
£Ø8·Ö£©°ŃĆŗ×÷ĪŖČ¼ĮĻæÉĶعżĻĀĮŠĮ½ÖÖĶ¾¾¶£ŗ
Ķ¾¾¶I£ŗC(s) +O2 (g)=CO2(g) ”÷H1<0 ¢Ł
Ķ¾¾¶II£ŗĻČÖĘ³ÉĖ®ĆŗĘų£ŗC(s) +H2O(g)=CO(g)+H2(g) ”÷H2>0 ¢Ś
ŌŁČ¼ÉÕĖ®ĆŗĘų£ŗ2CO(g)+O2 (g)=2CO2(g) ”÷H3<0 ¢Ū
2H2(g)+O2 (g)=2H2O(g) ”÷H4<0 ¢Ü
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©Ķ¾¾¶I·Å³öµÄČČĮæ ( Ģī”°“óÓŚ”±”°µČÓŚ”±»ņ”°Š”ÓŚ”±) Ķ¾¾¶II·Å³öµÄČČĮ攣
£Ø2£©”÷H1”¢”÷H2”¢”÷H3”¢”÷H4µÄŹżŃ§¹ŲĻµŹ½ŹĒ ”£
£Ø3£©12gĢæ·ŪŌŚŃõĘųÖŠ²»ĶźČ«Č¼ÉÕÉś³ÉŅ»Ńõ»ÆĢ¼£¬·Å³ö110.35kJČČĮ攣ĘäČČ»Æѧ·½³ĢŹ½ĪŖ ”£
£Ø4£©ĆŗĢæ×÷ĪŖČ¼ĮĻ²ÉÓĆĶ¾¾¶IIµÄÓŵćÓŠ ”£
£Ø1£©µČÓŚ £Ø2£©¦¤H1=¦¤H2+1/2£Ø¦¤H3+¦¤H4£© £Ø3£©C(s) +!/2O2(g)=CO(g) ¦¤H=-110.35KJ/mol £Ø4£©Č¼ĮĻČ¼ÉÕ³ä·Ö£¬ĄūÓĆĀŹøߣ¬·ÅČȶą£¬ĪŪČ¾ÉŁ”£
½āĪöŹŌĢā·ÖĪö£ŗ»Æѧ·“Ó¦²śÉśµÄČČĮæµÄ¶ąÉŁÖ»ÓėĪļÖŹµÄĘšŹ¼×“Ģ¬ŗĶÖÕĮĖדĢ¬ÓŠ¹Ų£¬¶ųÓė·“Ó¦Ķ¾¾¶ĪŽ¹Ų”£Čē¹ūĘšŹ¼×“Ģ¬ŗĶÖÕĮĖדĢ¬ĻąĶ¬£¬Ōņ·Å³öµÄČČĮæŅ²¾ĶĻąĶ¬”£¦¤H1=¦¤H2+1/2£Ø¦¤H3+¦¤H4£©.12g Ģæ·ŪŌŚŃõĘųÖŠ²»ĶźČ«Č¼ÉÕÉś³ÉŅ»Ńõ»ÆĢ¼£¬·Å³ö110.35kJČČĮ攣ĘäČČ»Æѧ·½³ĢŹ½ĪŖ£ŗC(s) +!/2O2(g)=CO(g) ¦¤H=-110.35KJ/mol”£ĆŗĢæ×÷ĪŖČ¼ĮĻ²ÉÓĆĶ¾¾¶IIæÉŅŌŹ¹Č¼ĮĻ³ä·ÖČ¼ÉÕ£¬Č¼ĮĻµÄĄūÓĆĀŹøߣ¬·Å³öČČĮæ¶ą£¬ĪŪČ¾Ņ²ÉŁ”£øüĒå½ą»·±£”£
æ¼µć£ŗæ¼²éøĒĖ¹¶ØĀɵÄÓ¦ÓĆ”£


 Š”ѧÄÜĮ¦²āŹŌ¾ķĻµĮŠ“š°ø
Š”ѧÄÜĮ¦²āŹŌ¾ķĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
¶ž¼×ĆŃ£ØCH3OCH3£©ŹĒĪŽÉ«ĘųĢ壬æÉ×÷ĪŖŅ»ÖÖŠĀŠĶÄÜŌ“£¬ÓÉŗĻ³ÉĘų£Ø×é³ÉĪŖH2”¢CO”¢ŗĶÉŁĮæCO2£©Ö±½ÓÖʱø¶ž¼×ĆŃ£¬ĘäÖŠÖ÷ŅŖ¹ż³Ģ°üĄØŅŌĻĀĖÄøö·“Ó¦£Ø¾łĪŖæÉÄę·“Ó¦£©£ŗ
¢ŁCO(g)+ 2H2(g) = CH3OH(g) ”÷H1=”Ŗ90£®1 kJ”¤mol-1
¢ŚCO2(g)+ 3H2(g) = CH3OH(g)+H2O(g) ”÷H2=”Ŗ49£®0 kJ”¤mol-1
Ė®ĆŗĘų±ä»»·“Ó¦¢ŪCO(g) + H2O (g)=CO2(g)+H2(g) ”÷H3=”Ŗ41£®1 kJ”¤mol-1
¶ž¼×ĆŃŗĻ³É·“Ó¦¢Ü2CH3OH(g)=CH3OCH3(g)+H2O(g) ”÷H4=”Ŗ24£®5 kJ”¤mol-1
£Ø1£©ÓÉH2ŗĶCOÖ±½ÓÖʱø¶ž¼×ĆŃ£ØĮķŅ»²śĪļĪŖĖ®ÕōĘų£©µÄČČ»Æѧ·½³ĢŹ½ĪŖ ”£
£Ø2£©Ņ»¶ØĪĀ¶ČĻĀ£¬ŌŚŗćČŻĆܱÕČŻĘ÷ÖŠ½ųŠŠ·“Ó¦¢Ł£¬ĻĀĮŠĆčŹöÄÜĖµĆ÷·“Ó¦µ½“ļĘ½ŗāדĢ¬µÄŹĒ ”£
a.ČŻĘ÷ÖŠĘųĢåĘ½¾łĻą¶Ō·Ö×ÓÖŹĮæ±£³Ö²»±ä
b.ČŻĘ÷ÖŠĘųĢåĆܶȱ£³Ö²»±ä
c.CH3OH(g)ÅØ¶Č±£³Ö²»±ä
d.CH3OH(g)µÄĻūŗÄĖŁĀŹµČÓŚH2 (g)µÄĻūŗÄĖŁĀŹ
£Ø3£©Ņ»¶ØĪĀ¶ČĻĀ£¬½«8mol CH3OH(g)³äČė5LĆܱÕČŻĘ÷ÖŠ½ųŠŠ·“Ó¦¢Ü£¬Ņ»¶ĪŹ±¼äŗ󵽓ļĘ½ŗāדĢ¬£¬·“Ó¦¹ż³ĢÖŠ¹²·Å³ö49kJČČĮ棬ŌņCH3OH(g)µÄĘ½ŗā×Ŗ»ÆĀŹĪŖ £¬øĆĪĀ¶ČĻĀ£¬Ę½ŗā³£ŹżK= £»øĆĪĀ¶ČĻĀ£¬ĻņČŻĘ÷ÖŠŌŁ³äČė2mol CH3OH(g)£¬¶ŌŌŁ“Ī“ļµ½µÄĘ½ŗāדĢ¬µÄÅŠ¶ĻÕżČ·µÄŹĒ ”£
a.CH3OH(g)µÄĘ½ŗā×Ŗ»ÆĀŹ¼õŠ”
b.CH3OCH3 (g)µÄĢå»ż·ÖŹżŌö“ó
c.H2O(g)ÅضČĪŖ0£®5mol”¤L-1
d.ČŻĘ÷ÖŠµÄŃ¹Ēæ±äĪŖŌĄ“µÄ1£®25±¶
£Ø4£©¶ž¼×ĆŃ”ŖŃõĘųČ¼ĮĻµē³Ų¾ßÓŠĘō¶Ææģ£¬Š§ĀŹøßµČÓÅµć£¬ĘäÄÜĮæĆܶČøßÓŚ¼×“¼Č¼ĮĻµē³Ų£¬Čōµē½āÖŹĪŖĖįŠŌ£¬¶ž¼×ĆŃ”ŖŃõĘųČ¼ĮĻµē³ŲµÄøŗ¼«·“Ó¦ĪŖ £»ĻūŗÄ2£®8L(±ź×¼×“æö)ŃõĘųŹ±£¬ĄķĀŪÉĻĮ÷¾ĶāµēĀ·µÄµē×Ó mol
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
¹ż¶ČÅÅ·ÅCO2»įŌģ³É”°ĪĀŹŅŠ§Ó¦”±£¬ĪŖĮĖ¼õÉŁĆŗČ¼ÉÕ¶Ō»·¾³Ōģ³ÉµÄĪŪČ¾£¬ĆŗµÄĘų»ÆŹĒøߊ§”¢Ēå½ąĄūÓĆĆŗĢæµÄÖŲŅŖĶ¾¾¶”£Ćŗ×ŪŗĻĄūÓƵÄŅ»ÖÖĶ¾¾¶ČēĶ¼ĖłŹ¾”£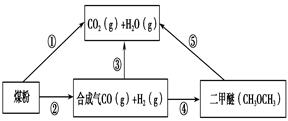
£Ø1£©ŅŃÖŖ¢ŁC(s) £« H2O(g) = CO(g)£«H2(g) ¦¤H1£½£«131.3 kJ”¤mol£1
¢ŚC(s) £« 2H2O(g) = CO2(g) £« 2H2(g) ¦¤H2£½£«90 kJ”¤mol£1
ŌņŅ»Ńõ»ÆĢ¼ÓėĖ®ÕōĘų·“Ӧɜ³É¶žŃõ»ÆĢ¼ŗĶĒāĘųµÄČČ»Æѧ·½³ĢŹ½ŹĒ ________________________£¬
£Ø2£©ÓĆĻĀĶ¼Ōµē³Ų×°ÖĆæÉŅŌĶź³É¹ż³Ģ¢ŻµÄ×Ŗ»Æ£¬øĆ×°ÖĆbµē¼«µÄµē¼«·“Ó¦Ź½ŹĒ_______________________”£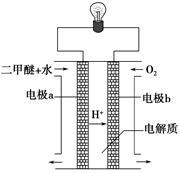
£Ø3£©ŌŚŃ¹ĒæĪŖ0.1 MPaĢõ¼žĻĀ£¬ČŻ»żĪŖV LµÄĆܱÕČŻĘ÷ÖŠa mol COÓė2a mol H2ŌŚ“߻ƼĮ×÷ÓĆĻĀ·“Ӧɜ³É¼×“¼£ŗ
CO(g)£«2H2(g)  CH3OH(g)£¬COµÄĘ½ŗā×Ŗ»ÆĀŹÓėĪĀ¶Č”¢Ń¹ĒæµÄ¹ŲĻµČēĻĀĶ¼ĖłŹ¾£¬Ōņ£ŗ
CH3OH(g)£¬COµÄĘ½ŗā×Ŗ»ÆĀŹÓėĪĀ¶Č”¢Ń¹ĒæµÄ¹ŲĻµČēĻĀĶ¼ĖłŹ¾£¬Ōņ£ŗ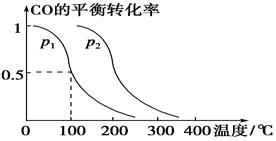
¢Łp1________p2(Ģī”°£¾”±”¢”°£¼”±»ņ”°£½”±)”£
¢ŚŌŚĘäĖūĢõ¼ž²»±äµÄĒéæöĻĀ£¬ĻņČŻĘ÷ÖŠŌŁŌö¼Óa mol COÓė2a mol H2£¬“ļµ½ŠĀĘ½ŗāŹ±£¬COµÄĘ½ŗā×Ŗ»ÆĀŹ________(Ģī”°Ōö“ó”±”¢”°¼õŠ””±»ņ”°²»±ä”±)”£
¢ŪŌŚp1ĻĀ£¬100 ”ꏱ£¬CO(g)£«2H2(g)  CH3OH(g)·“Ó¦µÄĘ½ŗā³£ŹżĪŖ________(ÓĆŗ¬a”¢VµÄ“śŹżŹ½±ķŹ¾)”£
CH3OH(g)·“Ó¦µÄĘ½ŗā³£ŹżĪŖ________(ÓĆŗ¬a”¢VµÄ“śŹżŹ½±ķŹ¾)”£
£Ø4£©ČēĶ¼±ķŹ¾CO2ÓėH2·“Ӧɜ³ÉCH3OHŗĶH2OµÄ¹ż³ĢÖŠÄÜĮæ(µ„Ī»ĪŖkJ”¤mol£1)µÄ±ä»Æ£ŗ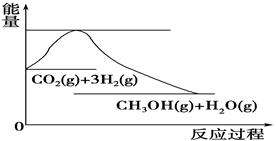
¹ŲÓŚøĆ·“Ó¦µÄĻĀĮŠĖµ·ØÖŠ£¬ÕżČ·µÄŹĒ________(Ģī±ąŗÅ)”£
A£®¦¤H£¾0£¬¦¤S£¾0 B£®¦¤H£¾0£¬¦¤S£¼0
C£®¦¤H£¼0£¬¦¤S£¼0 D£®¦¤H£¼0£¬¦¤S£¾0
£Ø5£©ĪŖĢ½¾æ·“Ó¦ŌĄķ£¬ĻÖ½ųŠŠČēĻĀŹµŃ飬ŌŚĢå»żĪŖ1 LµÄĆܱÕČŻĘ÷ÖŠ£¬³äČė1 mol CO2ŗĶ3 mol H2£¬Ņ»¶ØĢõ¼žĻĀ·¢Éś·“Ó¦£ŗCO2(g)£«3H2(g)  CH3OH(g)£«H2O(g)£¬²āµĆCO2(g)ŗĶCH3OH(g)µÄÅضČĖꏱ¼ä±ä»ÆµÄĒśĻßČēĶ¼ĖłŹ¾£ŗ
CH3OH(g)£«H2O(g)£¬²āµĆCO2(g)ŗĶCH3OH(g)µÄÅضČĖꏱ¼ä±ä»ÆµÄĒśĻßČēĶ¼ĖłŹ¾£ŗ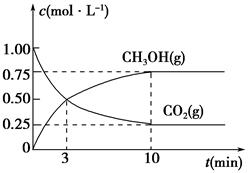
¢Ł“Ó·“Ó¦æŖŹ¼µ½Ę½ŗā£¬CO2µÄĘ½¾ł·“Ó¦ĖŁĀŹv(CO2)£½________”£
¢ŚĻĀĮŠ“ėŹ©ÖŠÄÜŹ¹»ÆŃ§Ę½ŗāĻņÕż·“Ó¦·½ĻņŅĘ¶ÆµÄŹĒ________(Ģī±ąŗÅ)”£
A£®ÉżøßĪĀ¶Č B£®½«CH3OH(g)¼°Ź±Ņŗ»ÆŅĘ³ö
C£®Ń”Ōńøߊ§“߻ƼĮ D£®ŌŁ³äČė1 mol CO2ŗĶ3 mol H2
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
£Ø1£© 8gŅŗĢ¬µÄCH3OHŌŚŃõĘųÖŠĶźČ«Č¼ÉÕ£¬Éś³É¶žŃõ»ÆĢ¼ĘųĢåŗĶŅŗĢ¬Ė®Ź±ŹĶ·Å³öQ kJµÄČČĮ攣ŹŌŠ“³öŅŗĢ¬CH3OHČ¼ÉÕČȵÄČČ»Æѧ·½³ĢŹ½ ”£
£Ø2£©ŌŚ»Æѧ·“Ó¦¹ż³ĢÖŠ£¬ĘĘ»µ¾É»Æѧ¼üŠčŅŖĪüŹÕÄÜĮ棬ŠĪ³ÉŠĀ»Æѧ¼üÓÖ»įŹĶ·ÅÄÜĮ攣
| »Æѧ¼ü | H”ŖH | N”ŖH | N”ŌN |
| ¼üÄÜ/kJ·mol£1 | 436 | 391 | 945 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ŅŃÖŖ£ŗ¢ŁČÜŅŗÖŠCrO42”ŖĻŌ»ĘÉ«£¬Cr2O72-ĻŌ³ČŗģÉ«
¢ŚPbCrO4ÄŃČÜÓŚĖ®£¬Ņ²ÄŃČÜÓŚĒæĖį
¢ŪH+(aq£©+OH-£Øaq£©=H2O(l£©£» ¦¤H=" ”Ŗa" KJ/mol
3Cl2£Øg£©+2Cr3+£Øaq£©+16OH-(aq£©=2CrO42-£Øaq£©+6Cl-£Øaq£©+8H2O(l)£»¦¤H="”Ŗb" KJ/mol
2CrO42-£Øaq£©+2H+(aq£©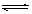 Cr2O72-£Øaq£©+H2O(l£©£»¦¤H="”Ŗc" KJ/mol
Cr2O72-£Øaq£©+H2O(l£©£»¦¤H="”Ŗc" KJ/mol
Ę½ŗā³£ŹżK=9£®5”Į104 £ØÉĻŹöa”¢b”¢c¾ł“óÓŚ0£©
¶ŌÉĻŹö·“Ó¦¢Ż£¬Č”50mLČÜŅŗ½ųŠŠŹŌŃ飬²æ·Ö²ā¶ØŹż¾ŻČēĻĀ£ŗ
| Ź±¼ä£Øs£© | 0 | 0£®01 | 0£®02 | 0£®03 | 0£®04 |
| n £ØCrO42”Ŗ£©£Ømol£© | 0£®01 | 8£®0”Į10-4 | 5£®4”Į10-4 | 5£®0”Į10-4 | |
| n £ØCr2O72”Ŗ£©£Ømol£© | 0 | | 4£®73”Į10-3 | | 4£®75”Į10-3 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
°ŃĆŗ×÷ĪŖČ¼ĮĻæÉĶعżĻĀĮŠĮ½ÖÖĶ¾¾¶£ŗ
Ķ¾¾¶I£ŗC(s) +O2 (g) == CO2(g) ”÷H1<0 ¢Ł
Ķ¾¾¶II£ŗĻČÖĘ³ÉĖ®ĆŗĘų£ŗC(s) +H2O(g) == CO(g)+H2(g) ”÷H2>0 ¢Ś
ŌŁČ¼ÉÕĖ®ĆŗĘų£ŗ2CO(g)+O2 (g) == 2CO2(g) ”÷H3<0 ¢Ū
2H2(g)+O2 (g) == 2H2O(g) ”÷H4<0 ¢Ü
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£© Ķ¾¾¶I·Å³öµÄČČĮæ ( Ģī”°“óÓŚ”±”°µČÓŚ”±»ņ”°Š”ÓŚ”±) Ķ¾¾¶II·Å³öµÄČČĮ攣
£Ø2£© ”÷H1”¢”÷H2”¢”÷H3”¢”÷H4µÄŹżŃ§¹ŲĻµŹ½ŹĒ ”£
£Ø3£©12gĢæ·ŪŌŚŃõĘųÖŠ²»ĶźČ«Č¼ÉÕÉś³ÉŅ»Ńõ»ÆĢ¼£¬·Å³ö110£®35kJČČĮ攣ĘäČČ»Æѧ·½³ĢŹ½ĪŖ ”£
£Ø4£©ĆŗĢæ×÷ĪŖČ¼ĮĻ²ÉÓĆĶ¾¾¶IIµÄÓŵćÓŠ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
£Ø1£©ŌŚ298KŹ±£¬1molC2H6ŌŚŃõĘųÖŠĶźČ«Č¼ÉÕÉś³É¶žŃõ»ÆĢ¼ŗĶŅŗĢ¬Ė®·Å³öČČĮæ1558£®3 kJ”£Š“³öøĆ·“Ó¦µÄČČ»Æѧ·½³ĢŹ½ ”£
£Ø2£©ĄūÓĆøĆ·“Ó¦Éč¼ĘŅ»øöČ¼ĮĻµē³Ų£ŗÓĆĒāŃõ»Æ¼ŲČÜŅŗ×÷µē½āÖŹČÜŅŗ£¬¶ąæ׏ÆÄ«×öµē¼«£¬ŌŚµē¼«ÉĻ·Ö±š
ĶØČėŅŅĶéŗĶŃõĘų”£ĶØČėŅŅĶéĘųĢåµÄµē¼«Ó¦ĪŖ ¼«£ØĢīŠ“”°Õż”±”°øŗ”±£©øƵē¼«ÉĻ·¢ÉśµÄµē¼«·“Ó¦
Ź½ŹĒ
£Ø3£©ČēĶ¼ĖłŹ¾ŹµŃé×°ÖĆÖŠ£¬ŹÆÄ«°ōÉĻµÄµē¼«·“Ó¦Ź½ĪŖ £»Čē¹ūĘšŹ¼Ź±Ź¢ÓŠ1000mLpH=5µÄĮņĖįĶČÜŅŗ£Ø25”ę£¬CuSO4×ćĮ棩£¬Ņ»¶ĪŹ±¼äŗóČÜŅŗµÄpH±äĪŖ1£¬“ĖŹ±ČōŅŖŹ¹ČÜŅŗ»Öø“µ½ĘšŹ¼ÅØ¶Č£ØĪĀ¶Č²»±ä£¬ŗöĀŌČÜŅŗĢå»żµÄ±ä»Æ£©£¬æÉĻņČÜŅŗÖŠ¼ÓČė __________£ØĢīĪļÖŹĆū³Ę£©”£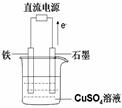
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
(1)CH4(g )+2O2(g )=CO2(g )+2H2O(g ) ¦¤H=-802.3kJ/mol
øĆČČ»Æѧ·“Ó¦·½³ĢŹ½µÄŅāŅåŹĒ_____________________________________”£
(2)ŅŃÖŖ2gŅŅ“¼ĶźČ«Č¼ÉÕÉś³ÉŅŗĢ¬Ė®·Å³öQ kJµÄČČĮ棬Š“³ö±ķŹ¾ŅŅ“¼Č¼ÉÕČȵÄČČ»Æѧ·½
³ĢŹ½£ŗ____________________________________________________________.
(3)ŅŃÖŖ²šæŖ1mol H-H¼ü£¬1mol N-H¼ü£¬1mol 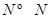 ¼ü·Ö±šŠčŅŖµÄÄÜĮæŹĒ436kJ”¢391KJ”¢946kJ£¬ŌņN2ÓėH2·“Ӧɜ³É1mol NH3(g)µÄČČ»Æѧ·½³ĢŹ½ŹĒ___________________.
¼ü·Ö±šŠčŅŖµÄÄÜĮæŹĒ436kJ”¢391KJ”¢946kJ£¬ŌņN2ÓėH2·“Ӧɜ³É1mol NH3(g)µÄČČ»Æѧ·½³ĢŹ½ŹĒ___________________.
(4)ŅĄ¾ŻøĒĖ¹¶ØĀÉæÉŅŌ¶ŌijŠ©ÄŃŅŌĶعżŹµŃéÖ±½Ó²ā¶ØµÄ»Æѧ·“Ó¦µÄģŹ±ä½ųŠŠĶĘĖć”£
ŅŃÖŖ£ŗC(ŹÆÄ«£¬s)+O2(g)=CO2(g) ¦¤H=-393.5kJ/mol ¢Ł
2H2(g)+O2(g)=2H2O(l) ¦¤H=-571.6kJ/mol ¢Ś
2C2H2(g)+5O2(g)=4CO2(g)+2H2O(l) ¦¤H=-2599kJ/mol ¢Ū
øł¾ŻøĒĖ¹¶ØĀÉ£¬¼ĘĖć298KŹ±ÓÉC£ØŹÆÄ«£¬s£©ŗĶH2(g)Éś³É1mol C2H2(g)·“Ó¦µÄģŹ±ä£ŗ
____________________________.
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
°±ĘųŹĒÉś²ś»Æ·Ź”¢ĻõĖįµČµÄÖŲŅŖŌĮĻ£¬Ī§ČĘŗĻ³É°±ČĖĆĒ½ųŠŠĮĖŅ»ĻµĮŠµÄŃŠ¾æ
£Ø1£©ĒāĘų¼ČÄÜÓėµŖĘųÓÖÄÜÓėŃõĘų·¢Éś·“Ó¦£¬µ«ŹĒ·“Ó¦µÄĢõ¼žČ“²»ĻąĶ¬”£
ŅŃÖŖ£ŗ2H2 (g) + O2 (g) = 2H2O (g) ¦¤H =" -483.6" kJ/mol
3H2 (g) + N2 (g)  2NH3 (g) ¦¤H =" -92.4" kJ/mol
2NH3 (g) ¦¤H =" -92.4" kJ/mol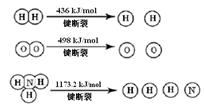
¼ĘĖć¶ĻĮŃ1 mol N”ŌN¼üŠčŅŖÄÜĮæ kJ £¬ µŖĘų·Ö×ÓÖŠ»Æѧ¼ü±ČŃõĘų·Ö×ÓÖŠµÄ»Æѧ¼ü¼ü £ØĢī”°Ēæ”±»ņ”°Čõ”±£©£¬Ņņ“ĖĒāĘųÓė¶žÕß·“Ó¦µÄĢõ¼ž²»Ķ¬”£
£Ø2£©¹ĢµŖŹĒæĘѧ¼ŅÖĀĮ¦ŃŠ¾æµÄÖŲŅŖæĪĢā”£×ŌČ»½ēÖŠ“ęŌŚĢģČ»µÄ“óĘų¹ĢµŖ¹ż³Ģ£ŗN2 (g) + O2 (g) =" 2NO" (g) ¦¤H =" +180.8" kJ/mol £¬¹¤ŅµŗĻ³É°±ŌņŹĒČĖ¹¤¹ĢµŖ”£
·ÖĪöĮ½ÖÖ¹ĢµŖ·“Ó¦µÄĘ½ŗā³£Źż£¬ĻĀĮŠ½įĀŪÕżČ·µÄŹĒ ”£
| ·“Ó¦ | “óĘų¹ĢµŖ | ¹¤Ņµ¹ĢµŖ | ||||
| ĪĀ¶Č/”ę | 27 | 2000 | 25 | 350 | 400 | 450 |
| K | 3.84”Į10-31 | 0.1 | 5”Į108 | 1.847 | 0.507 | 0.152 |
 2NH3 (g)²āµĆ¼×ČŻĘ÷ÖŠH2µÄ×Ŗ»ÆĀŹĪŖ40%”£
2NH3 (g)²āµĆ¼×ČŻĘ÷ÖŠH2µÄ×Ŗ»ÆĀŹĪŖ40%”£| | N2 | H2 | NH3 |
| ¼× | 1 | 3 | 0 |
| ŅŅ | 0.5 | 1.5 | 1 |
| ±ū | 0 | 0 | 4 |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com