
����Ŀ���������ߣ��������Ͽ�֪�����ʾ���ݣ�
���� | �Ҵ� | ���� | �������� | Ũ���� |
�е�/�� | 78.5 | 117.9 | 77.5 | 338.0 |
[ʵ�鲽��]
ijѧ����ʵ������ȡ������������Ҫ�������£�
����30 mL�Ĵ��Թ�A�а������1��4��4����Ũ���ᡢ�Ҵ�������Ļ����Һ��
�ڰ���ͼ��ʾ���Ӻ�װ��(װ������������)����С����ȵؼ���װ�л����Һ�Ĵ��Թ�5��10 min��

�۴��Թ�B�ռ���һ�����IJ����ֹͣ���ȣ���ȥ�Թ�B��������Ȼ���ô��ֲ㣻
�ܷ�������������㡢ϴ�ӡ����
�������ĿҪ��ش��������⣺
(1)д����ȡ���������Ļ�ѧ����ʽ��___________________________��
(2)B�Թ��ñ���̼��������������ռ�����������_____________________��
(3)���������������Ϊ�˸�������������ѡ�õĸ����Ϊ__________(����ĸ)��
A.P2O5 B.��ˮNa2SO4 C.��ʯ�� D.NaOH����

(4)ij��ѧ����С���������ͼ��ʾ����ȡ����������װ��(ͼ�е�����̨�����С�����װ������ȥ)������ͼװ����ȣ���װ�õ���Ҫ�ŵ���__________________��
���𰸡�CH3COOH+C2H5OH![]() CH3COOC2H5+H2O �к����ᣬ�����Ҵ�������Ϊ���������ڱ���̼������Һ�е��ܽ��С�������ڷֲ����� B ���������¶ȼƣ����ڿ��Ʒ���װ���з�ӦҺ���¶ȣ����ٸ�����IJ������������˷�Һ©���������ڼ�ʱ���䷴Ӧ���Һ����������������IJ�����������������װ�ã��������ռ�������������
CH3COOC2H5+H2O �к����ᣬ�����Ҵ�������Ϊ���������ڱ���̼������Һ�е��ܽ��С�������ڷֲ����� B ���������¶ȼƣ����ڿ��Ʒ���װ���з�ӦҺ���¶ȣ����ٸ�����IJ������������˷�Һ©���������ڼ�ʱ���䷴Ӧ���Һ����������������IJ�����������������װ�ã��������ռ�������������
��������
���������������Ʊ�ԭ����������ʵ����ʲ����װ��ͼ�������
��1���������Ҵ���Ӧ��������������ˮ����ѧ��Ӧ����ʽ��CH3COOH+C2H5OH![]() CH3COOC2H5+H2O���ʴ�Ϊ��CH3COOH+C2H5OH
CH3COOC2H5+H2O���ʴ�Ϊ��CH3COOH+C2H5OH![]() CH3COOC2H5+H2O��
CH3COOC2H5+H2O��
��2������̼���ƿ����кͻӷ������ᣬ�����Ҵ�������Ϊ���������ڱ���̼������Һ�е��ܽ��С�������ڷֲ��������ʴ�Ϊ���к����ᣬ�����Ҵ�������Ϊ���������ڱ���̼������Һ�е��ܽ��С�������ڷֲ�������
��3�����������ֲ�Ʒ���ᴿ��������Ϊ������ֲ�Ʒ�м���̼���Ʒ�ĩ(Ŀ���dz�ȥ�ֲ�Ʒ�е�����)���������м��뱥��ʳ��ˮ�뱥���Ȼ�����Һ�������á���Һ(Ŀ���dz�ȥ�ֲ�Ʒ�е��Ҵ�)���������м�����ˮ������(Ŀ���dz�ȥ�ֲ�Ʒ�е�ˮ)��������������������Һ�������һ���������ƿ�ڣ���������ȥ�ͷе���֣��ռ��¶���76��78��֮�����ּ��ô����������������Ի���Ը��������ʹ��������ˮ�⣬���Ը�������������ѡ�õĸ����Ϊ��ˮNa2SO4��ѡB���ʴ�Ϊ��B��
��4���Ա�����ʵ��װ��ͼ��������������Ʊ������еĸ����������ƣ����Կ������ߵ�����ͻ�����ŵ㣺���������¶ȼƣ����ڿ��Ʒ���װ���з�ӦҺ���¶ȣ����ٸ�����IJ������������˷�Һ©���������ڼ�ʱ���䷴Ӧ���Һ����������������IJ�������������ˮ����װ�ã��������ռ����������������ʴ�Ϊ�����������¶ȼƣ����ڿ��Ʒ���װ���з�ӦҺ���¶ȣ����ٸ�����IJ������������˷�Һ©���������ڼ�ʱ���䷴Ӧ���Һ����������������IJ�����������������װ�ã��������ռ���������������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����֪�������衢��������Ͷ�����̼���������������ѧ���ʾ���һ���������ԣ�þ���ƵĻ�ѧ����Ҳ����һ���������ԡ�
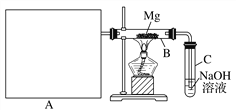
������ͼ��ʾװ�ý���þ�Ͷ��������ʵ�飬����A���Ʊ���������ķ���װ�á�
��1��ѡ����ȡ��������ĺ����Լ�________(�����)��
��10%��������Һ����80%������Һ�����������ƹ��塡��������ƹ���
��2��д��װ��B�з�����Ӧ�Ļ�ѧ����ʽ�� ______________________��
��3������Ϊ��װ�õIJ���֮����_______________________________(��д2��)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������йػ�ѧ��Ӧ��������ȷ���ǣ� ��
A.MnO2��ϡ���ᷴӦ��ȡCl2
B.������ˮ��SO2��Ӧ����(NH4)2SO3
C.����Ũ�����з����ۻ�
D.����������������Ӧ����Na2O2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��X��Y��Z��W��ԭ���������ε����Ķ���������Ԫ�ء�Y��Wͬ���壬��W�ĺ˵������Y��2����X��Y��W����Ԫ��ԭ�ӵ�����������֮��Ϊ17��Y��Z���γ����ӻ�����Z2Y��Z2Y2��
��1��W��Ԫ�����ڱ��е�λ��Ϊ___��
��2��Z������������ˮ����ĵ���ʽ��___�����ۻ�ʱ�˷���������Ϊ___��
��3��Y��Z��W��ԭ�Ӱ뾶�Ӵ�С��˳����___����Ԫ�ط��ű�ʾ����
��4��X�ļ���̬�⻯���W����������ˮ���ﷴӦ�����ӷ���ʽ��___��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����ſ�ѧ�����ķ�չ�������ӵ������IJⶨ�ֶ�Խ��Խ�࣬�ⶨ��ȷ��ҲԽ��Խ�ߣ�����һ�ּ��еIJⶨ���������岽��Ϊ��
(1)������NaCl��ϸ�������ȷ��ȡmgNaCl���岢ת�Ƶ���������A�С�
(2)�õζ�����A�����еμӱ��������������ӱ���A�����Ŀ̶��ߣ������NaCl��������ΪVcm3��
�ٲ���(1)��A���������__________���������ƣ���
�ڲ���(2)������ʽ�ζ��ܺû��Ǽ�ʽ�ζ��ܺã�__________��������______________��
���ܷ��ý�ͷ�ιܴ��沽��(2)�еĵζ���__________��������____________________��
����֪NaCl����Ľṹ����ͼ��ʾ����X���߲��NaCl�����п��������Na+��Cl-���ƽ������Ϊacm�����������ⶨ������ð����ӵ�����NA�ı���ʽΪ��NA=______mol-1��

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������˵�����ʾ������ȷ����
A.1mol��������2mol��������ȫȼ��ʱ��ȼ������ͬ
B.��֪��2H2(g)��O2(g)=2H2O(l) ��H=��571.6kJ��mol��1����H2��ȼ����Ϊ285.8kJ��mol��1
C.��ϡ��Һ�У�H��(aq)��OH��(aq)=H2O(aq)��H=��57.3kJ��mol��1��������1molCH3COOH�Ĵ�����Һ�뺬1molBa(OH)2����Һ��ϣ��ų�������С��57.3kJ
D.��101kPa��25��ʱ��2gH2��ȫȼ������Һ̬ˮ���ų�285.8kJ����������ȼ�յ��Ȼ�ѧ����ʽ��ʾΪ2H2(g)��O2(g)=2H2O(l) ��H=��285.8kJ��mol��1
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������ij�л���![]() ������������ȷ����( )
������������ȷ����( )
A. 1 mol���л��������3 mol Na������Ӧ
B. 1 mol���л��������3 mol NaOH������Ӧ
C. 1 mol���л��������6 mol H2�����ӳɷ�Ӧ
D. 1 mol���л���ֱ�������Na��NaHCO3��Ӧ����������������ͬ������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������ʽΪC12H14O2���л���F�㷺�����Ʊ��㾫�ĵ������Ϊ�˺ϳɸ��л��ijʵ���ҵĿƼ���Ա��������кϳ�·�ߡ�

�Իش��������⣺
(1) C�����к��еĹ�����������__________��________��A���ʵ�����Ϊ_____________��
(2) �ϳ�·���з�Ӧ�۵ķ�Ӧ����Ϊ_________��
(3) д��E�Ľṹ��ʽ_________________����Ӧ�ݵĻ�ѧ����ʽΪ______________________��
(4) ��Ҫ��д��E��һ��ͬ���칹��Ľṹ��ʽ��Ҫ�� ���ڷ����廯����� ��Ũ��ˮ�ܲ�����ɫ������___________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͭ����ѧ����ѧ�ۺ��������õĺϽ𣬹㷺Ӧ�������������Ԫ���������Ǵ�ij�Ͼ���ͭԪ��(��BeO25%��CuS71%������FeS��SiO2)�л������ͭ���ֽ���������

��֪��I.�롢��Ԫ�ش������ڱ��еĶԽ���λ�ã���ѧ��������
��.�����£� Ksp[Cu(OH)2]=2.2��10-20 Ksp[Fe(OH)3]=4.0��10-38 K sp[Mn(OH)2]=2.1��10-13
��1����ҺA����Ҫ�ɷֳ�NaOH�⣬����______________ (�ѧʽ)
д����ӦI�к��뻯������������ᷴӦ�����ӷ���ʽ____________________________
��2������ҺC�к�NaCl��BeCl2������HCl��Ϊ�ᴿBeCl2��ѡ��������貢����______________��
a��������� NaOH b.ͨ�������CO2 c��������İ�ˮ
d.����������HCl e.���� fϴ��
�ڴ�BeCl2��Һ�еõ�BeCl2����IJ�����____________________________��
��3����MnO2�ܽ����������е���Ԫ������Ϊ������д����Ӧ����CuS������Ӧ�Ļ�ѧ����ʽ________________________________________________________��
������ŨHNO3�ܽ�������ȱ����______________ (��дһ��)��
��4����ҺD�к�c(Cu2+)=2.2mol��L-1��c(Fe3+)=0.008mol��L-1��c(Mn2+)=0.01mol��L-1����μ���ϡ��ˮ����pH�����η��룬���ȳ�������______________ (�����ӷ���)��Ϊʹͭ���ӿ�ʼ������������Ӧ������Һ��pHֵ����______________��
��5��ȡ��ͭԪ��1000g�����ջ��Be������Ϊ81g���������______________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com