
 2CH3CHO+2H2O��
2CH3CHO+2H2O�� 2CH3CHO+2H2O��
2CH3CHO+2H2O�� ��
�� ��
��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
| ||
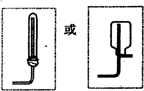
 ��2����ͼ��ʾװ��Ҳ������ȡNH3����Բ����ƿ�еĹ������ѡ��
��2����ͼ��ʾװ��Ҳ������ȡNH3����Բ����ƿ�еĹ������ѡ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
| ||

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�����ʡ����8���¿������Ծ��������棩 ���ͣ�ʵ����
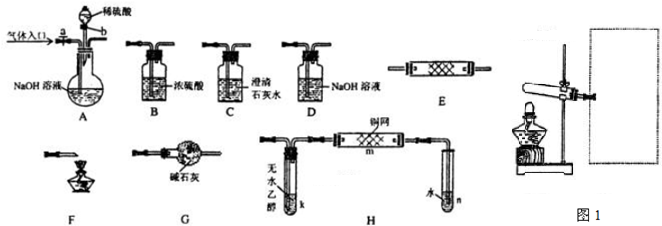
(14�֣���ŨCaCl2��Һ��ͨ��NH3��CO2�������Ƶ�����̼��ƣ�����ֱ����1~100nm֮�䣩����ͼ��ʾA~EΪʵ���ҳ���������װ�ã����̶ֹ��г�װ����ȥ���������Ҫ��ش����⡣

��1��ʵ������ȡ���ռ������NH3����ѡ����������װ�õĽӿ�����˳���ǣ�ѡ ����ĸ����a�� �� �� �� ��h ����Aװ����ȡNH3�Ļ�ѧ��Ӧ����ʽΪ ��
��2����ͼ��ʾװ��Ҳ������ȡNH3����Բ����ƿ�еĹ������ѡ�� ��ѡ����ĸ��ţ���

A����ʯ�� B����ʯ�� C����ˮ�Ȼ��� D����ˮ����ͭ
E���ռ�
��3����ŨCaCl2��Һ��ͨ��NH3��CO2����������̼���ʱ��Ӧ��ͨ��������� ����д��������̼��ƵĻ�ѧ����ʽ ��
��4������Ƽ�ʵ�鷽�����ж�����̼�����Ʒ�����Ƿ�Ϊ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�갲��ʡ�������Ĵ��¿���ѧ�Ծ� ���ͣ�ʵ����
(11�֣���ŨCaCl2��Һ��ͨ��NH3��CO2�������Ƶ�����̼��ƣ�����ֱ����1~100nm֮�䣩����ͼ��ʾA~EΪʵ���ҳ���������װ�ã����̶ֹ��г�װ����ȥ���������Ҫ��ش����⡣

��1��ʵ������ȡ���ռ������NH3����ѡ����������װ�õĽӿ�����˳���ǣ�ѡ ����ĸ����a�� �� �� �� ��h��
��Aװ����ȡNH3�Ļ�ѧ��Ӧ����ʽΪ
��2������ͼ��ʾװ��Ҳ������ȡNH3����Բ����ƿ�еĹ������ѡ�� ��ѡ����ĸ��ţ���

A����ʯ�� B����ʯ�� C����ˮ�Ȼ��� D����ˮ����ͭ E���ռ�
��3����ŨCaCl2��Һ��ͨ��NH3��CO2����������̼��� ʱ��Ӧ��ͨ��������� ����д��������̼��ƵĻ�ѧ����ʽ ��
��4������Ƽ�ʵ�鷽�����ж�����̼�����Ʒ�����Ƿ�Ϊ����
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com