
���������ߵ�ֱ��Ӱ��������������������Խ��Խ�ܵ����ǵĹ�ע������Ⱦ�Ŀ��������ʵijɷ��ж��֣����м��롶���������ձ���������Ⱦָ������Ŀ��SO2��CO��NO2��O3�Ϳ����������ȡ�
��ش��������⣺
��1��S��N��O�ĵĵ�һ�������ɴ�С��˳��Ϊ ��
��2��SO2��CO��NO2��O3�����¾�Ϊ���壬��̬ʱ������ ���塣
��3����������������������ߣ����ڵĻ�����ȫ��ʳƷ��ȫԽ��ԽΪ��������ע����ȩ��HCHO����������Ҫ������Ⱦ��֮һ����е��ǨC19.5 �棩���״���CH3OH���ǡ��پơ��е���Ҫ�к����ʣ���е���64.65 �棩����ȩ������Cԭ�Ӳ�ȡ �ӻ������ʽ���״��ķе����Ը��ڼ�ȩ����Ҫԭ���ǣ�
__________ ��
��4��CuCl��������Һ�ܹ���CO������Ӧ��CuCl+CO+H2O=Cu(CO)Cl��H2O���÷�Ӧ�����ڲⶨ������CO������
��д��ͭԭ�ӵĻ�̬�����Ų�ʽ ��
��CuCl�ľ���ṹ����ͼ����ʾ����ͬһ��Cl���������������Cu���� ����
��Cu(CO)Cl��H2O�Ľṹ����ͼ����ʾ��ͼ�б�ʾ��8���ǹ��ۼ������� ������λ����

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���������ߵ�ֱ��Ӱ��������������������Խ��Խ�ܵ����ǵĹ�ע������Ⱦ�Ŀ��������ʵijɷ��ж��֣����м��롶���������ձ���������Ⱦָ������Ŀ��SO2��CO��NO2��O3�Ϳ����������ȡ�
��ش��������⣺
��1��S��N��O�ĵĵ�һ�������ɴ�С��˳��Ϊ ��
��2��SO2��CO��NO2��O3�����¾�Ϊ���壬��̬ʱ������ ���塣
��3����������������������ߣ����ڵĻ�����ȫ��ʳƷ��ȫԽ��ԽΪ��������ע����ȩ��HCHO����������Ҫ������Ⱦ��֮һ����е��ǨC19.5 �棩���״���CH3OH���ǡ��پơ��е���Ҫ�к����ʣ���е���64.65 �棩����ȩ������Cԭ�Ӳ�ȡ �ӻ������ʽ���״��ķе����Ը��ڼ�ȩ����Ҫԭ���ǣ�
__________ ��
��4��CuCl��������Һ�ܹ���CO������Ӧ��CuCl+CO+H2O=Cu(CO)Cl��H2O���÷�Ӧ�����ڲⶨ������CO������
��д��ͭԭ�ӵĻ�̬�����Ų�ʽ ��
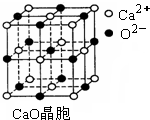
��CuCl�ľ���ṹ����ͼ����ʾ����ͬһ��Cl���������������Cu���� ����
��Cu(CO)Cl��H2O�Ľṹ����ͼ����ʾ��ͼ�б�ʾ��8���ǹ��ۼ������� ������λ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010-2011ѧ�������и���5���¿������ۣ���ѧ���� ���ͣ�ѡ����
���������ߵ�ֱ��Ӱ�����������������������������ձ����п�����Ⱦָ������Ŀ��SO2�� CO��NOx��O3�Ϳ����������ȡ�����˵������ȷ���� �� ��
A�������еij�����Ե����кܺõı������ã����Դ����к��д���O3����������
B���ڴ��������£�CO��NOx��ת��Ϊ������
C��Ѫ�쵰���к���Fe2+��NO��CO������Ѫ�쵰��ϳ��ȶ����ʶ�ʹ���ж�
D��SO2��NOx�Ĵ����ŷŻᵼ������IJ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010-2011ѧ�긣��ʡ�����и�����Ӧ����ϰ�����ۣ���ѧ���� ���ͣ������
��13�֣�
���������ߵ�ֱ��Ӱ��������������������Խ��Խ�ܵ����ǵĹ�ע������Ⱦ�Ŀ��������ʵijɷ��ж��֣����м��롶���������ձ���������Ⱦָ������Ŀ��SO2��CO��NO2��O3�Ϳ����������ȡ�
��ش��������⣺
��1��S��N��O�ĵĵ�һ�������ɴ�С��˳��Ϊ ��
��2��SO2��CO��NO2��O3�����¾�Ϊ���壬��̬ʱ������ ���塣
��3����������������������ߣ����ڵĻ�����ȫ��ʳƷ��ȫԽ��ԽΪ��������ע����ȩ��HCHO����������Ҫ������Ⱦ��֮һ����е��ǨC19.5 �棩���״���CH3OH���ǡ��پơ��е���Ҫ�к����ʣ���е���64.65 �棩����ȩ������Cԭ�Ӳ�ȡ �ӻ������ʽ���״��ķе����Ը��ڼ�ȩ����Ҫԭ���ǣ�__________ ��
��4��CuCl��������Һ�ܹ���CO������Ӧ��CuCl+CO+H2O=Cu(CO)Cl��H2O���÷�Ӧ�����ڲⶨ������CO������
��д��ͭԭ�ӵĻ�̬�����Ų�ʽ ��
��CuCl�ľ���ṹ����ͼ����ʾ����ͬһ��Cl���������������Cu���� ����

��Cu(CO)Cl��H2O�Ľṹ����ͼ����ʾ��ͼ�б�ʾ��8���ǹ��ۼ�������6������λ��������ͼ���ü�ͷ��ʾ����
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com