
ĒæĖįÖĘČõĖįŹĒø“·Ö½ā·“Ó¦µÄŅ»ĢõÖŲŅŖ¹ęĀÉ”£ÕāĄļµÄ”°ĒæĖį”±”¢”°ČõĖį”±ÖøĻą¶ŌĒæČõ£¬ÄܳŹĻÖĖįŠŌµÄŅ»Š©·ĒĖįĄąĪļÖŹ£¬Čē·ÓĄą”¢Į½ŠŌĒāŃõ»ÆĪļ”¢ĖįŹ½ŃĪµČ²ĪÓėµÄ·“Ó¦Ņ²æÉøł¾ŻĘäĖįŠŌĒæČõŌĖÓĆÉĻŹö¹ęĀÉĄ“ÅŠ¶ĻĘä²śĪļ”£
(1)HA”¢H2BŹĒĮ½ÖÖČõĖį£¬ÓŠČēĻĀ¹ŲĻµ£ŗH2B(ÉŁĮæ)£«2A£=B2££«2HA£¬ŌņA£”¢HB£”¢B2£ČżÖÖĄė×ÓÖŠ£¬×īŅ×½įŗĻÖŹ×Ó(H£«)µÄŹĒ________”£
(2)ĖįŠŌĒæČõ³żÓėĪļÖŹµÄ±¾ŠŌÓŠ¹ŲĶā£¬»¹ÓėČܼĮÓŠ¹Ų£¬ČēCH3COOHÓėHFŌŚŅŗ°±ÖŠŹÜNH3Ó°ĻģæÉ·¢ÉśĶźČ«µēĄė”£ŌŚŅŗ°±ÖŠCH3COONa£«HClØD”śNaCl£«CH3COOHÕāŅ»·“Ó¦ÄÜ·ń·¢Éś________(Ģī”°ÄÜ”±»ņ”°·ń”±)£¬ĄķÓÉŹĒ____________________”£
(3)ijĶ¬Ń§ŹµŃé·¢ĻÖ£¬½«H2SĘųĢåĶØČėCuSO4ČÜŅŗÖŠ£¬Éś³ÉŗŚÉ«³Įµķ”£ÅŖĒå³ĮµķŹĒCuSŗ󣬊“³öĮĖ»Æѧ·½³ĢŹ½£ŗH2S£«CuSO4=CuS”ż£«H2SO4”£µ«ĖęŗóĖūĻŻČėĮĖĄ§»ó£ŗÕā²»ŹĒČõĖįÖʵĆĒæĖįĮĖĀš£æÓėĒæĖįÖĘČõĖįµÄ¹ęĀÉƬ¶ÜĮĖ”£ĒėÄć°ļÖś½āŹĶ__________________________________________”£
(4)Ńõ»Æ»¹Ō·“Ó¦ÖŠŅ²ÓŠĄąĖĘ¹ęĀÉ£ŗ”°ĒæŃõ»ÆŠŌĪļÖŹÖĘČõŃõ»ÆŠŌĪļÖŹ”±”¢”°Ē滹ŌŠŌĪļÖŹÖĘČõ»¹ŌŠŌĪļÖŹ”±£¬¾Ż“ĖÅŠ¶ĻĻĀĮŠ·“Ó¦Äܹ»·¢ÉśµÄŹĒ________(Ģī×ÖÄø±ąŗÅ)”£
A£®FeCl2£«Cl2 FeCl3
FeCl3
B£®Fe£«I2 FeI3
FeI3
C£®Fe£«CuSO4 FeSO4£«Cu
FeSO4£«Cu
D£®FeCl3£«Cu CuCl2£«FeCl2
CuCl2£«FeCl2
E£®FeBr3£«Cl2 FeCl2£«Br2
FeCl2£«Br2
F£®FeI2£«Br2 FeBr3£«I2
FeBr3£«I2

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
£Ø8·Ö£©½«0£®08mol KMnO4¹ĢĢå£ØÖŹĮæĪŖ12£®64g£©¼ÓČČŅ»¶ĪŹ±¼äŗó£¬ŹÕ¼Æµ½a molO2£»Ļņ·“Ó¦ŗó²ŠĮōµÄ¹ĢĢåÖŠ¼ÓČė×ćĮæµÄÅØŃĪĖį£¬ÓÖŹÕ¼Æµ½b molCl2£¬“ĖŹ±MnŌŖĖŲČ«²æŅŌMn2+µÄŠĪŹ½“ęŌŚÓŚČÜŅŗÖŠ”£
£Ø1£©ĒėÅäĘ½ĻĀĮŠ·½³ĢŹ½£ŗ
KMnO4+ HCl £ KCl+ MnCl2+ Cl2”ü+ H2O
£Ø2£©ÉĻŹ½·“Ó¦ÖŠµÄ»¹Ō¼ĮŹĒ £¬µ±»¹Ō¼ĮŹ§Č„1molµē×ÓŹ±£¬Ńõ»Æ²śĪļµÄĪļÖŹµÄĮæĪŖ ”£
£Ø3£©a +bµÄ×ī“óÖµĪŖ £¬a +b×īŠ”ÖµĪŖ ”£
£Ø4£©µ± a +b £½0£®18Ź±£¬²ŠĮō¹ĢĢåµÄÖŹĮæĪŖ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
(15·Ö)2012Äź4ŌĀ15ČÕ£¬ŃėŹÓĘŲ¹āŗÓ±±Ņ»Ņ©ÓĆ½ŗÄŅ³§ÓĆʤøļ·ĻĮĻĖłÉś²śĆ÷½ŗ×÷ŌĮĻ”£ÕāŠ©ĘóŅµÓĆÉśŹÆ»Ņ“¦Ąķʤøļ·ĻĮĻ½ųŠŠĶŃÉ«ĘÆ°×ŗĶĒåĻ“£¬Ėęŗó°¾ÖĘ³É¹¤ŅµĆ÷½ŗ£¬ĀōøųÕć½ŠĀ²żĻŲŅ©ÓĆ½ŗÄŅÉś²śĘóŅµ£¬×īÖÕĮ÷ĻņŅ©Ę·ĘóŅµ£¬½ųČėĻū·ŃÕßø¹ÖŠ”£¼ĒÕßµ÷²é·¢ĻÖ9¼ŅŅ©³§µÄ13øöÅś“ĪŅ©Ę·ĖłÓĆ½ŗÄŅÖŲ½šŹōøõŗ¬Į泬±ź”£ÖŲøõĖįÄĘĖ׳Ęŗģ·ÆÄĘ(Na2Cr2O7”¤2H2O)ŹĒÖŲŅŖµÄ»Æ¹¤²śĘ·ŗĶĒæŃõ»Æ¼Į”£¹¤ŅµÖʱøµÄĮ÷³ĢČēĻĀ£ŗ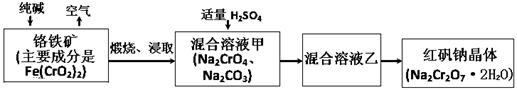
£Ø1£©»ÆѧÉĻæɽ«Ä³Š©ŃĪŠ“³ÉŃõ»ÆĪļµÄŠĪŹ½£¬ČēNa2SiO3æÉŠ“³ÉNa2O”¤SiO2£¬ŌņFe(CrO2)2æÉŠ“³É ”£
£Ø2£©ģŃÉÕøõĢśæóŹ±£¬æóŹÆÖŠÄŃČܵÄFe(CrO2)2Éś³ÉæÉČÜÓŚĖ®µÄNa2CrO4£¬·“Ó¦»Æѧ·½³ĢŹ½ČēĻĀ£ŗ4Fe(CrO2)2+8Na2CO3+7O2 = 2Fe2O3+8Na2CrO4+8CO2ĪŖĮĖ¼ÓæģøĆ·“Ó¦µÄ·“Ó¦ĖŁĀŹ£¬æɲÉČ”µÄ“ėŹ©ŹĒ ”££ØŠ“Ņ»ÖÖ¼“æÉ£©
£Ø3£©ŅŃÖŖCrO42-ŌŚ²»Ķ¬µÄĖįŠŌČÜŅŗÖŠÓŠ²»Ķ¬µÄ·“Ó¦£¬Čē£ŗ2CrO42-+2H+= Cr2O72-+H2O ;
3CrO42-+4H+= Cr3O102-+2H2O ¢Ł Ķł»ģŗĻČÜŅŗ¼×ÖŠ¼ÓČėĮņĖį±ŲŠėŹŹĮæµÄŌŅņŹĒ ”£
¢Ś »ģŗĻČÜŅŗŅŅÖŠČÜÖŹµÄ»ÆѧŹ½ŹĒ ”£
£Ø4£©ŌŚŗ¬Cr2O72-·ĻĖ®ÖŠ“ęŌŚ×ÅĘ½ŗā£ŗCr2O72-+H2O  2CrO42-+2H+£¬ĒėŠ“³öøĆĘ½ŗāµÄĘ½ŗā³£Źż±ķ“ļŹ½K= £¬Čō¼ĢŠų¼ÓĖ®Ļ”ŹĶ£¬Ę½ŗā½« ŅʶÆ(Ģī”°ÕżĻņ”±”¢”°ÄęĻņ”±”°²»”±)”£
2CrO42-+2H+£¬ĒėŠ“³öøĆĘ½ŗāµÄĘ½ŗā³£Źż±ķ“ļŹ½K= £¬Čō¼ĢŠų¼ÓĖ®Ļ”ŹĶ£¬Ę½ŗā½« ŅʶÆ(Ģī”°ÕżĻņ”±”¢”°ÄęĻņ”±”°²»”±)”£
£Ø5£©ĒėÅäĘ½¼īŠŌČÜŅŗ»¹Ō·ØÖŠ·¢ÉśµÄĄė×Ó·“Ó¦£ŗ”õCr2O72-+”õS2-+”õH2O - ”õCr(OH)3+”õS2O32-+”õOH-
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ŠŪ»Ę(As4S4)ŗĶ“Ę»Ę(As2S3)ŹĒĢįČ”ÉéµÄÖ÷ŅŖæóĪļŌĮĻ£¬¶žÕßŌŚ×ŌČ»½ēÖŠ
¹²Éś”£øł¾ŻĢāŅāĶź³ÉĻĀĮŠĢīæÕ£ŗ
(1)As2S3ŗĶSnCl2ŌŚŃĪĖįÖŠ·“Ó¦×Ŗ»ÆĪŖAs4S4ŗĶSnCl4²¢·Å³öH2SĘųĢ唣ČōAs2S3ŗĶSnCl2ÕżŗĆĶźČ«·“Ó¦£¬As2S3ŗĶSnCl2µÄĪļÖŹµÄĮæÖ®±ČĪŖ________”£
(2)ÉĻŹö·“Ó¦ÖŠµÄŃõ»Æ¼ĮŹĒ________£¬·“Ó¦²śÉśµÄĘųĢåæÉÓĆ________ĪüŹÕ”£
(3)As2S3ŗĶHNO3ÓŠČēĻĀ·“Ó¦£ŗAs2S3£«10H£«£«10NO3”Ŗ=2H3AsO4£«3S”ż£«10NO2”ü£«2H2O£¬ČōÉś³É2 mol H3AsO4£¬Ōņ·“Ó¦ÖŠ×ŖŅʵē×ÓµÄĪļÖŹµÄĮæĪŖ________”£Čō½«øĆ·“Ó¦Éč¼Ę³ÉŅ»Ōµē³Ų£¬ŌņNO2Ó¦øĆŌŚ________(Ģī”°Õż¼«”±»ņ”°øŗ¼«”±)ø½½üŅŻ³ö”£
(4)Čō·“Ó¦²śĪļNO2Óė11.2 L O2(±ź×¼×“æö)»ģŗĻŗóÓĆĖ®ĪüŹÕČ«²æ×Ŗ»Æ³ÉÅØĻõĖį£¬Č»ŗóÓė¹żĮæµÄC·“Ó¦£¬Ėł²śÉśµÄCO2µÄĮæ________(Ń”Ģī±ąŗÅ)”£
a£®Š”ÓŚ0.5 mol b£®µČÓŚ0.5 mol
c£®“óÓŚ0.5 mol d£®ĪŽ·ØČ·¶Ø
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
µŖŃõ»ÆĪļŹĒ“óĘųĪŪČ¾ĪļÖ®Ņ»£¬Ļū³żµŖŃõ»ÆĪļµÄ·½·ØÓŠ¶ąÖÖ”£
£Ø1£©ĄūÓĆ¼×Ķé“߻ƻ¹ŌµŖŃõ»ÆĪļ”£ŅŃÖŖ£ŗ
¢ŁCH4 (g)£«4NO2(g)£½4NO(g)£«CO2(g)£«2H2O(g£© ”÷H £½£574 kJ/mol
¢ŚCH4(g)£«4NO(g£©£½ 2N2(g)£«CO2(g)£«2H2O(g£© ”÷H £½£1160 kJ/mol
ŌņCH4 ½«NO2 »¹ŌĪŖN2 µÄČČ»Æѧ·½³ĢŹ½ĪŖ£ŗ ”£
£Ø2£©ĄūÓĆNH3“߻ƻ¹ŌµŖŃõ»ÆĪļ£ØSCR¼¼Źõ)”£øĆ¼¼ŹõŹĒÄæĒ°Ó¦ÓĆ×ī¹ć·ŗµÄŃĢĘųµŖŃõ»ÆĪļĶŃ³ż¼¼Źõ”£ ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ: ĪŖĢįøßµŖŃõ»ÆĪļµÄ×Ŗ»ÆĀŹæɲÉČ”µÄ“ėŹ©ŹĒ £ØŠ“³ö1Ģõ¼“æÉ£©”£
ĪŖĢįøßµŖŃõ»ÆĪļµÄ×Ŗ»ÆĀŹæɲÉČ”µÄ“ėŹ©ŹĒ £ØŠ“³ö1Ģõ¼“æÉ£©”£
£Ø3£©ĄūÓĆClO2Ńõ»ÆµŖŃõ»ÆĪļ”£Ęä×Ŗ»ÆĮ÷³ĢČēĻĀ: NO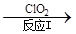 NO2
NO2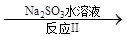 N2”£ŅŃÖŖ·“Ó¦¢ńµÄ»Æѧ·½³ĢŹ½ĪŖ2NO+ ClO2 + H2O £½NO2 + HNO3 + HCl£¬Ōņ·“Ó¦¢ņµÄ»Æѧ·½³ĢŹ½ŹĒ £»ČōÉś³É11.2 L N2£Ø±ź×¼×“æö£©£¬ŌņĻūŗÄClO2 g ”£
N2”£ŅŃÖŖ·“Ó¦¢ńµÄ»Æѧ·½³ĢŹ½ĪŖ2NO+ ClO2 + H2O £½NO2 + HNO3 + HCl£¬Ōņ·“Ó¦¢ņµÄ»Æѧ·½³ĢŹ½ŹĒ £»ČōÉś³É11.2 L N2£Ø±ź×¼×“æö£©£¬ŌņĻūŗÄClO2 g ”£
£Ø4£©ÓĆ»īŠŌĢ滹Ō·Ø“¦ĄķµŖŃõ»ÆĪļ£®ÓŠ¹Ų·“Ó¦ĪŖ£ŗC£Øs£©+2NO£Øg£© N2 £Øg£©+CO2 £Øg£©”÷H£®Ä³ŃŠ¾æŠ”×éĻņijĆܱÕČŻĘ÷¼ÓČėŅ»¶ØĮæµÄ»īŠŌĢæŗĶNO£¬ŗćĪĀ£ØT1”ę£©Ģõ¼žĻĀ·“Ó¦£¬·“Ó¦½ųŠŠµ½²»Ķ¬Ź±¼ä²āµĆø÷ĪļÖŹµÄÅضČČēĻĀ£ŗ
N2 £Øg£©+CO2 £Øg£©”÷H£®Ä³ŃŠ¾æŠ”×éĻņijĆܱÕČŻĘ÷¼ÓČėŅ»¶ØĮæµÄ»īŠŌĢæŗĶNO£¬ŗćĪĀ£ØT1”ę£©Ģõ¼žĻĀ·“Ó¦£¬·“Ó¦½ųŠŠµ½²»Ķ¬Ź±¼ä²āµĆø÷ĪļÖŹµÄÅضČČēĻĀ£ŗ
| ÅضČ/mol?L-1/ Ź±¼ä/min | NO | N2 | CO2 |
| 0 | 0.100 | 0 | 0 |
| 10 | 0.058 | 0.021 | 0.021 |
| 20 | 0.040 | 0.030 | 0.030 |
| 30 | 0.040 | 0.030 | 0.030 |
| 40 | 0.032 | 0.034 | 0.017 |
| 50 | 0.032 | 0.034 | 0.017 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ĄūÓĆŗ£Ė®æÉŅŌĢįČ”äåŗĶĆ¾£¬ĢįČ”¹ż³ĢČēĻĀ”£
(1)ĢįČ”äåµÄ¹ż³ĢÖŠ£¬¾¹żĮ½“ĪBr£”śBr2×Ŗ»ÆµÄÄæµÄŹĒ________£¬ĪüŹÕĖžÖŠ·¢Éś·“Ó¦µÄĄė×Ó·½³ĢŹ½ŹĒ__________”£ÓĆĘ½ŗāŅʶÆŌĄķ½āŹĶĶØæÕĘųµÄÖ÷ŅŖÄæµÄŹĒ_______”£
(2)“ÓMgCl2ČÜŅŗÖŠµĆµ½MgCl2”¤6H2O¾§ĢåµÄÖ÷ŅŖ²Ł×÷ŹĒ________________”¢¹żĀĖ”¢Ļ“µÓ”¢øÉŌļ”£
(3)ŅĄ¾ŻÉĻŹöĮ÷³Ģ£¬Čō½«10 m3ŗ£Ė®ÖŠµÄäåŌŖĖŲ×Ŗ»ÆĪŖ¹¤Ņµä壬ÖĮÉŁŠčŅŖ±ź×¼×“æöĻĀCl2µÄĢå»żĪŖ________L(ŗöĀŌCl2µÄČܽā)”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
(1)µē½ā±„ŗĶŹ³ŃĪĖ®Ö®Ē°Ź³ŃĪĖ®ŠčŅŖ¾«ÖĘ£¬ÄæµÄŹĒ³żČ„“ÖŃĪÖŠµÄCa2£«”¢Mg2£«”¢SO42£µČŌÓÖŹĄė×Ó£¬Ź¹ÓƵďŌ¼ĮŗĶ²Ł×÷ÓŠa.Na2CO3ČÜŅŗ£¬b.Ba(OH)2ČÜŅŗ£¬c.Ļ”ŃĪĖį£¬d.¹żĀĖ£¬ĘäŗĻĄķµÄ¼ÓČėĖ³ŠņĪŖ________(Ģī×ÖÄø“śŗÅ)”£
(2)µē½ā±„ŗĶŹ³ŃĪĖ®Ź±Ąė×Ó½»»»Ä¤µÄ×÷ÓĆŹĒ_____________________________
(3)µē½ā±„ŗĶŹ³ŃĪĖ®Ź±£¬Čē¹ūŌŚČŻ»żĪŖ10 LµÄĄė×Ó½»»»Ä¤µē½ā²ŪÖŠ£¬1 minŌŚŃō¼«æɲśÉś11.2 L(±ź×¼×“æö)Cl2£¬ÕāŹ±ČÜŅŗµÄpHŹĒ(ÉčĢå»żĪ¬³Ö²»±ä)________”£
(4)Cl2³£ÓĆÓŚ×ŌĄ“Ė®µÄɱ¾śĻū¶¾£¬ĻÖÓŠŅ»ÖÖŠĀŠĶĻū¶¾¼ĮClO2£¬ČōĖüĆĒŌŚÉ±¾ś¹ż³ĢÖŠ»¹Ō²śĪļ¾łĪŖCl££¬Ļū¶¾µČĮæµÄ×ŌĄ“Ė®£¬ĖłŠčCl2ŗĶClO2µÄĪļÖŹµÄĮæÖ®±ČĪŖ________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
Ļņ100 mL FeI2ČÜŅŗÖŠÖš½„ĶØČėCl2£¬»įŅĄ“ĪÉś³ÉI2”¢Fe3£«”¢IO3-£¬ĘäÖŠFe3£«”¢I2µÄĪļÖŹµÄĮæĖęn(Cl2)µÄ±ä»ÆČēĶ¼ĖłŹ¾£¬Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ÓÉĶ¼æÉÖŖ£¬I£”¢Fe2£«”¢I2ČżÖÖĮ£×ӵĻ¹ŌŠŌÓÉĒæµ½ČõµÄĖ³ŠņĪŖ________£¾________£¾________£»
£Ø2£©µ±n(Cl2)£½0.12 molŹ±£¬ČÜŅŗÖŠµÄĄė×ÓÖ÷ŅŖĪŖ________________________________£¬
“ÓæŖŹ¼ĶØČėCl2µ½n(Cl2)£½0.12 molŹ±µÄ×Ü·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ______________________£»
£Ø3£©µ±ČÜŅŗÖŠn(Cl£)”Ćn(IO3-)£½8”Ć1Ź±£¬ĶØČėµÄCl2ŌŚ±ź×¼×“æöĻĀµÄĢå»żĪŖ________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
H2O2ŌŚ¹¤Ņµ”¢Å©Ņµ”¢Ņ½Ņ©ÉĻ¶¼ÓŠ¹ć·ŗµÄÓĆĶ¾”£
£Ø1£©H2O2ŹĒ¶žŌŖČõĖį£¬Š“³öµŚŅ»²½µÄµēĄė·½³ĢŹ½ £¬µŚ¶ž²½µÄµēĄėĘ½ŗā³£Źż±ķ“ļŹ½Ka2£½ ”£
£Ø2£©Šķ¶ąĪļÖŹ¶¼æÉŅŌ×öH2O2·Ö½āµÄ“߻ƼĮ”£Ņ»ÖÖ¹ŪµćČĻĪŖ£ŗŌŚ·“Ó¦¹ż³ĢÖŠ“߻ƼĮĻȱ»H2O2Ńõ»Æ£Ø»ņ»¹Ō£©£¬ŗóÓÖ±»H2O2»¹Ō£Ø»ņŃõ»Æ£©”£ĻĀĮŠĪļÖŹ¶¼æÉ×öH2O2·Ö½āµÄ“߻ƼĮ£¬ŌŚ·“Ó¦¹ż³ĢÖŠĻȱ»Ńõ»Æ£¬ŗó±»»¹ŌµÄŹĒ ”£
¢ŁI£ ¢ŚFe3£« ¢ŪCu2£« ¢ÜFe2£«
£Ø3£©ÓĆ¼īŠŌĒāŃõČ¼ĮĻµē³ŲŗĻ³ÉH2O2£¬¾ßÓŠŠ§ĀŹøߣ¬ĪŽĪŪČ¾µČĢŲµć”£µē³Ų×Ü·“Ó¦ĪŖ£ŗ
H2 + O2 + OH£ £½ H2O + HO2£”£Š“³öÕż¼«·“Ó¦Ź½£ŗ ”£
£Ø4£©H2O2ŹĒŅ»ÖÖ»·¾³ÓŃŗƵÄĒæŃõ»Æ¼Į”£µē¶Ę·ĻĖ®£ØÖ÷ŅŖŗ¬Cu2£«”¢Ni2£«£¬»¹ŗ¬ÉŁĮæFe3£«”¢Fe2£«”¢Cr3£« µČ£©ÖʱøĮņĖįÄųµÄŅ»ÖÖĮ÷³ĢČēĻĀ£ŗ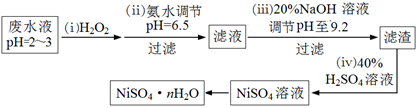
¢ŁµŚ£Ø¢”£©²½£¬¼ÓČėH2O2·“Ó¦µÄĄė×Ó·½³ĢŹ½ ”£
¢ŚµŚ£Ø¢¢£©²½£¬ĀĖŌüÖŠµÄÖ÷ŅŖ³É·ÖŌŚŅ½ĮĘÉĻµÄÓĆĶ¾ŹĒ ”£
¢ŪĪŖ²ā¶ØNiSO4”¤n H2OµÄ×é³É£¬½ųŠŠČēĻĀŹµŃé£ŗ³ĘČ”2.627gѳʷ£¬ÅäÖĘ³É250.00 mLČÜŅŗ”£×¼Č·ĮæČ”ÅäÖʵÄČÜŅŗ25.00 mL£¬ÓĆ0.04000 mol”¤L£1µÄEDTA£ØNa2H2Y£©±ź×¼ČÜŅŗµĪ¶ØNi2+£ØĄė×Ó·½³ĢŹ½ĪŖNi2£«+ H2Y2££½NiY2£+ 2H£«£©£¬ĻūŗÄEDTA±ź×¼ČÜŅŗ25.00 mL”£ŌņĮņĖįÄų¾§ĢåµÄ»ÆѧŹ½ĪŖ ”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com