
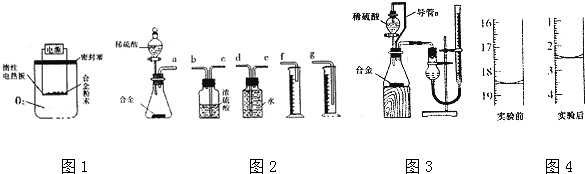
���� ��1����������������Һ��Ӧ����ƫ��������������
��2��þ������������Сʱ�������������������Ҫ������������Һ��࣬ʵ����Ҫ����������Һ�����Ӧ���ڻ�������ֵ���ݴ˼��㣻
��3�����ݷ�Ӧ��Ϊ���������Һ�����֣�Ҫ�õ����������Ҫ��ʵ��������
��4������Mg��Al���ܹ���������ѧ��Ӧ�����������������Ҫ֪�������ݣ�
��5���������þ���������ʵ��������ݺϽ�����������պ��������ʽ�����þ�����ʵ������ټ����þ������������
��6������װ�õ���װ˳�Ͻ����ᷴӦ������ˮ�������ⶨ���������������ʢˮ���Լ�ƿ����һ��Ҫ�̽�������������
��7���ٱ��ַ�Һ©��������ѹǿ����ƿ������ѹǿ��ȣ���Һ©������ʱϡ������˳�����£�������ƿ��ϡ����������ڽ����Һ©��������������Ӷ��������ڼ���ϡ������������������
�ڵζ��ܵ���ֵ��̶����Ϸ������ε����֮��Ϊ�ⶨ�������������ע��Ӧ���ָ������ζ�����Һ��ȸߣ����ռ�������ζ�����Һ��������������С��
�۸���ϡ���ᶨ����ƿ�ܹ��������ų�������
��� �⣺��1����������������Һ��Ӧ����ƫ����������������Ӧ����ʽΪ2Al+2NaOH+2H2O=2NaAlO2+3H2����
�ʴ�Ϊ��2Al+2NaOH+2H2O=2NaAlO2+3H2����
��2����þΪ3%ʱ���������ĺ�����ߣ�5.4g�Ͻ�����������Ϊ��5.4g����1-3%��=5.4��97%g����
2Al+2NaOH+2H2O=2NaAlO2 +3H2��
54g 2mol
5.4g��97% V��10-3L��2.0mol/L
����54g����5.4g��97%��=2mol����V��10-3L��2.0mol/L������ã�V=97����V��NaOH��Һ����97mL��
�ʴ�Ϊ��97��
��3����Ӧ���������ȫ���ģ�û�з�Ӧ��Ϊ����þ����������þ֮ǰ��Ҫ�������ˡ�ϴ�ӡ����������Ȼ���ٳ��������������Ӷ�������Ͻ���þ�ĺ�����
�ʴ�Ϊ�����ˡ�ϴ�ӡ�������壻
��4��Mg��Al����������Ӧ�����ɽ������������ⶨ�������������
�ʴ�Ϊ�����պ�����������
��5����xg��þ�Ͻ��ĩ�к���n molþ��zmol������24n+27z=x�٣�
�ٸ��ݷ�Ӧ��ϵʽ��Mg��MgO��Al��Al2O3�����õ���yg������ݴ���ʽΪ��40n+51z=y�ڣ�
���ݢ٢ڽ�ã�z=$\frac{3y-5x}{18}$mol����������Ϊ��27g/mol��$\frac{3y-5x}{18}$mol=$\frac{9y-15x}{2}$g���Ͻ���þ����������Ϊ��$\frac{x-\frac{9y-15x}{2}}{x}$=$\frac{17x-9y}{2x}$��
�ʴ�Ϊ��$\frac{17x-9y}{2x}$��
��6��װ�õ���װ˳�Ͻ����ᷴӦ������ˮ�������ⶨ���������������ʢˮ���Լ�ƿ����һ��Ҫ�̽���������������ѹǿԭ����ˮ�ų�����Ͳ��ˮ��������������������������Ͳ�ڵ���Ӧ������Ͳ�ײ���������˳��Ϊ����a���ӣ�e����d���ӣ�g����
�ʴ�Ϊ��e��d��g��
��7����װ���е���a�������ǣ����ַ�Һ©��������ѹǿ����ƿ������ѹǿ��ȣ���Һ©������ʱϡ������˳�����£�������ƿ��ϡ����������ڽ����Һ©��������������Ӷ��������ڼ���ϡ������������������
�ʴ�Ϊ�����ַ�Һ©��������ѹǿ����ƿ������ѹǿ��ȣ���Һ©������ʱϡ������˳�����£�������ƿ��ϡ����������ڽ����Һ©��������������Ӷ��������ڼ���ϡ������������������
�ڵζ��ܵ���ֵ��̶����Ϸ������ε����֮��Ϊ�ⶨ��������������ռ�������ζ�����Һ�������С����Ӧǰ�ζ��ܶ���Ϊ18.50mL����Ӧ��ζ��ܶ���Ϊ2.50mL�����Բⶨ���������Ϊ��18.50mL-2.50mL=16.00mL��
�ʴ�Ϊ��16.00mL��
������ϡ���������ƿ�У���ʹ������������Ҳ�Ὣƿ�ڿ����ų���ʹ�����������ƫ��ʵ�����ʱ�����ӹ��ƿ����Ͳ�ĵ�����������ˮ���ڣ�ʹ�����������ƫС��
�ʴ�Ϊ������ϡ���������ƿ�У���ʹ������������Ҳ�Ὣƿ�ڿ����ų���ʹ�����������ƫ��ʵ�����ʱ�����ӹ��ƿ����Ͳ�ĵ�����������ˮ���ڣ�ʹ�����������ƫС��
���� ���⿼�����ʺ����IJⶨ����ʵ��ԭ����װ�õ����⡢ʵ�鷽����Ƶȣ��Ѷ��еȣ�����ʵ��ԭ���ǽ���Ĺؼ����Ƕ�֪ʶ���ۺϿ��飬Ҫ��ѧ��������ʵ�Ļ������ۺ�����֪ʶ�������⡢��������������


 ����ȫ���ִʾ��ƪ��ϵ�д�
����ȫ���ִʾ��ƪ��ϵ�д� �����߿����ϵ�д�
�����߿����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �٢ڢ� | B�� | �٢ۢ� | C�� | �ڢۢ� | D�� | �٢ۢܢ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
 ��ͼ��Zn��Cu�γɵ�ԭ��أ�ijʵ��С��ʵ���¼��ͼ��ʾ�������������� ��������
��ͼ��Zn��Cu�γɵ�ԭ��أ�ijʵ��С��ʵ���¼��ͼ��ʾ�������������� ��������| ��CuΪ������ZnΪ���� ��Cu���������ݲ��� ��SO42-��Cu���ƶ� ������0.5mol�����������ߣ��ɲ���0.25mol���� �ݵ��ӵ�������Cu�����ߡ�Zn ��������Ӧʽ��Cu+2e-�TCu2+ |
| A�� | �٢ڢ� | B�� | �ڢ� | C�� | �ܢݢ� | D�� | �ۢܢ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �������ֽ� | B�� | ��������Һ������ķ�Ӧ | ||
| C�� | Ư��ʧЧ | D�� | ������������Ϊ�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
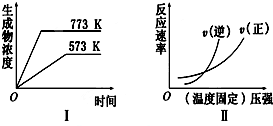
| A�� | 2SO3��g��?2SO2��g��+O2��g����H��0 | B�� | N2��g��+3H2��g��?2NH3��g����H��0 | ||
| C�� | 4NH3��g��+5O2��g��?4NO��g��+6H2O��g����H��0 | D�� | H2��g��+CO��g��?C��s��+H2O��g����H��0 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | 2mol/L | B�� | 3mol/L | C�� | 4mol/L | D�� | 5mol/L |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
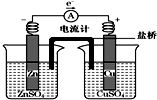
| A�� | �����ǽ��ݹ�KCl��Һ����֬�γɵģ�����Cl-������� | |
| B�� | ͭƬ�������� | |
| C�� | ������ͭƬ����������пƬ | |
| D�� | ͭ������ͭƬ���汻��ԭ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ���³�ѹ�£�11.2 L��������ԭ����ĿΪNA | |
| B�� | 2 L 0.2 mol/L K2SO4��ҺSO42-���ʵ���Ũ��Ϊ0.4 mol/L | |
| C�� | 1 mol Na����ԭ�����ṩ������ΪNA | |
| D�� | ͬ�¡�ͬѹ��NA��CO2���Ӻ�NA��O2���ӵ������ͬ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����

| A�� | �ù����ĩ��һ��������BaCl2 | |
| B�� | �ù����ĩ��һ������KNO3 | |
| C�� | ������ɿ�����CaCO3��BaCl2��CuSO4 | |
| D�� | �������һ����CaCO3��Na2SO4��KNO3 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com