
(10·Ö£©ÓŠA”¢B”¢C”¢DĖÄÖÖĄė×Ó»ÆŗĻĪļ£¬×é³ÉĖüĆĒµÄĄė×Ó·Ö±šĪŖ£ŗ
ŃōĄė×Ó£ŗNa£«”¢Al3£«”¢NH4+£» ŅõĄė×Ó£ŗOH£”¢NO3£”¢CO32£”¢HSO4£
ĪŖ¼ų±šĖÄÖÖ»ÆŗĻĪļ£¬Ä³Ń§Éś·Ö±šČ”ÉŁĮæ¹ĢĢåÅä³ÉČÜŅŗ£¬±ąŗÅĪŖA”¢B”¢C”¢D½ųŠŠŹµŃ锣ŹµŃé¹ż³ĢŗĶ¼ĒĀ¼ČēĻĀĶ¼ĖłŹ¾£ØĪŽ¹ŲĪļÖŹŅŃĀŌČ„£©
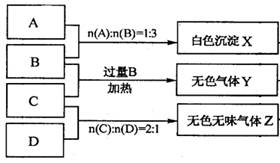
(1) Y”¢ZµÄ»ÆѧŹ½·Ö±šĪŖ£ŗY £»Z
(2)Š“³öÖø¶Ø·“Ó¦µÄĄė×Ó·½³ĢŹ½£ŗ
¢Ł¼ÓČČĢõ¼žĻĀ£¬CÓė¹żĮæB·“Ó¦£ŗ
¢ŚDČÜŅŗĻŌČõ¼īŠŌµÄŌŅņŹĒ(ÓĆĄė×Ó·½³ĢŹ½±ķŹ¾)
(3)µČĪļÖŹµÄĮæÅØ¶ČµÄA”¢B”¢C”¢DČÜŅŗpHÓɓ󵽊”µÄĖ³ŠņŹĒ(ÓĆ»ÆѧŹ½±ķŹ¾)
(4)ČōB”¢CµÄĻ”ČÜŅŗ»ģŗĻŗó(²»¼ÓČČ)ČÜŅŗ³ŹÖŠŠŌ£¬ŌņČÜŅŗÖŠĄė×ÓÅØ¶Č“Ó“óµ½Š”µÄĖ³ŠņŹĒ£ŗ
(1)NH3(1·Ö) CO2(1·Ö)
(2)¢ŁNH4++H++2OH-£½NH3”ü+2H2O(2·Ö)
¢ŚCO32-+H2O HCO3-+OH-
(2·Ö)
HCO3-+OH-
(2·Ö)
(3) NaOH>Na2CO3>Al(NO3)3>NH4HSO4(2·Ö)
(4) c(Na+)>c(SO42-)>c(NH4+)>c(H+)=c(OH-)(2·Ö)
”¾½āĪö”æ£Ø1£©ÄÜŹ¹×ĻÉ«µÄŹÆČļŹŌŅŗĻŌĄ¶É«µÄŹĒ°±Ęų£¬ÄÜŹ¹×ĻÉ«ŹÆČļŹŌŅŗĻŌŗģÉ«µÄŹĒĖįŠŌĘųĢ壬ĖłŅŌøł¾ŻĢāŅāæÉÖŖYŹĒ°±Ęų£¬ZŹĒCO2”£
£Ø2£©°×É«³ĮµķXÄÜČÜÓŚBÖŠ£¬Ņņ“Ė³ĮµķĒāŃõ»ÆĀĮ£¬ĖłŅŌøł¾Ż·“Ó¦µÄĪļÖŹµÄĮæÖ®±ČæÉÖŖ£¬AŹĒĻõĖįĀĮ£¬BŹĒĒāŃõ»ÆÄĘ”£CŹĒĮņĖįĒāļ§£¬DŹĒĢ¼ĖįÄĘ”£
¢Ł¼ÓČČĢõ¼žĻĀ£¬CÓė¹żĮæB·“Ó¦µÄ·½³ĢŹ½ĪŖNH4++H++2OH-£½NH3”ü+2H2O”£
¢ŚĢ¼ĖįÄĘŹĒĒæ¼īČõĖįŃĪ£¬CO32£Ė®½āĻŌ¼īŠŌ£¬·½³ĢŹ½ĪŖCO32-+H2O HCO3-+OH-”£
HCO3-+OH-ӣ
£Ø3£©ĒāŃõ»ÆÄĘŹĒĒæ¼ī£¬¼īŠŌ×īĒ攣Ģ¼ĖįÄĘĖ®½āĻŌ¼īŠŌ£¬ĻõĖįĀĮĖ®½āĻŌĖįŠŌ£¬ĮņĖįĒāļ§µēĄė³öĒāĄė×ÓĻŌĖįŠŌ£¬ĖłŅŌµČĪļÖŹµÄĮæÅØ¶ČµÄA”¢B”¢C”¢DČÜŅŗpHÓɓ󵽊”µÄĖ³ŠņŹĒNaOH>Na2CO3>Al(NO3)3>NH4HSO4”£
£Ø4£©B”¢CµÄĻ”ČÜŅŗ»ģŗĻŗó(²»¼ÓČČ)ČÜŅŗ³ŹÖŠŠŌ£¬ŌņČÜŅŗÖŠŗ¬ÓŠµÄČÜÖŹŹĒĮņĖįÄĘ”¢ĮņĖįļ§ŗĶ°±Ė®£¬ĖłŅŌČÜŅŗÖŠĄė×ÓÅØ¶Č“Ó“óµ½Š”µÄĖ³ŠņŹĒ c(Na+)>c(SO42-)>c(NH4+)>c(H+)=c(OH-)”£



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
£Ø±¾Ģā¹²10·Ö£©ÓŠA”¢B”¢C”¢D”¢E”¢FĮłÖÖŌŖĖŲ”£
¢ŁA”¢B”¢CŹĒ½šŹōŌŖĖŲ£¬Ī»ÓŚĶ¬Ņ»ÖÜĘŚ£¬Ō×ÓŗĖĶā¶¼ÓŠ3øöµē×Ó²ć£¬AµÄŌ×Ó°ė¾¶ŌŚĖłŹōÖÜĘŚÖŠ×ī“ó£¬ĒŅŌ×Ó°ė¾¶A>B>C”£
¢ŚD”¢EŹĒ·Ē½šŹōŌŖĖŲ£¬ĖüĆĒøśĒā»ÆŗĻæÉÉś³ÉĘųĢ¬Ēā»ÆĪļHDŗĶHE£¬ŌŚŹŅĪĀŹ±£¬DµÄµ„ÖŹŹĒŅŗĢ壬EµÄµ„ÖŹŹĒ¹ĢĢ唣
¢ŪFµÄµ„ÖŹŌŚ³£ĪĀĻĀŹĒĘųĢ壬ŠŌÖŹŗÜĪČ¶Ø£¬ŹĒ³żĒāĶā×īĒįµÄĘųĢ唣
Ēė»Ų“š£ŗ
£Ø1£©BĪ»ÓŚÖÜĘŚ±ķÖŠµŚ________ÖÜĘŚ______×壬CµÄŌ×Ó½į¹¹Ź¾ŅāĶ¼ŹĒ________”£
£Ø2£©Fµ„ÖŹµÄ»ÆѧŹ½ŹĒ________”£
£Ø3£©ŌŚÉĻŹöĮłÖÖŌŖĖŲÖŠ£¬×īøß¼ŪŃõ»ÆĪļ¶ŌÓ¦µÄĖ®»ÆĪļ¼īŠŌ×īĒæµÄĪļÖŹŹĒ_____£¬ĖįŠŌ×īĒæµÄĪļÖŹŹĒ________£¬ĘųĢ¬Ēā»ÆĪļ×īĪČ¶ØµÄŹĒ________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2009”Ŗ2010ѧğ¹ćÖŻŹŠĘßĒųĮŖæ¼øßŅ»»ÆѧĻĀѧʌʌĩ¼ą²ā ĢāŠĶ£ŗĢīæÕĢā
£Ø±¾Ģā¹²10·Ö£©ÓŠA”¢B”¢C”¢D”¢E”¢FĮłÖÖŌŖĖŲ”£
¢ŁA”¢B”¢CŹĒ½šŹōŌŖĖŲ£¬Ī»ÓŚĶ¬Ņ»ÖÜĘŚ£¬Ō×ÓŗĖĶā¶¼ÓŠ3øöµē×Ó²ć£¬AµÄŌ×Ó°ė¾¶ŌŚĖłŹōÖÜĘŚÖŠ×ī“ó£¬ĒŅŌ×Ó°ė¾¶A>B>C”£
¢ŚD”¢EŹĒ·Ē½šŹōŌŖĖŲ£¬ĖüĆĒøśĒā»ÆŗĻæÉÉś³ÉĘųĢ¬Ēā»ÆĪļHDŗĶHE£¬ŌŚŹŅĪĀŹ±£¬DµÄµ„ÖŹŹĒŅŗĢ壬EµÄµ„ÖŹŹĒ¹ĢĢ唣
¢ŪFµÄµ„ÖŹŌŚ³£ĪĀĻĀŹĒĘųĢ壬ŠŌÖŹŗÜĪČ¶Ø£¬ŹĒ³żĒāĶā×īĒįµÄĘųĢ唣
Ēė»Ų“š£ŗ
£Ø1£©BĪ»ÓŚÖÜĘŚ±ķÖŠµŚ________ÖÜĘŚ______×壬CµÄŌ×Ó½į¹¹Ź¾ŅāĶ¼ŹĒ________”£
£Ø2£©Fµ„ÖŹµÄ»ÆѧŹ½ŹĒ________”£
£Ø3£©ŌŚÉĻŹöĮłÖÖŌŖĖŲÖŠ£¬×īøß¼ŪŃõ»ÆĪļ¶ŌÓ¦µÄĖ®»ÆĪļ¼īŠŌ×īĒæµÄĪļÖŹŹĒ_____£¬ĖįŠŌ×īĒæµÄĪļÖŹŹĒ________£¬ĘųĢ¬Ēā»ÆĪļ×īĪČ¶ØµÄŹĒ________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2011-2012Äźŗž±±ŅĖ²ż½š¶«·½Ń§Š£ø߶žÉĻŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌ¾ķ£Ø“ų½āĪö£© ĢāŠĶ£ŗĢīæÕĢā
(10·Ö£©ÓŠA”¢B”¢C”¢DĖÄÖÖĄė×Ó»ÆŗĻĪļ£¬×é³ÉĖüĆĒµÄĄė×Ó·Ö±šĪŖ£ŗ
ŃōĄė×Ó£ŗNa£«”¢Al3£«”¢NH4+£» ŅõĄė×Ó£ŗOH£”¢NO3£”¢CO32£”¢HSO4£
ĪŖ¼ų±šĖÄÖÖ»ÆŗĻĪļ£¬Ä³Ń§Éś·Ö±šČ”ÉŁĮæ¹ĢĢåÅä³ÉČÜŅŗ£¬±ąŗÅĪŖA”¢B”¢C”¢D½ųŠŠŹµŃ锣ŹµŃé¹ż³ĢŗĶ¼ĒĀ¼ČēĻĀĶ¼ĖłŹ¾£ØĪŽ¹ŲĪļÖŹŅŃĀŌČ„£©
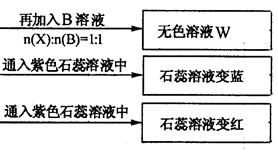
(1) Y”¢ZµÄ»ÆѧŹ½·Ö±šĪŖ£ŗY £»Z
(2)Š“³öÖø¶Ø·“Ó¦µÄĄė×Ó·½³ĢŹ½£ŗ
¢Ł¼ÓČČĢõ¼žĻĀ£¬CÓė¹żĮæB·“Ó¦£ŗ
¢ŚDČÜŅŗĻŌČõ¼īŠŌµÄŌŅņŹĒ(ÓĆĄė×Ó·½³ĢŹ½±ķŹ¾)
(3)µČĪļÖŹµÄĮæÅØ¶ČµÄA”¢B”¢C”¢DČÜŅŗpHÓɓ󵽊”µÄĖ³ŠņŹĒ(ÓĆ»ÆѧŹ½±ķŹ¾)
(4)ČōB”¢CµÄĻ”ČÜŅŗ»ģŗĻŗó(²»¼ÓČČ)ČÜŅŗ³ŹÖŠŠŌ£¬ŌņČÜŅŗÖŠĄė×ÓÅØ¶Č“Ó“óµ½Š”µÄĖ³ŠņŹĒ£ŗ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2009-2010ѧğ¹ćÖŻŹŠĘßĒųĮŖæ¼øßŅ»»ÆѧĻĀѧʌʌĩ¼ą²ā ĢāŠĶ£ŗĢīæÕĢā
£Ø±¾Ģā¹²10·Ö£©ÓŠA”¢B”¢C”¢D”¢E”¢FĮłÖÖŌŖĖŲ”£
¢ŁA”¢B”¢CŹĒ½šŹōŌŖĖŲ£¬Ī»ÓŚĶ¬Ņ»ÖÜĘŚ£¬Ō×ÓŗĖĶā¶¼ÓŠ3øöµē×Ó²ć£¬AµÄŌ×Ó°ė¾¶ŌŚĖłŹōÖÜĘŚÖŠ×ī“ó£¬ĒŅŌ×Ó°ė¾¶A>B>C”£
¢ŚD”¢EŹĒ·Ē½šŹōŌŖĖŲ£¬ĖüĆĒøśĒā»ÆŗĻæÉÉś³ÉĘųĢ¬Ēā»ÆĪļHDŗĶHE£¬ŌŚŹŅĪĀŹ±£¬DµÄµ„ÖŹŹĒŅŗĢ壬EµÄµ„ÖŹŹĒ¹ĢĢ唣
¢ŪFµÄµ„ÖŹŌŚ³£ĪĀĻĀŹĒĘųĢ壬ŠŌÖŹŗÜĪČ¶Ø£¬ŹĒ³żĒāĶā×īĒįµÄĘųĢ唣
Ēė»Ų“š£ŗ
£Ø1£©BĪ»ÓŚÖÜĘŚ±ķÖŠµŚ________ÖÜĘŚ______×壬CµÄŌ×Ó½į¹¹Ź¾ŅāĶ¼ŹĒ________”£
£Ø2£©Fµ„ÖŹµÄ»ÆѧŹ½ŹĒ________”£
£Ø3£©ŌŚÉĻŹöĮłÖÖŌŖĖŲÖŠ£¬×īøß¼ŪŃõ»ÆĪļ¶ŌÓ¦µÄĖ®»ÆĪļ¼īŠŌ×īĒæµÄĪļÖŹŹĒ_____£¬ĖįŠŌ×īĒæµÄĪļÖŹŹĒ________£¬ĘųĢ¬Ēā»ÆĪļ×īĪČ¶ØµÄŹĒ________”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com