
���绷�����˽���ȫ���ֹʹ��������������ˮ����������������ø�Ч����ɫ���������������ȡ�����������һ�ּ��ױ�ը��ǿ���������壬������ˮ�����ȶ����ʻ���ɫ����������ʹ��ʱ���뾡����ϡ���������ϡ�ͣ�ͬʱҪ������ա�����ȡ�ʵ�����Ե�ⷨ�Ʊ�ClO2���������£���ش��������⡣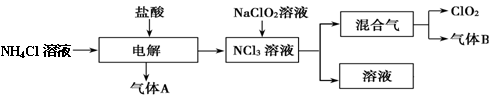
��1��ClO2������ԭ�� ����ǡ����ǡ���������8���ӽṹ����ͼ��ʾ��ⷨ�ƵõIJ�������������B��ʹʯ����Һ����ɫ����ȥ���������ѡ�� ��
A������ʳ��ˮ B����ʯ�� C��Ũ���� D������ˮ
��2���ȶ��Զ���������Ϊ�ƹ�������ȶ����������Ͳ�Ʒ������˵����ȷ���� ��
A���������ȿɹ㷺���ڹ�ҵ������ˮ����
B��Ӧ����ʳƷ��ҵ������Ч���ӳ�ʳƷ������
C���ȶ��Զ������ȵij��ִ�������˶������ȵ�ʹ�÷�Χ
D���ڹ������ͳ�Ʒ�������ڣ�Ҫ��ͨ��װ�úͼ�⼰����װ��
��3��ŷ������Ҫ��������������Ũ�����Ʊ�����ѧ��Ӧ����ʽΪ ���˷�ȱ����Ҫ�Dz��ʵ͡���Ʒ���Է��룬��������Ⱦ������
��4���ҹ��㷺���þ��������ϡ�͵�����������������ƣ�NaClO2����Ӧ�Ʊ�����ѧ����ʽ�� ���˷����ŷ�������ŵ��� ��
��5����ѧ��������о�����һ���µ��Ʊ����������������ữ�IJ��ᣨH2C2O4����Һ��ԭ�����ƣ���ѧ��Ӧ����ʽΪ ���˷���������������桢����İ�ȫ�ԣ�ԭ���� ��
��1������ C ��2��A��B��C��D
��3��2NaClO3��4HCl(Ũ) =2NaCl��Cl2����2ClO2����2H2O
��4��2NaClO2 + Cl2 ="2NaCl" + 2ClO2 ��ȫ�Ժã�û�в����ж�����Ʒ
��5��H2C2O4��2NaClO3��H2SO4 = Na2SO4��2CO2����2ClO2����2H2O
��Ӧ���������ɵĶ�����̼��ϡ������
���������������1����ClO2��ÿ��Clԭ����2��Oԭ���γ����ĶԹ��õ��Ӷԡ�����Oԭ�ӵõ���8���ӵ��ȶ��ṹ����Clԭ������11���������ӡ���˲��Ƕ�����8���ӽṹ������ͼ��ʾ��ⷨ�ƵõIJ�������������B��ʹʯ����Һ����ɫ ���������ΪNH3����ȥ������ʱҪ���������ռ������ա����ѡ��ΪC����2��A. ��Ч����ɫ���������������ȡ����Զ������ȿɹ㷺���ڹ�ҵ������ˮ��������ȷ��B.���ڶ���������һ��ǿ���������壬ʳƷ���ڸû����оͿ��Է�ֹ���������︯ʴ���������Ч���ӳ�ʳƷ�����ڡ���ȷ��C���������������Ķ������Ȳ��ȶ������ױ�ը��Ҫ������ա�����ȡ�ʹ��ʹ�ô���ܵ������ƣ����ȶ��Զ������ȵij��ֱؽ������Ӷ������ȵ�ʹ�÷�Χ�����õķ�����ǿ�������ԡ���ȷ��D.����������һ�ּ��ױ�ը��ǿ����������,���ȶ����ʻ���ɫ����������ʹ��ʱ���뾡����ϡ���������ϡ�ͣ�ͬʱҪ������ա�����ȡ������ڹ������ͳ�Ʒ�������ڣ�Ҫ��ͨ��װ�úͼ�⼰����װ�á���ȷ����3��ŷ������Ҫ��������������Ũ�����Ʊ�ClO2�Ļ�ѧ��Ӧ����ʽΪ2NaClO3��4HCl(Ũ)��2NaCl��Cl2����2ClO2����2H2O����4���ҹ��㷺���þ��������ϡ�͵�����������������ƣ�NaClO2����Ӧ�Ʊ�ClO2�Ļ�ѧ����ʽ��2NaClO2 + Cl2��2NaCl + 2ClO2���ɷ���ʽ���Կ������˷���Ϊ��������к�����Cl2����̬������٣���ը�Ը�С��������ŷ�������ŵ��ǰ�ȫ�Ժã�û�в����ж�����Ʒ����5�����������ữ�IJ��ᣨH2C2O4����Һ��ԭ��������ȡClO2�Ļ�ѧ��Ӧ����ʽΪH2C2O4��2NaClO3��H2SO4 = Na2SO4��2CO2����2ClO2����2H2O���˷������ڷ�Ӧ���������ɵĶ�����̼��ϡ�����ã�������������������桢����İ�ȫ�ԡ�
���㣺�����Ч����ɫ���������������ȵķ��ӽṹ�����ʼ�������ȡ�����Լ����ӵıȽϵ�֪ʶ��


 ��У���һ��ͨϵ�д�
��У���һ��ͨϵ�д� �γ̴����Ծ�����100��ϵ�д�
�γ̴����Ծ�����100��ϵ�д� �¾�����ĩ���100��ϵ�д�
�¾�����ĩ���100��ϵ�д� ȫ�ܴ���100��ϵ�д�
ȫ�ܴ���100��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ͭ���ʼ��仯������Ӧ�ü���㷺�����ʡ�
(1)ͭ�������������������ᷢ���û���Ӧ����������ͭ����Ũ������У����п�ȼ�����弰��ɫ�������ɣ���֪�����ԣ�Cu2��>I2����ͭ������ᷴӦ�Ļ�ѧ����ʽΪ__________________________________
(2)��֪Cu2O�����ڴ�����Һ�������У�ͬʱ�õ���ɫ��Һ�ͺ�ɫ���壬��Cu2O��ϡ���ᷴӦ�����ӷ���ʽΪ____________________________________��
Cu2O��ϡ���ᷴӦ�����ӷ���ʽΪ_____________________________��
ֻ��ϡ������ȷ��ij��ɫ������ Cu2O��Cu��ɵĻ����ķ�������ȡm g�ú�ɫ������������ϡ�����У���ַ�Ӧ����ˣ�Ȼ��___________________��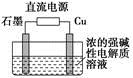
(3)Cu2O��һ�ְ뵼����ϣ�������ɫ��ѧ������Ƶ���ȡCu2O�ĵ��װ����ͼ��ʾ������ܷ�Ӧ��2Cu��H2O Cu2O��H2������ʯīӦ���Դ��________��������ͭ�缫�ϵĵ缫��ӦʽΪ________���������У���������Χ��ҺpH________(��������С�����䡱)��
Cu2O��H2������ʯīӦ���Դ��________��������ͭ�缫�ϵĵ缫��ӦʽΪ________���������У���������Χ��ҺpH________(��������С�����䡱)��
(4)����Cu��Cu2O��CuO��ɵĻ�����м���1 L 0.6 mol/L HNO3ǡ��ʹ������ܽ⣬ͬʱ�ռ���2 240 mL NO(��״��)�����������������������������ԭ�����ù��������Ϊ________����������к���0.1 mol Cu�����û������ϡ�����ַ�Ӧ������������������ʵ���Ϊ________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���ʼ��仯�����ڹ�ũҵ������������Ҫ��Ӧ�á�
��1����֪25��ʱ��SO2��g����2CO��g����2CO2��g����1/xSx��s�� ��H��akJ/mol
2COS��g����SO2��g����2CO2��g����3/xSx��s�� ��H��bkJ/mol��
��COS��g������CO��g����Sx��s����Ӧ���Ȼ�ѧ����ʽ�� ��
��2���ۻ�(As4S4)�ʹƻ�(As2S3)����ȡ�����Ҫ����ԭ�ϡ���֪As2S3��HNO3�����·�Ӧ��As2S3+10H++ 10NO3?=2H3AsO4+3S+10NO2��+ 2H2O��������H3AsO4�����ʵ���Ϊ0.6 mol��Ӧ��ת�Ƶ��ӵ���ĿΪ ��
��3��������ʵ���Ũ��Na2S��NaOH�����Һ�еμ�ϡ����������������H2S��HS?��S2?�ķֲ�������ƽ��ʱij���ֵ�Ũ��ռ������Ũ��֮�͵ķ�������μ���������Ĺ�ϵ����ͼ��ʾ�����Եμӹ���H2S������ݳ�����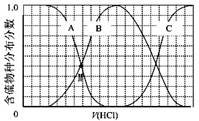
��B��ʾ ��
�ڵμӹ����У���Һ����Ũ�ȴ�С��ϵ��ȷ���� (����ĸ)��
a��c(Na+)= c(H2S)+c(HS?)+2c(S2?)
b��2c(Na+)=c(H2S)+c(HS?)+c(S2?)
c��c(Na+)=3[c(H2S)+c(HS?)+c(S2?)]
��NaHS��Һ�ʼ��ԣ����μ�������M��ʱ����Һ�и�����Ũ���ɴ�С��˳��Ϊ ��
��4����ҵ������·ѭ����������������Ĺ�����������ͼ��ʾ��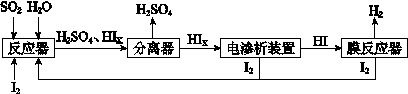
�� д����Ӧ���з�����Ӧ�Ļ�ѧ����ʽ�� ��
�� ������װ����ͼ��ʾ��д�������ĵ缫��Ӧʽ ����װ���з������ܷ�Ӧ�Ļ�ѧ����ʽ�� ��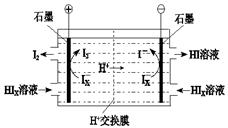
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������Խ����Ⱥ͵�֮�䣬������һ���ʽ����������⡣
(1)����Ϊ���������������ַ��ӻ����ӵ��Լ����뵽����Br������Һ�У����Խ�Br������ΪBr2__________��
| A��I2 | B��I | C��Cl2 | D��Cl�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(1)��NaBr��KI�Ļ����Һ�У�ͨ��������Cl2����Һ���ɲ����գ������������ʣ���������________��д����Ӧ�Ļ�ѧ����ʽ��________________________________��
(2)��KI��Һ�е��������Һ��������____________________���ٵ�����ˮ��������________���йط�Ӧ�����ӷ���ʽΪ______________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��1�����Ϸ�����(��Ҫ�ɷ�V2O5)��ϡ���ᡢ���������Һ��ϣ���ַ�Ӧ��������Һ�����ԣ���VO2����K����SO42-�ȡ�д���÷�Ӧ�Ļ�ѧ����ʽ_________________________________��
��2��������������Һ�м���KClO3��Һ����ַ�Ӧ����Һ����������VO2+��Cl����д������ƽ�÷�Ӧ�����ӷ���ʽ�����������ת�Ƶ���Ŀ�ͷ���______________________��
��3����20.00 mL��0.1 mol��L��1 VO2+��Һ�У�����0.195 gп�ۣ�ǡ����ɷ�Ӧ����ԭ���������______________________________________________________________��
a��V b��V2�� c��VO2+ d��VO2��
��4����֪V2O5�ܺ����ᷴӦ����������VO2��������дһ�����ӷ�Ӧ����ʽ��˵����ԭ�ԣ�SO32-��Cl����VO2��__________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������ʵĵ���ƽ�⡢�����ˮ��ƽ�����������ܽ�ƽ������ڻ�ѧƽ�⡣
��.��֪H2A��ˮ�д�������ƽ�⣺H2A=H����HA����HA��??H����A2����
��1��������NaHA��Һ��pH________(�����)��ԭ����_________________��
A������7 B����7
C������7 D����ȷ��
��2��ij�¶��£�����0.1 mol��L��1��NaHA��Һ����εμ�0.1 mol��L��1KOH��Һ����Һ������(���Ի�Ϻ���Һ������仯)����ʱ�û����Һ�е����й�ϵһ����ȷ����________��
A��c(H��)��c(OH��)��1.0��10��14
B��c(Na��)��c(K��)��c(HA��)��2c(A2��)
C��c(Na��)��c(K��)
D��c(Na��)��c(K��)��0.05 mol��L��1
��3����֪������H2A�ĸ���(CaA)�ı�����Һ�д�������ƽ�⣺CaA(s)??Ca2��(aq)��A2��(aq)����H��0����Ҫʹ����Һ��Ca2��Ũ�ȱ�С���ɲ�ȡ�Ĵ�ʩ��________��
A�������¶� B�������¶�
C������NH4Cl���� D������Na2A����
��.����Cr2O72-�ķ�ˮ���Խϴ�ij������ˮ�к�5.0��10��3 mol��L��1��Cr2O72-��Ϊ��ʹ��ˮ���ŷŴ�꣬�������´�����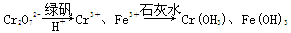
(1)�÷�ˮ�м����̷���H����������Ӧ�����ӷ���ʽΪ____________________��
(2)��������ķ�ˮ�в�����c(Fe3��)��2.0��10��13 mol��L��1���������Cr3����Ũ��Ϊ________��
(��֪��Ksp[Fe(OH)3]��4.0��10��38��Ksp[Cr(OH)3]��6.0��10��31)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����������Ч����SO2�Կ�������Ⱦ��
(1)��ú�м���ʯ��ʯ�ɼ���ȼ�ղ�����SO2�ĺ������÷�Ӧ�Ļ�ѧ����ʽ��
_______________________________��
(2)��ˮ�������ԣ���Ҫ����Na����K����Ca2����Mg2����Cl����SO42����Br����HCO3���ȡ���SO2�����������ú�ˮ�����乤��������ͼ��ʾ��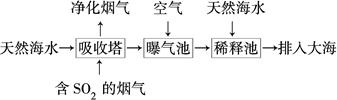
������������ͨ�������Ŀ����_____________________________________��
��ͨ��������������к�ˮ����Ȼ��ˮ��ȣ�Ũ�������Բ�ͬ��������________��
a��Cl�������� b��SO42�������� c��Br�������� d��HCO3��
(3)��NaOH��Һ���������е�SO2�������õ�Na2SO3��Һ���е�⣬�ɵõ�NaOH��ͬʱ�õ�H2SO4����ԭ����ͼ��ʾ(�缫����Ϊʯī)��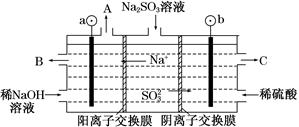
��ͼ��a��Ҫ���ӵ�Դ��________(���������)����C��������������________��
��SO32���ŵ�ĵ缫��ӦʽΪ____________________________��
�۵�����������������������ǿ����ƽ���ƶ���ԭ������ԭ��
__________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ס�����ͬѧ�о�Na2SO3��Һ��FeCl3��Һ��Ӧ�������
| ���� | ���� | ���� |
| I | ��2 mL 1 mol��L-1FeCl3��Һ�м���һ������Na2SO3��Һ | ��Һ���ػ�ɫ��Ϊ���ɫ�� ���������̼�����ζ�������ݳ� |
 SO32- +
SO32- + =
= F e2+ +
F e2+ + +
+

| 1��Fe2+��SO32-��Ӧ����ī��ɫ����״����FeSO3�� 2��ī��ɫ��FeSO3���ɫ��FeCl3��Һ��Ϻ���Һ�ʺ��ɫ�� |
| ���� | ���� | ���� |
| II | �ü�������䲽��I�еĺ��ɫ��Һ | ���֡������ЧӦ�� |
| ���� | ���� | ���� |
| III | ��1 mol?L-1��FeCl3��Һ��ͨ��һ������SO2 | ��Һ�ɻ�ɫ��Ϊ���ɫ |
| IV | �ü�������䲽��III�еĺ��ɫ��Һ | û�г��֡������ЧӦ�� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com