
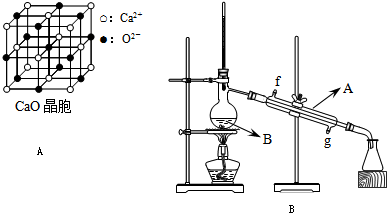
ÓŠa”¢bĮ½øöµČĢå»żĒŅ¼«Ņ×µ¼ČȵÄĆܱÕČŻĘ÷£¬a±£³ÖČŻ»ż²»±ä£¬bÖŠµÄ»īČūæÉÉĻĻĀŅĘ¶Æ£¬ŅŌ±£³ÖÄŚĶāŃ¹ĒæĻąµČ£®ŌŚĻąĶ¬Ģõ¼žĻĀ½«3 mol””A£¬1 mol””B·Ö±šĶ¬Ź±»ģŗĻÓŚa”¢bĮ½ČŻĘ÷ÖŠ£¬·¢Éś·“Ó¦£ŗ3A(g)£«B(g)![]() 2C(g)£«D(g)
2C(g)£«D(g)
(1)“ļµ½Ę½ŗāŹ±£¬aÖŠAµÄÅضČĪŖM mol/L£®AµÄ×Ŗ»ÆĀŹĪŖN£¬bÖŠAµÄÅضČĪŖm mol/L£¬AµÄ×Ŗ»ÆĀŹĪŖn£¬ŌņM________m£»N________n(Ģī£¾”¢£¼”¢£½”¢ĪŽ·Ø±Č½Ļ)
(2)±£³ÖĪĀ¶Č²»±ä£¬°“ĻĀĮŠÅä±Č·Ö±š³äČėa”¢bĮ½ČŻĘ÷£¬“ļĘ½ŗāŗó£¬aÖŠAµÄÅضČĪŖM mol”¤L£1µÄŹĒ________£®
A£®6 mol””A£«2 mol””B
B£®3 mol””A£«2 mol””C
C£®2 mol””C£«1 mol””B£«1 mol””D
D£®2 mol””C£«1 mol””D
E£®1.5 mol””C£«0.5 mol””B£«1 mol””C£«0.5 mol””D

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
| ||
| ||


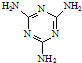 £©Ė×³Ę”°µ°°×¾«”±£®¶ÆĪļÉćČėČż¾ŪĒč°·ŗĶČż¾ŪĒčĖį£Ø
£©Ė×³Ę”°µ°°×¾«”±£®¶ÆĪļÉćČėČż¾ŪĒč°·ŗĶČż¾ŪĒčĖį£Ø 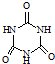 £©ŗó£¬Čż¾ŪĒčĖįÓėČż¾ŪĒč°··Ö×ÓĻą»„Ö®¼äĶعż
£©ŗó£¬Čż¾ŪĒčĖįÓėČż¾ŪĒč°··Ö×ÓĻą»„Ö®¼äĶعż| »ÆŗĻĪļ | ĆܶČ/g?cm-3 | ·Šµć/”ę | Čܽā¶Č/100gĖ® |
| Õż¶”“¼ | 0.810 | 118.0 | 9 |
| ±ł“×Ėį | 1.049 | 118.1 | ”Ž |
| ŅŅĖįÕż¶”õ„ | 0.882 | 126.1 | 0.7 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2011-2012ѧğ°²»ÕŹ”°ö²ŗŹŠø߶žµŚ¶žŃ§ĘŚĘŚÖŠĮŖæ¼ŹŌ»ÆѧŹŌ¾ķ£Ø“ų½āĪö£© ĢāŠĶ£ŗĢīæÕĢā
£Ø6·Ö£©ÓŠa”¢bĮ½øöµČĢå»żĒŅ¼«Ņ×µ¼ČȵÄĆܱÕČŻĘ÷£¬a±£³ÖČŻ»ż²»±ä£¬bÖŠµÄ»īČūæÉÉĻĻĀŅĘ¶Æ£¬ŅŌ±£³ÖÄŚĶāŃ¹ĒæĻąµČ”£ŌŚĻąĶ¬Ģõ¼žĻĀ½«3mol A£¬1mol B·Ö±šĶ¬Ź±»ģŗĻÓŚa”¢bĮ½ČŻĘ÷ÖŠ£¬·¢Éś·“Ó¦£ŗ3A(g)£«B(g) 2C(g)£«D(g)
2C(g)£«D(g)
£Ø1£©“ļµ½Ę½ŗāŹ±£¬aÖŠAµÄÅضČĪŖM mol/L”£AµÄ×Ŗ»ÆĀŹĪŖN£¬bÖŠAµÄÅضČĪŖm mol/L£¬AµÄ×Ŗ»ÆĀŹĪŖn£¬ŌņM____m£»N_____n£ØĢī£¾”¢£¼”¢£½”¢ĪŽ·Ø±Č½Ļ£©
£Ø2£©±£³ÖĪĀ¶Č²»±ä£¬°“ĻĀĮŠÅä±Č·Ö±š³äČėa”¢bĮ½ČŻĘ÷£¬“ļĘ½ŗāŗó£¬aÖŠAµÄÅضČĪŖM mol”¤L£1µÄŹĒ
A 6mol A£«2mol B B 3mol A£«2mol C C 2mol C£«1mol B£«1mol D
D 2mol C£«1mol D E 1.5mol C£«0.5mol B£«1mol C£«0.5mol D
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2013½ģ°²»ÕŹ”°ö²ŗŹŠø߶žµŚ¶žŃ§ĘŚĘŚÖŠĮŖæ¼ŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
£Ø6·Ö£©ÓŠa”¢bĮ½øöµČĢå»żĒŅ¼«Ņ×µ¼ČȵÄĆܱÕČŻĘ÷£¬a±£³ÖČŻ»ż²»±ä£¬bÖŠµÄ»īČūæÉÉĻĻĀŅĘ¶Æ£¬ŅŌ±£³ÖÄŚĶāŃ¹ĒæĻąµČ”£ŌŚĻąĶ¬Ģõ¼žĻĀ½«3mol
A£¬1mol B·Ö±šĶ¬Ź±»ģŗĻÓŚa”¢bĮ½ČŻĘ÷ÖŠ£¬·¢Éś·“Ó¦£ŗ3A(g)£«B(g)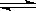 2C(g)£«D(g)
2C(g)£«D(g)
£Ø1£©“ļµ½Ę½ŗāŹ±£¬aÖŠAµÄÅضČĪŖM mol/L”£AµÄ×Ŗ»ÆĀŹĪŖN£¬bÖŠAµÄÅضČĪŖm mol/L£¬AµÄ×Ŗ»ÆĀŹĪŖn£¬ŌņM____m£»N_____n£ØĢī£¾”¢£¼”¢£½”¢ĪŽ·Ø±Č½Ļ£©
£Ø2£©±£³ÖĪĀ¶Č²»±ä£¬°“ĻĀĮŠÅä±Č·Ö±š³äČėa”¢bĮ½ČŻĘ÷£¬“ļĘ½ŗāŗó£¬aÖŠAµÄÅضČĪŖM mol”¤L£1µÄŹĒ
A 6mol A£«2mol B B 3mol A£«2mol C C 2mol C£«1mol B£«1mol D
D 2mol C£«1mol D E 1.5mol C£«0.5mol B£«1mol C£«0.5mol D
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ°²»ÕŹ”ĘŚÖŠĢā ĢāŠĶ£ŗĢīæÕĢā
 2C(g)£«D(g)
2C(g)£«D(g) ²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com