
ĻĀĮŠĖµ·Ø²»ÕżČ·µÄŹĒ(””””)
¢Ł½«BaSO4·ÅČėĖ®ÖŠ²»Äܵ¼µē£¬ĖłŅŌBaSO4ŹĒ·Ēµē½āÖŹ
¢Ś°±ČÜÓŚĖ®µĆµ½µÄ°±Ė®Äܵ¼µē£¬ĖłŅŌ°±Ė®ŹĒµē½āÖŹ””¢Ū¹ĢĢ¬¹²¼Ū»ÆŗĻĪļ²»µ¼µē£¬ČŪČŚĢ¬µÄ¹²¼Ū»ÆŗĻĪļæÉŅŌµ¼µē””¢Ü¹ĢĢ¬µÄĄė×Ó»ÆŗĻĪļ²»µ¼µē£¬ČŪČŚĢ¬µÄĄė×Ó»ÆŗĻĪļŅ²²»µ¼µē””¢ŻĒæµē½āÖŹČÜŅŗµÄµ¼µēÄÜĮ¦Ņ»¶Ø±ČČõµē½āÖŹČÜŅŗµÄµ¼µēÄÜĮ¦Ēæ
A£®¢Ł¢Ü B£®¢Ł¢Ü¢Ż
C£®¢Ł¢Ś¢Ū¢Ü D£®¢Ł¢Ś¢Ū¢Ü¢Ż
 ŌĶĮæģ³µĻµĮŠ“š°ø
ŌĶĮæģ³µĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ĻÖÓŠ1”«20ŗÅŌŖĖŲA”¢B”¢C”¢DĖł¶ŌÓ¦µÄĪļÖŹµÄŠŌÖŹ»ņĪ¢Į£½į¹¹ČēĻĀ±ķ£ŗ
| ŌŖĖŲ | ĪļÖŹŠŌÖŹ»ņĪ¢Į£½į¹¹ |
| A | M²ćÉĻÓŠ2¶Ō³É¶Ōµē×Ó |
| B | BµÄĄė×ÓÓėDµÄĄė×Ó¾ßÓŠĻąĶ¬µē×Ó²ć½į¹¹£¬ĒŅæÉŅŌĻą»„×éŗĻŠĪ³ÉøÉŌļ¼Į |
| C | ³£ĪĀĻĀµ„ÖŹĪŖĖ«Ō×Ó·Ö×Ó£¬ĘäĒā»ÆĪļĖ®ČÜŅŗ³Ź¼īŠŌ |
| D | ŌŖĖŲ×īøßÕż¼ŪŹĒ£«7¼Ū |
(1)ŌŖĖŲAµÄŌ×Ó×īĶā²ć¹²ÓŠ________ÖÖ²»Ķ¬ŌĖ¶ÆדĢ¬µÄµē×Ó£¬ÓŠ________ÖÖÄÜĮæ²»Ķ¬µÄµē×Ó”£BµÄĄė×ÓÓėDµÄĄė×ÓĻą»„×éŗĻŠĪ³ÉµÄøÉŌļ¼ĮµÄ»ÆѧŹ½ŹĒ________”£
(2)ŌŖĖŲCÓėĒāŌŖĖŲŠĪ³É“ųŅ»øöµ„Ī»Õżµēŗɵĥė×Ó£¬Š“³öøĆĪ¢Į£µÄµē×ÓŹ½________________________________________________________________________(ÓĆŌŖĖŲ·ūŗűķŹ¾)”£
(3)ŌŖĖŲAÓėŌŖĖŲDĻą±Č£¬·Ē½šŹōŠŌ½ĻĒæµÄŹĒ________(ÓĆŌŖĖŲ·ūŗűķŹ¾)£¬ĻĀĮŠ±ķŹöÖŠÄÜÖ¤Ć÷ÕāŅ»ŹĀŹµµÄŹĒ________”£
A£®³£ĪĀĻĀAµÄµ„ÖŹŗĶDµÄµ„֏דĢ¬²»Ķ¬
B£®AµÄĒā»ÆĪļ±ČDµÄĒā»ÆĪļĪȶØ
C£®Ņ»¶ØĢõ¼žĻĀDÄÜ“ÓAµÄĒā»ÆĪļĖ®ČÜŅŗÖŠÖĆ»»³öAµ„ÖŹ
D£®HDµÄĖįŠŌ±ČHAĖįŠŌĒæ
(4)CµÄĒā»ÆĪļ¹ĢĢ¬Ź±ŹōÓŚ________¾§Ģ壬øĆĒā»ÆĪļÓėAµÄ×īøß¼ŪŃõ»ÆĪļ¶ŌÓ¦µÄĖ®»ÆĪļ·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ________________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
25 ”ꏱ£¬H2SO3??HSO £«H£«µÄµēĄė³£ŹżKa£½1”Į10£2mol·L£1£¬ŌņøĆĪĀ¶ČĻĀNaHSO3Ė®½ā·“Ó¦µÄĘ½ŗā³£ŹżKh£½______mol·L£1£¬ČōĻņNaHSO3ČÜŅŗÖŠ¼ÓČėÉŁĮæµÄI2£¬ŌņČÜŅŗÖŠ
£«H£«µÄµēĄė³£ŹżKa£½1”Į10£2mol·L£1£¬ŌņøĆĪĀ¶ČĻĀNaHSO3Ė®½ā·“Ó¦µÄĘ½ŗā³£ŹżKh£½______mol·L£1£¬ČōĻņNaHSO3ČÜŅŗÖŠ¼ÓČėÉŁĮæµÄI2£¬ŌņČÜŅŗÖŠ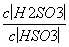 ½«________(Ģī”°Ōö“ó”±”¢”°¼õŠ””±»ņ”°²»±ä”±)”£
½«________(Ģī”°Ōö“ó”±”¢”°¼õŠ””±»ņ”°²»±ä”±)”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
Š“³öĻĀĮŠµäŠĶĪļÖŹµÄµēĄė·½³ĢŹ½
(1)H2SO4_______________________________________________________________________£»
(2)H2CO3_______________________________________________________________________£»
(3)Ca(OH)2____________________________________________________________________£»
(4)Fe(OH)3_____________________________________________________________________£»
(5)NH3·H2O____________________________________________________________________£»
(6)NaCl________________________________________________________________________£»
(7)BaSO4______________________________________________________________________£»
(8)NaHSO4_____________________________________________________________________£»
(9)NaHCO3_____________________________________________________________________£»
(10)NaHSO4(ČŪČŚ)______________________________________________________________£»
(11)Al2O3(ČŪČŚ)_______________________________________________________________£»
(12)CH3COOH_________________________________________________________________ӣ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
Ļņŗ¬µā·ĻŅŗÖŠ¼ÓČėÉŌ¹żĮæµÄNa2SO3ČÜŅŗ£¬½«·ĻŅŗÖŠµÄI2»¹ŌĪŖI££¬ĘäĄė×Ó·½³ĢŹ½ĪŖ
________________________________________________________________________ӣ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ĻĀĮŠ·“Ó¦µÄĄė×Ó·½³ĢŹ½ÕżČ·µÄŹĒ(””””)
A£®NaAlO2ČÜŅŗÖŠµĪ¼ÓNaHCO3ČÜŅŗ£ŗAlO £«HCO
£«HCO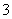 £«H2O===Al(OH)3”ż£«CO
£«H2O===Al(OH)3”ż£«CO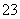
B£®µāĖ®ÖŠĶØČėŹŹĮæµÄSO2£ŗI2£«SO2£«2H2O===2HI£«SO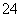 £«2H£«
£«2H£«
C£®FeSO4ČÜŅŗÖŠµĪ¼ÓĖ«ŃõĖ®£ŗ2Fe2£«£«2H2O2===2Fe3£«£«O2”ü£«2H2O
D£®Na2CO3·¢ÉśĖ®½ā·“Ó¦£ŗCO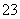 £«2H2O===H2CO3£«2OH£
£«2H2O===H2CO3£«2OH£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ĻĀĮŠŹµŃéĖł¶ŌÓ¦µÄĄė×Ó·½³ĢŹ½ÕżČ·µÄŹĒ(””””)
A£®ĻņNaAlO2ČÜŅŗÖŠĶØČėÉŁĮæCO2£ŗAlO £«CO2£«2H2O===Al(OH)3”ż£«HCO
£«CO2£«2H2O===Al(OH)3”ż£«HCO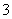
B£®Ļņ·ŠĖ®ÖŠµĪ¼Ó±„ŗĶµÄĀČ»ÆĢśČÜŅŗ£ŗFe3£«£«3H2O===Fe(OH)3”ż£«3H£«
C£®ŌŚĢ¼ĖįĒā±µČÜŅŗÖŠ¼ÓČė¹żĮæµÄĒāŃõ»ÆÄĘČÜŅŗ£ŗBa2£«£«2HCO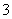 £«2OH£===BaCO3”ż£«CO
£«2OH£===BaCO3”ż£«CO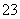 £«2H2O
£«2H2O
D£®ĻņFeCl2ČÜŅŗÖŠ¼ÓČėÉŁĮæK3[Fe(CN)6]ČÜŅŗ£ŗ4Fe2£«£«2[Fe(CN)6]4£===Fe4[Fe(CN)6]2”ż
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ÓÉōĒ»ł·Ö±šøśĻĀĮŠ»łĶÅĻą»„½įŗĻĖł¹¹³ÉµÄ»ÆŗĻĪļÖŠ£¬ŹōÓŚ“¼ĄąµÄŹĒ________£¬ŹōÓŚ·ÓĄąµÄŹĒ________(Ģī×ÖÄø)”£
A£®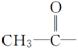 B£®C6H5—
B£®C6H5—
C£®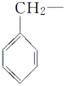 D£®
D£®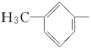
E£®HO—CH2—CH2—
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ĻĀĮŠĖµ·Ø“ķĪóµÄŹĒ(””””)
A£®¹²¼Ū¼ü¾ßÓŠ·½ĻņŠŌ
B£®¹²¼Ū¼ü¾ßÓŠ±„ŗĶŠŌ
C£®¹²¼Ū¼üµÄŹµÖŹŹĒŌ×Ó¹ģµĄµÄĻą»„ÖŲµž
D£®¹²¼Ū¼üŹĒĶعżµē×ӵƏ§¶ųŠĪ³ÉµÄ»Æѧ¼ü
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com