
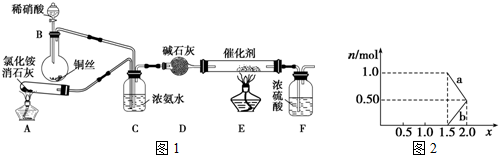
·ÖĪö £Ø1£©AÖŠĻūŹÆ»ŅÓėĀČ»Æļ§ŌŚ¼ÓČČĢõ¼žĻĀ·“Ӧɜ³É°±Ęų£¬BÖŠĻ”ĻõĖįÓėĶ·“Ӧɜ³ÉNO£¬°±ĘųŗĶNO¾C³żŌÓ”¢DøÉŌļŗóŌŚEÖŠ“ß»Æ×÷ÓĆĻĀ·“Ӧɜ³ÉµŖĘų£¬FĪŖøÉŌļ×°ÖĆ£¬
¢ŁŌŚ¼ÓČČĢõ¼žĻĀ£¬ĀČ»Æļ§ŗĶĒāŃõ»ÆøĘ·“Ӧɜ³ÉĀČ»ÆøĘ”¢°±ĘųŗĶĖ®£»
¢Ś°±ĘųŹōÓŚ¼īŠŌĘųĢ壬ÄÜÓĆ¼īŠŌĪļÖŹøÉŌļ£»
£Ø2£©¢Łµ±NOx±»Na2CO3ČÜŅŗĶźČ«ĪüŹÕŹ±£¬Ōņn£ØNO2£©”Żn£ØNO£©£»
¢ŚĄūÓĆ¼«ĻŽ·ØŗĶŹŲŗć·ØĄ“·ÖĪö½ā“š£»
¢ŪĄūÓĆ²īĮæ·Ø¼ĘĖćNO”¢NO2µÄĪļÖŹµÄĮ棬ŌŁøł¾ŻĘ½¾łÄ¦¶ūÖŹĮæ·Ø¼ĘĖćxÖµ£®
½ā“š ½ā£ŗ£Ø1£©AÖŠĻūŹÆ»ŅÓėĀČ»Æļ§ŌŚ¼ÓČČĢõ¼žĻĀ·“Ӧɜ³É°±Ęų£¬BÖŠĻ”ĻõĖįÓėĶ·“Ӧɜ³ÉNO£¬°±ĘųŗĶNO¾C³żŌÓ”¢DøÉŌļŗóŌŚEÖŠ“ß»Æ×÷ÓĆĻĀ·“Ӧɜ³ÉµŖĘų£¬FĪŖøÉŌļ×°ÖĆ£¬
¢ŁŌŚ¼ÓČČĢõ¼žĻĀ£¬ĀČ»Æļ§ŗĶĒāŃõ»ÆøĘ·“Ӧɜ³ÉĀČ»ÆøĘ”¢°±ĘųŗĶĖ®£¬2NH4Cl+Ca£ØOH£©2$\frac{\underline{\;\;”÷\;\;}}{\;}$CaCl2+2NH3”ü+2H2O£¬
¹Ź“š°øĪŖ£ŗ2NH4Cl+Ca£ØOH£©2$\frac{\underline{\;\;”÷\;\;}}{\;}$CaCl2+2NH3”ü+2H2O£»
¢Ś¼īŹÆ»ŅÄÜĪüŹÕĖ®·Ö¶ų×÷øÉŌļ¼Į£¬°±ĘųŹōÓŚ¼īŠŌĘųĢ壬ĖłŅŌÄÜÓĆ¼īŹÆ»ŅøÉŌļ£¬
¹Ź“š°øĪŖ£ŗ³żČ„ĘųĢåÖŠŗ¬ÓŠµÄĖ®ÕōĘų£»
£Ø2£©¢Łµ±NOx±»Na2CO3ČÜŅŗĶźČ«ĪüŹÕŹ±£¬Ōņn£ØNO2£©”Żn£ØNO£©£¬µ±n£ØNO2£©£ŗn£ØNO£©=1Ź±xÖµ×īŠ”£¬x×īŠ”ÖµĪŖ$\frac{2+1}{2}$=1.5£¬ŅņĪŖ»ģÓŠNO£¬ĖłŅŌx×ī“óÖµ£¼2£¬¹ŹxµÄȔֵ·¶Ī§ĪŖ1.5”Üx£¼2£¬ĖłŅŌxµÄÖµ²»æÉÄÜŹĒ1.2£¬
¹Ź“š°øĪŖ£ŗC£»
¢ŚÓĆ¼«ĻŽ·Ø£ŗČōx=1.5ĘųĢåÓ¦ĪŖNOŗĶNO2»ģŗĻĪļ£¬ĪļÖŹµÄĮæ±ČĪŖ1£ŗ1£¬°“¢ńŹ½·“Ó¦£¬Ć»ÓŠNO3-£¬ĻßaÓ¦øƱķŹ¾NO2-£»
ÓĆŹŲŗć·Ø£ŗ·“Ӧɜ³ÉµÄNaNO3ŗĶNaNO2ÖŠµŖŌŖĖŲÓėÄĘŌŖĖŲÖ®±ČĪŖ1£ŗ1£¬ĖłŅŌ1mol NOx±»ĶźČ«ĪüŹÕÖĮÉŁŠčĢ¼ĖįÄĘ0.5mol£¬ÖŹĮæĪŖ53g£¬¼ĘĖćµĆĢ¼ĖįÄĘČÜŅŗµÄÖŹĮæĪŖ$\frac{53g}{42.4%}$=125g£¬
¹Ź“š°øĪŖ£ŗNO2-£»125£»
¢ŪÉčÓÉNO2ŗĶ“æ¼ī·“Ó¦²śÉśCO2ĪŖamol£¬
ÓÉNOŗĶNO2Óė“æ¼ī·“Ó¦²śÉśµÄCO2ĪŖbmol£¬
2NO2+Na2CO3=NaNO2+NaNO3+CO2 ÖŹĮæŌö¼Ó
1mol”÷m=48g
amol 48ag
NO+NO2+Na2CO3=2NaNO2+CO2 ÖŹĮæŌö¼Ó
1mol”÷m=32g
bmol 32bg
$\left\{\begin{array}{l}{a+b=1}\\{48a+32b=44}\end{array}\right.$½āµĆ$\left\{\begin{array}{l}{a=0.75mol}\\{b=0.25mol}\end{array}\right.$
n£ØNO2£©=0.75mol”Į2+0.25mol=1.75mol
n£ØNO£©=0.25mol
x=$\frac{0.25mol”Į1+1.75mol”Į2}{0.25mol+1.75mol}=\frac{15}{8}$=1.875£¬
¹Ź“š°øĪŖ£ŗ1.875£®
µćĘĄ ±¾ĢāŅŌµŖŃõ»ÆĪļĪŖŌŲĢåæ¼²éĮĖĪļÖŹ¼äµÄ·“Ó¦£¬ĪŖøßæ¼³£¼ūĢāŠĶ£¬²ąÖŲӌѧɜµÄ·ÖĪö”¢ŹµŃéŗĶ¼ĘĖćÄÜĮ¦µÄ漲飬Ć÷Č·ĪļÖŹµÄŠŌÖŹŹĒ½ā±¾Ģā¹Ų¼ü£¬ÄѵćŹĒ£Ø2£©ĢāµÄ¼ĘĖć£¬ŅŖ½įŗĻ·½³ĢŹ½ÖŠø÷øöĪļĄķĮæÖ®¼äµÄ¹ŲĻµŹ½½ā“š£¬»įøł¾ŻĢāøųŠÅĻ¢¼ĘĖćxÖµ£¬ÄѶČÖŠµČ£®



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
 ±ķŹ¾µÄ·Ö×ÓŹ½ĪŖC6H14£»Ćū³ĘŹĒ2-¼×»łĪģĶ飮
±ķŹ¾µÄ·Ö×ÓŹ½ĪŖC6H14£»Ćū³ĘŹĒ2-¼×»łĪģĶ飮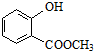 ÖŠŗ¬ÓŠµÄ¹ŁÄÜĶŵÄĆū³ĘĪŖōĒ»ł”¢õ„»ł£®
ÖŠŗ¬ÓŠµÄ¹ŁÄÜĶŵÄĆū³ĘĪŖōĒ»ł”¢õ„»ł£®²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
| Ęų»Æ·“Ó¦Ź½ | 1gK | ||
| 700K | 900K | 1200K | |
| C£Øs£©+H2O£Øg£©=CO£Øg£©+H2£Øg£© | -2.64 | -0.39 | 1.58 |
| C£Øs£©+2H2O£Øg£©=CO2£Øg£©+2H2£Øg£© | -1.67 | -0.03 | 1.44 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | ČŪµć£ŗNa£¾MgO£¾SiO2 | B£® | Ė®ČÜŠŌ£ŗSO2£¾H2S£¾HCl | ||
| C£® | ČČĪČ¶ØŠŌ£ŗHF£¾H2O£¾NH3 | D£® | ·Šµć£ŗHF£¾HCl£¾HBr |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
| ŹµŃé | ·“Ó¦ĪĀ¶Č/”ę | Na2S2O3ČÜŅŗ | Ļ”H2SO4 | H2O | ||
| V/mL | c/£Ømol•L-1£© | V/mL | c/£Ømol•L-1£© | V/mL | ||
| A | 25 | 5 | 0.1 | 10 | 0.1 | 5 |
| B | 25 | 5 | 0.2 | 5 | 0.2 | 10 |
| C | 35 | 5 | 0.1 | 10 | 0.1 | 5 |
| D | 35 | 5 | 0.2 | 5 | 0.2 | 10 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | °“ČŪµćÓɵĶµ½øßÅÅĮŠI2”¢CO2”¢Hg”¢KCl”¢SiO2 | |
| B£® | ŌŚŹÆÓ¢¾§ĢåÖŠ£¬Ćæøö¹čŌ×ÓŗĶĻąĮŚµÄĮ½øöŃõŌ×ÓŅŌ¹²¼Ū¼ü½įŗĻ | |
| C£® | ÓŠ»śĪļ·Ö×ÓÖŠĢ¼Ō×ӳɼü·½Ź½ŗĶÅÅĮŠ·½Ź½ÓŠ¶ąÖÖŅŌ¼°Ķ¬·ÖŅģ¹¹ĻÖĻóµÄ“ęŌŚ¶¼ŹĒÓŠ»śĪļÖÖĄą·±¶ąµÄŌŅņ | |
| D£® | ŅņĪŖŹÆÄ«ŹĒ½šøÕŹÆµÄĶ¬ĖŲŅģŠĪĢ壬ĖłŅŌ¶žÕßæÕ¼ä½į¹¹ĻąĖĘ£¬»ÆѧŠŌÖŹĻąĖĘ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com