺
(1)�����Ļ�ѧʽΪ________��
(2)����Һ�����ɳ����ҵ����ӷ���ʽΪ______________��
(3)��������һ������___________(�ѧʽ����ͬ)�����ܺ���__________��
(4)�ۺ�������Ϣ������Һ�п϶����ڵ�������_________��
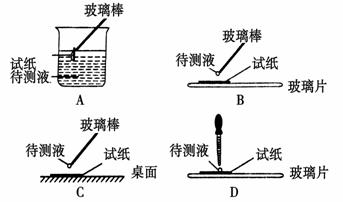 (5)����Һ���� �������ԡ����Ի����ԣ�����Ҫ����������ԣ���ͼ������ȷ���� ��
(5)����Һ���� �������ԡ����Ի����ԣ�����Ҫ����������ԣ���ͼ������ȷ���� ��
�������ӷ���ʽ��ʾ�Լ��Ե�ԭ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���б��ﲻ��ȷ����
A��NaHS��ˮ�еĵ��뷽��ʽΪ��NaHS=Na++HS-��HS-=H++S2-
B��ͬ���ʵ���Ũ�ȵİ�ˮ�����ᷴӦ������ʱ���������V��NH3��H2O����V��HCl��
C��Na2SO3��Һ�У�c��H+��+c��HSO3-��+ 2c��H2SO3��=c��OH-��
D��ͬŨ�ȵ�������Һ�У�c ��CH3COO-���Ĵ�С��CH3COONa��CH3COONH4��CH3COOH
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����˵���в���ȷ���ǣ� ( )
A��ά����C���л�ԭ�ԣ�������������������
B��Ϊ��ֹ�±��ȸ�֬ʳƷ�������ʣ����ڰ�װ���з�����������
C����ˮ���������c(H+) = 1��10��13 mol/L����Һ�У�K+��CO32����Cl����NO3�����ܴ�������
D��ʹʯ�������Һ�У� Na+��I����Cl����NO3�����ܴ�������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��Դ���������Ϊ��Լ������ᾭ�÷�չ��ƿ����Խ��Խ��Ĺ��ҿ�ʼʵ�С�����ƻ���������̫������Դ��Ѱ�÷�չ���¶�����

��1��̫������ˮ���г�ʹ��һ�����������Ͻ������Ϊ���ռ���̫��������Ϳ�㣬д����̬��ԭ�ӵ���Χ�����Ų�ʽ__________����λ�����ڱ�____________����
��2�� ����ϩ���������ھ������õĹ�����ܣ���̫���ܵ�ص�Ӧ���Ͼ��зdz�������ǰ;������ϩ��C60���Ľṹ����ͼ��������̼ԭ�ӹ�����ӻ�����Ϊ________��1 mol C60�����ЦҼ�����ĿΪ____________����
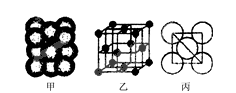
��3��Cu���ʾ�����ԭ�ӵĶѻ���ʽ����ͼ����ʾ���侧����������ͼ����ʾ��ԭ��֮���λ�ù�ϵ��ƽ��ͼ����ͼ����ʾ��������Cuԭ�ӵ���λ��Ϊ____________��һ��������Cuԭ�ӵ���ĿΪ________��
��4��Fe��CO��5�����³�Һ̬���۵�Ϊ��20��5 �棬�е�Ϊ103 �棬�����ڷǼ����ܼ����ݴ˿��ж�Fe��CO��5��������________��������ͣ���Fe��CO��5���������壮��λ���ֱ���________��________��
��5������˵����ȷ����________��
A����һ�����ܴ�С��S��P��Si
B���縺��˳��C��N��O��F
C����Ϊ������CaO��KCl�ߣ�����KCl��CaO�۵��
D��SO2��CO2�Ļ�ѧ�������ƣ����ӽṹҲ����ֱ���ͣ���ͬ������SO2���ܽ�ȸ���
E�����Ӿ����У����ۼ�����Խ�÷��Ӿ�����ۣ��е�Խ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��֪:�� Fe2O3(s)+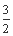 C(s)
C(s)
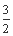 CO2(g)+2Fe(s)����H=+234.1 kJ��mol-1
CO2(g)+2Fe(s)����H=+234.1 kJ��mol-1
��C(s)+O2(g) CO2(g)����H=-393.5 kJ��mol-1
CO2(g)����H=-393.5 kJ��mol-1
��Fe(s)��O2��Ӧ����Fe2O3���Ȼ�ѧ����ʽ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
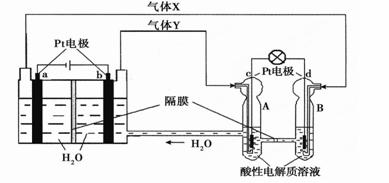
�ռ�ʵ���ҡ��칬һ�š��Ĺ���ϵͳ������������ȼ�ϵ�أ�RFC����RFC��һ�ֽ�ˮ��⼼��������ȼ�ϵ�ؼ������ϵĿɳ�ŵ�ء���ͼΪRFC����ԭ��ʾ��ͼ���й�˵����ȷ����( )
A. ����0.1mol����ת��ʱ��a������1.12L O2����״���£�
B. b���Ϸ����ĵ缫��Ӧ�ǣ�4H2O + 4e- = 2H2��+ 4OH-
C. d���Ϸ����ĵ缫��Ӧ�ǣ�O2 + 4H+ + 4e- = 2H2O
D. c���Ͻ��л�ԭ��Ӧ��B�е�H+����ͨ����Ĥ����A
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com