
ijͬѧ�����ͼ��ʾװ�÷ֱ����̽��ʵ�飨�г�װ������ȥ������ش��������⣺
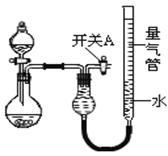 |
| ʵ�� | ҩƷ | ��ȡ���� | �������е�Һ�� |
| �� | Cu��ϡHNO3 | H2O | |
| �� | NaOH���塢Ũ��ˮ | NH3 | |
| �� | þ���Ͻ�NaOH��Һ�������� | H2 | H2O |
��1��ʵ������У�ʹ�÷�Һ©���μ�Һ��IJ�����
��2����ͬѧ��Ϊʵ��I��ͨ���ռ�������NO����������̽��ͭ��Ʒ�Ĵ��ȣ�����Ϊ
�Ƿ���У� ������С������С�����ԭ����
��3��ʵ������������е�Һ�������__ __������ĸ��ţ���ͬ��
A��ŨNaOH��Һ B����ˮ C��ú�� D���Ȼ����Һ
��ʵ��ʣ���NH3�����մ��������¸���β������װ���У�������ֹ�������� ��
��4����ʵ��Ӧ�������ܶ�ζ���������ʱӦע�⣺�ٽ�ʵ��װ�ûָ������£��� ���������밼Һ����ʹ���ƽ��
��5��д��ʵ������漰�����ӷ�Ӧ����ʽ
(6)ʵ������������ݣ���������������ѻ���ɱ�״�������Ե���Һ����������������Ӱ�죩
| ��� | þ���Ͻ����� | �����ܵ�һ�ζ��� | �����ܵڶ��ζ��� |
| �� | 1.0g | 10.0mL | 346.3mL |
| �� | 1.0g | 10.0mL | 335.0mL |
| �� | 1.0g | 10.0mL | 345.7mL |
�����������ݣ��ɼ����þ���Ͻ���������������Ϊ__ __��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����

| ���� |
| ʵ�鲽�� | ���� | ��������� |
| ��һ�� | ȡ����Һ��װ���Թܣ����� ���е��뼸��KSCN��Һ�� |
|
| �ڶ��� | ����Һ��ɫ��ȥ����˵������Fe2+�� �������Ա仯����˵������Fe2+�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��10�֣��������й�SO2��Cl2������ʵ�顣
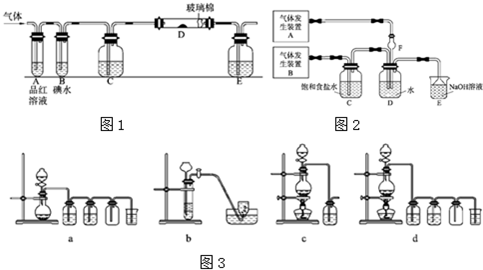
��1��ijС�������ͼ��ʾ��װ��ͼ��ͼ�мгֺͼ���װ����ȥ�����ֱ��о�SO2��Cl2�����ʡ�
��������˷ֱ�ͨ��SO2��Cl2��װ��A�й۲쵽�������Ƿ���ͬ�� �����ͬ������ͬ����������Dװ�������ۣ�ͨ��Cl2�����۳�ַ�Ӧʱ����Ϊ
����װ��D��װ������������������������ͨ��SO2ʱ����Kͨ������O2�Ļ�ѧ��Ӧ����ʽΪ ��
����װ��B��װ��5.0mL 1.0��10-3mol/L�ڵ�ˮ����ͨ������Cl2��ȫ��Ӧ��ת����5.0��10-5mol���ӣ��÷�Ӧ�Ļ�ѧ����ʽΪ ��
��E��ʢ�� ��Һ��
��2��ijͬѧ��������SO2ͨ��һ֧װ����������Һ���Թܣ�δ���������ɣ�������Թ��м������������ĸ�� ������Կ�����ɫ����������
A����ˮ B��ϡ���� C��ϡ���� D���Ȼ���
��3������Ԫ��S��O���-2���������X��X��S��O��������Ϊ4��3����Cl2���뺬X����Һ��ȫ��Ӧ����dz��ɫ����������ȡ�ϲ���Һ�����Ȼ�����Һ���а�ɫ����������д��Cl2�뺬X����Һ��Ӧ�����ӷ���ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�����ʡ����11���¿���ѧ���Ծ� ���ͣ�ʵ����
��10�֣��������й�SO2��Cl2������ʵ�顣
��1��ijС�������ͼ��ʾ��װ��ͼ��ͼ�мгֺͼ���װ����ȥ�����ֱ��о�SO2��Cl2�����ʡ�

��������˷ֱ�ͨ��SO2��Cl2��װ��A�й۲쵽�������Ƿ���ͬ�� �����ͬ������ͬ����������Dװ�������ۣ�ͨ��Cl2�����۳�ַ�Ӧʱ����Ϊ
����װ��D��װ������������������������ͨ��SO2ʱ����Kͨ������O2�Ļ�ѧ��Ӧ����ʽΪ ��
����װ��B��װ��5.0 mL 1.0��10-3mol/L�ڵ�ˮ����ͨ������Cl2��ȫ��Ӧ��ת����5.0��10-5mol���ӣ��÷�Ӧ�Ļ�ѧ����ʽΪ ��
��E��ʢ�� ��Һ��
��2��ijͬѧ��������SO2ͨ��һ֧װ����������Һ���Թܣ�δ���������ɣ�������Թ��м������������ĸ�� ������Կ�����ɫ����������
A����ˮ B��ϡ���� C��ϡ���� D���Ȼ���
��3������Ԫ��S��O���-2���������X��X��S��O��������Ϊ4��3����Cl2���뺬X����Һ��ȫ��Ӧ����dz��ɫ����������ȡ�ϲ���Һ�����Ȼ�����Һ���а�ɫ����������д��Cl2�뺬X����Һ��Ӧ�����ӷ���ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�������й�SO2��Cl2������ʵ�顣
��1��ijС�������ͼ��ʾ��װ��ͼ��ͼ�мгֺͼ���װ����ȥ�����ֱ��о�SO2��Cl2�����ʡ�

��������˷ֱ�ͨ��SO2��Cl2��װ��A�й۲쵽�������Ƿ���ͬ�� �����ͬ������ͬ����������Dװ�������ۣ�ͨ��Cl2�����۳�ַ�Ӧʱ����Ϊ
����װ��D��װ������������������������ͨ��SO2ʱ����Kͨ������O2�Ļ�ѧ��Ӧ����ʽΪ ��
����װ��B��װ��5.0 mL 1.0��10-3mol/L�ڵ�ˮ����ͨ������Cl2��ȫ��Ӧ��ת����5.0��10-5mol���ӣ��÷�Ӧ�Ļ�ѧ����ʽΪ ��
��E��ʢ�� ��Һ��
��2��ijͬѧ��������SO2ͨ��һ֧װ����������Һ���Թܣ�δ���������ɣ�������Թ��м������������ĸ�� ������Կ�����ɫ����������
A����ˮ B��ϡ���� C��ϡ���� D���Ȼ���
��3������Ԫ��S��O���-2���������X��X��S��O��������Ϊ4��3����Cl2���뺬X����Һ��ȫ��Ӧ����dz��ɫ����������ȡ�ϲ���Һ�����Ȼ�����Һ���а�ɫ����������д��Cl2�뺬X����Һ��Ӧ�����ӷ���ʽ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com