
�״���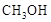 ������Ҫ����Դ���ʣ��о��״�������Ҫ���塣
������Ҫ����Դ���ʣ��о��״�������Ҫ���塣
��1�����ù�ҵ�����е�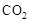 ����ȡ�״����䷴ӦΪ��
����ȡ�״����䷴ӦΪ��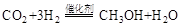
���³�ѹ����֪���з�Ӧ�������仯��ͼ��ʾ��

д���ɶ�����̼�������Ʊ��״����Ȼ�ѧ����ʽ��
��
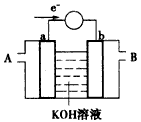
��2��Ϊ��״�ȼ�ϵ������ʣ���ѧ�ҷ�����һ��ȼ�ϵ�أ���ص�һ���缫ͨ���������һ���缫ͨ��״����壬������Dz�����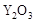 ��
��  ���壬�ڸ��������ܴ���
���壬�ڸ��������ܴ��� ���ӡ���ع���ʱ������ӦΪ ��
���ӡ���ع���ʱ������ӦΪ ��
���Ըõ��Ϊ��Դ����ʯī���缫���100mL�����������ӵ���Һ��
���һ��ʱ��������ռ�����ͬ�������ͬ������������ʱ��������Һ����ı仯���缫������ܴ��ڵ��ܽ������������ռ������������ʵ���Ϊ mol��
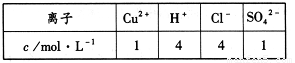
��3���״���ˮ�ʻ����һ������Ⱦ����һ�ֵ绯ѧ��������������Ⱦ����ԭ���ǣ�ͨ��� ������
������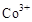 ��Ȼ����
��Ȼ����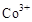 ����������ˮ�еļ״�������
����������ˮ�еļ״�������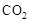 ��������ʵ��������ͼװ��ģ���������̣�
��������ʵ��������ͼװ��ģ���������̣�
��д�������缫��Ӧʽ ��
�ڳ�ȥ�״������ӷ�ӦΪ��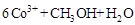
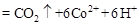 ���ù����б�������Ԫ���� ����������״����2.24L
���ù����б�������Ԫ���� ����������״����2.24L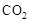 ʱ����ת�Ƶ��� mol��
ʱ����ת�Ƶ��� mol��
��1��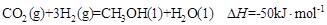 ��2�֣�
��2�֣�
��2��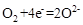 ��2�֣� 0.1��2�֣�
��2�֣� 0.1��2�֣�
��3����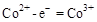 ��2�֣� ��C��̼��2�֣� 0.6��2�֣�
��2�֣� ��C��̼��2�֣� 0.6��2�֣�
��������
�����������1����ͼ��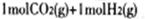 ת��Ϊ
ת��Ϊ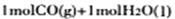 ʱ�����仯Ϊ��41 Kl��mol��1����ͼ��
ʱ�����仯Ϊ��41 Kl��mol��1����ͼ��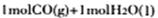 ת��Ϊ
ת��Ϊ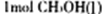 ʱ�����仯Ϊ��419 Kl��mol��1��510
Kl��mol��1= ��9Kl��mol��1�����ɶ�����̼�������Ʊ��״��������仯Ϊ��41 Kl��mol��1+����9Kl��mol��1��=��50 Kl��mol��1
ʱ�����仯Ϊ��419 Kl��mol��1��510
Kl��mol��1= ��9Kl��mol��1�����ɶ�����̼�������Ʊ��״��������仯Ϊ��41 Kl��mol��1+����9Kl��mol��1��=��50 Kl��mol��1
��2��Ϊ��״�ȼ�ϵ������ʣ���ȼ�ϵ�ص�һ���缫ͨ���������ʱ������Ϊ���������״�������Ϊ������������ӦʽΪ��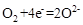 ���������ռ�����ͬ�������ͬ������������ʱ��˵�������̷ֱ������O2��Cl2��H2�����ʱ�������ռ������������ʵ���Ϊ0.1mol��
���������ռ�����ͬ�������ͬ������������ʱ��˵�������̷ֱ������O2��Cl2��H2�����ʱ�������ռ������������ʵ���Ϊ0.1mol��
��3�������⣬ͨ��� ������
������ ˵��
˵��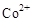 ���ڵĵ缫Ϊ��������������ӦʽΪ��
���ڵĵ缫Ϊ��������������ӦʽΪ��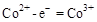 ��CH3OH��Ϊ�����������������б��������Ǽ״��е�CԪ�أ���������״����2.24L
��CH3OH��Ϊ�����������������б��������Ǽ״��е�CԪ�أ���������״����2.24L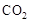 ʱ��˵����������0.1mol
ʱ��˵����������0.1mol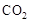 ����ת�Ƶ���0.6 mol��
����ת�Ƶ���0.6 mol��
���㣺��ѧ��Դ��ȼ�ϵ�ء����ӷ�Ӧ
���������л���������������Ӧ��ȼ�ϵ�أ�������ɫ��ѧ��ɳ�����չ����������͵���ĿΪ�߿���Ҫ���㣬�����˻�ѧ������ϢϢ��أ�Ҳ��ǿ��ѧ���Ļ�ѧ�����̽����ʶ��


 Сѧ��ĩ���Ծ�ϵ�д�
Сѧ��ĩ���Ծ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 һ�������£������Ϊ3L���ܱ������У�һ����̼��������Ӧ���ɼ״�������ΪCu2O/ZnO����CO��g��+2H2��g��?CH3OH��g��
һ�������£������Ϊ3L���ܱ������У�һ����̼��������Ӧ���ɼ״�������ΪCu2O/ZnO����CO��g��+2H2��g��?CH3OH��g��| c(CH3OH) |
| c(CO)?C2(H2) |
| c(CH3OH) |
| c(CO)?C2(H2) |
| 2nB |
| 3tB |
| 2nB |
| 3tB |
| 1 |
| 2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013��ɽ��ʡΫ���и���3�µ�һ��ģ�⿼�Ի�ѧ�Ծ����������� ���ͣ������
�״���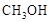 ������Ҫ����Դ���ʣ��о��״�������Ҫ���塣
������Ҫ����Դ���ʣ��о��״�������Ҫ���塣
��1�����ù�ҵ�����е�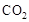 ����ȡ�״����䷴ӦΪ��
����ȡ�״����䷴ӦΪ��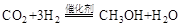
���³�ѹ����֪���з�Ӧ�������仯��ͼ��ʾ��
д���ɶ�����̼�������Ʊ��״����Ȼ�ѧ����ʽ��
��
��2��Ϊ��״�ȼ�ϵ������ʣ���ѧ�ҷ�����һ��ȼ�ϵ�أ���ص�һ���缫ͨ���������һ���缫ͨ��״����壬������Dz�����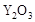 ��
�� 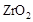 ���壬�ڸ��������ܴ���
���壬�ڸ��������ܴ���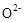 ���ӡ���ع���ʱ������ӦΪ ��
���ӡ���ع���ʱ������ӦΪ ��
���Ըõ��Ϊ��Դ����ʯī���缫���100mL�����������ӵ���Һ��
���һ��ʱ��������ռ�����ͬ�������ͬ������������ʱ��������Һ����ı仯���缫������ܴ��ڵ��ܽ������������ռ������������ʵ���Ϊ mol��
��3���״���ˮ�ʻ����һ������Ⱦ����һ�ֵ绯ѧ��������������Ⱦ����ԭ���ǣ�ͨ���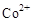 ������
������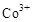 ��Ȼ����
��Ȼ����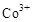 ����������ˮ�еļ״�������
����������ˮ�еļ״�������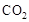 ��������ʵ��������ͼװ��ģ���������̣�
��������ʵ��������ͼװ��ģ���������̣�
��д�������缫��Ӧʽ ��
�ڳ�ȥ�״������ӷ�ӦΪ��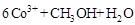
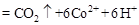 ���ù����б�������Ԫ���� ����������״����2.24L
���ù����б�������Ԫ���� ����������״����2.24L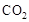 ʱ����ת�Ƶ��� mol��
ʱ����ת�Ƶ��� mol��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��ģ���� ���ͣ������
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com