
سأNa2CO3¹ججهإنضئ1mol/LµؤNa2CO3بـز؛100mL،£
£¨1£©¹©ر،سأµؤزائ÷سذ£؛¢ظ²£ء§°ô£»¢ع½؛ح·µخ¹ـ£»¢غ100mLء؟ح²£»¢ـ100mLبفء؟ئ؟£»¢ف250mLء؟ح²£»¢قجىئ½£»¢ك50mLةص±£»س¦ر،سأµؤزائ÷£¨جî±à؛إ) ،£
£¨2£©س¦³ئب،Na2CO3µؤضتء؟خھ
(3)سذبئ؟±êا©حرآنµؤخقة«ح¸أ÷بـز؛£¬·ض±ًتاد،رخثل،¢د،ءٍثل؛حد،دُثل،£اëؤم°ïضْتµرéہدت¦ةè¼ئء½ضض·½·¨¼ّ±ًثüأا£¬²¢جùةد±êا©،£
| | ²ظ ×÷ ²½ ضè | دض دَ ¼° ½ل آغ |
| ·½·¨ز» | | |
| ·½·¨¶ | | |
£¨1£©¢ظ¢ع¢ـ¢ق¢ك
£¨2£©10.6g
(3)£¨؛دہيµؤ´ً°¸¾ù؟ة£¬µ«¶¼±طذëسذ²ظ×÷،¢دضدَ،¢½لآغµؤذًتِ) ²ظ ×÷ ²½ ضè دض دَ ¼° ½ل آغ ·½·¨ز» £¨1£©¸÷ب،ةظء؟بـز؛سعتش¹ـضذ£¬·ض±ًµخ¼سBaCl2بـز؛
£¨2£©¸÷ب،ت£سàµؤء½ضضبـز؛سعتش¹ـضذ£¬·ض±ًµخ¼سAgNO3بـز؛²ْةْ°×ة«³ءµيµؤتاد،ءٍثل
²ْةْ°×ة«³ءµيµؤتاد،رخثل£¬ت£سàµؤتاد،دُثل·½·¨¶ £¨1£©¸÷ب،ةظء؟بـز؛سعتش¹ـضذ£¬·ض±ً¼سبëحئ¬
£¨2£©¸÷ب،ت£سàµؤء½ضضبـز؛سعتش¹ـضذ£¬·ض±ًµخ¼سBaCl2بـز؛سذئّإف²ْةْµؤتاد،دُثل
²ْةْ°×ة«³ءµيµؤتاد،ءٍثل£¬ت£سàµؤتاد،رخثل
½âخِتشجâ·ضخِ£؛£¨1£©¹ججهإنضئبـز؛ذèزھجىئ½،¢ةص±،¢²£ء§°ô،¢100mlبفء؟ئ؟،¢½؛ح·µخ¹ـ£¬ثùزشر،¢ظ¢ع¢ـ¢ق¢ك
£¨2£©س¦³ئب،Na2CO3µؤضتء؟خھ0.1L،ء1mol/L،ء106g/mol=10.6g
£¨3£©¼ّ±ًرخثل،¢د،ءٍثل،¢د،دُثل£¬؟ةسأآب»¯±µ،¢دُثلزّبـز؛£¬»ٍحسëآب»¯±µبـز؛£¬»ٍحسëدُثلزّبـز؛£¬¾كجه²½ضèبçدآ ²ظ ×÷ ²½ ضè دض دَ ¼° ½ل آغ ·½·¨ز» £¨1£©¸÷ب،ةظء؟بـز؛سعتش¹ـضذ£¬·ض±ًµخ¼سBaCl2بـز؛
£¨2£©¸÷ب،ت£سàµؤء½ضضبـز؛سعتش¹ـضذ£¬·ض±ًµخ¼سAgNO3بـز؛²ْةْ°×ة«³ءµيµؤتاد،ءٍثل
²ْةْ°×ة«³ءµيµؤتاد،رخثل£¬ت£سàµؤتاد،دُثل·½·¨¶ £¨1£©¸÷ب،ةظء؟بـز؛سعتش¹ـضذ£¬·ض±ً¼سبëحئ¬
£¨2£©¸÷ب،ت£سàµؤء½ضضبـز؛سعتش¹ـضذ£¬·ض±ًµخ¼سBaCl2بـز؛سذئّإف²ْةْµؤتاد،دُثل
²ْةْ°×ة«³ءµيµؤتاد،ءٍثل£¬ت£سàµؤتاد،رخثل
؟¼µم£؛؟¼²éبـز؛µؤإنضئ£¬د،رخثل،¢د،ءٍثل،¢د،دُثلµؤ¼ّ±ً


 جىجىء·؟عثمدµءذ´ً°¸
جىجىء·؟عثمدµءذ´ً°¸
| ؤ꼶 | ¸كضذ؟خ³ج | ؤ꼶 | ³ُضذ؟خ³ج |
| ¸كز» | ¸كز»أâ·ر؟خ³جحئ¼ِ£، | ³ُز» | ³ُز»أâ·ر؟خ³جحئ¼ِ£، |
| ¸ك¶ | ¸ك¶أâ·ر؟خ³جحئ¼ِ£، | ³ُ¶ | ³ُ¶أâ·ر؟خ³جحئ¼ِ£، |
| ¸كب | ¸كبأâ·ر؟خ³جحئ¼ِ£، | ³ُب | ³ُبأâ·ر؟خ³جحئ¼ِ£، |
؟ئؤ؟£؛¸كضذ»¯ر§ ہ´ش´£؛ جâذح£؛µ¥ر،جâ
ءٍ´ْءٍثلؤئ£¨Na2S2O3£©؟ةسةراءٍثلؤئ؛حءٍ·غح¨¹»¯؛د·´س¦ضئµأ£؛Na2SO3 + S  Na2S2O3£¬³£خآدآبـز؛ضذخِ³ِ¾§جهخھNa2S2O3?5H2O،£Na2S2O3،¤5H2Oسع40،«45،وبغ»¯£¬48،و·ض½â£»Na2S2O3ز×بـسعث®£¬²»بـسعزز´¼،£شعث®ضذسذ¹طخïضتµؤبـ½â¶باْدكبçسزح¼ثùت¾،£
Na2S2O3£¬³£خآدآبـز؛ضذخِ³ِ¾§جهخھNa2S2O3?5H2O،£Na2S2O3،¤5H2Oسع40،«45،وبغ»¯£¬48،و·ض½â£»Na2S2O3ز×بـسعث®£¬²»بـسعزز´¼،£شعث®ضذسذ¹طخïضتµؤبـ½â¶باْدكبçسزح¼ثùت¾،£
¢ٌ£®دض°´بçدآ·½·¨ضئ±¸Na2S2O3،¤5H2O£؛
½«ءٍ»¯ؤئ؛حج¼ثلؤئ°´·´س¦زھاَ±بہز»²¢·إبëب¾±ةصئ؟ضذ£¬×¢بë150mLصôءَث®ت¹ئنبـ½â£¬شع·ضز؛آ©¶·ضذ£¬×¢بëإ¨رخثل£¬شع×°ضأ2ضذ¼سبëراءٍثلؤئ¹ججه£¬²¢°´دآح¼°²×°؛أ×°ضأ،£
£¨1£©زائ÷2µؤأû³ئخھ £¬
×°ضأ6ضذ؟ة·إبë ،£
| A£®BaCl2بـز؛ | B£®إ¨H2SO4 | C£®ثلذشKMnO4بـز؛ | D£®NaOHبـز؛ |
 Na2S2O3
Na2S2O3
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛¸كضذ»¯ر§ ہ´ش´£؛ جâذح£؛µ¥ر،جâ
دآءذسذ¹ط»¯ر§تµرéµؤأèتِضذصب·µؤتا
| A£®·ضہëزز´¼؛حززثلزھسأ·ضز؛µؤ·½·¨ |
| B£®إنضئز»¶¨خïضتµؤء؟إ¨¶بµؤNaClبـز؛£¬بç¹ûNaCl¹ججهضذ؛¬سذNa2CO3,ثùإنضئبـز؛ضذc(Na+)½«ئ«¸ك |
| C£®ؤ³خقة«بـز؛ضذ¼سد،ءٍثل²ْةْخقة«خقخ¶ئّجه£¬¸أئّجهؤـت¹³خاهت¯»زث®±ن»ë×ا£¬ثµأ÷¸أبـز؛ضذ؛¬CO32- |
D£®ئ½؛â2NO2 N2O4(خقة«)£¬¼ُر¹ت±£¬ئّجهرصة«±نا³£¬ئ½؛âدٍص·´س¦·½دٍزئ¶¯ N2O4(خقة«)£¬¼ُر¹ت±£¬ئّجهرصة«±نا³£¬ئ½؛âدٍص·´س¦·½دٍزئ¶¯ |
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛¸كضذ»¯ر§ ہ´ش´£؛ جâذح£؛µ¥ر،جâ
دآءذتµرé²ظ×÷»ٍتآتµسëش¤ئعتµرéؤ؟µؤ»ٍثùµأ½لآغز»ضآµؤتا( )
| ر،دî | تµرé²ظ×÷»ٍتآتµ | تµرéؤ؟µؤ»ٍ½لآغ |
| A | ؤ³بـز؛ أ°³ِئّإف أ°³ِئّإف | ثµأ÷شبـز؛ضذز»¶¨؛¬سذCO32- |
| B | SiO2سë´؟¼î¸كخآ؟ةةْ³ةCO2 | ثµأ÷¹èثلµؤثلذش±بج¼ثلا؟ |
| C | µâ»¯¼طبـز؛¾أضأدش»ئة« | تاسةسعI-±»»¹ش£¬خِ³ِµؤI2بـسعبـز؛ضذ |
| D | جْ¶¤·إشعإ¨دُثلضذ½إف؛َ£¬شظسأصôءَث®³هد´£¬ب»؛َ·إبëCuSO4بـز؛ضذ²»·´س¦ | ثµأ÷جْ¶¤±يأوذخ³ةءثز»²مضآأـخب¶¨µؤرُ»¯ؤ¤ |
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛¸كضذ»¯ر§ ہ´ش´£؛ جâذح£؛µ¥ر،جâ
دآءذثµ·¨ضذصب·µؤتا£¨ £©
| A£®³£خآدآ£¬إ¨ءٍثل،¢إ¨دُثل¾ùؤـ½«½ًتôحرُ»¯£¬از¾ùؤـت¹جْ¶غ»¯ |
| B£®ززد©³£سأ×÷ض²خï´كتى¼ء£¬ززثل³£سأ×÷µ÷خ¶¼ء£¬زز´¼³£سأسعة±¾ْدû¶¾ |
| C£®آêè§،¢ث®¾§،¢×êت¯µؤض÷زھ³ة·ض¾ùخھSiO2 |
| D£®سأCCI4فحب،µâث®ضذµؤI2ت±£¬×دة«بـز؛´س·ضز؛آ©¶·µؤةد؟عµ¹³ِ |
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛¸كضذ»¯ر§ ہ´ش´£؛ جâذح£؛جî؟صجâ
£¨6·ض£©دآح¼ضذA،¢B،¢C،¢Dتا³£¼ûµؤئصح¨²£ء§تش¼ءئ؟،£اë°ردآءذتش¼ءµؤذٍ؛إجîذ´شعتت؛دت¢·إµؤتش¼ءئ؟دآأوµؤہ¨؛إؤع£؛
a£®إ¨ءٍثل b£®ج¼ثلؤئبـز؛ c£®آءئ¬ d£®إ¨دُثل e£®اâرُ»¯ؤئبـز؛ f£®¶رُ»¯أج
| A | B | C | D |
 |  |  |  |
| £¨ £© | £¨ £© | £¨ £© | £¨ £© |
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛¸كضذ»¯ر§ ہ´ش´£؛ جâذح£؛جî؟صجâ
خھءث½«»ىسذK2SO4،¢MgSO4µؤKNO3¹ججهجل´؟£¬²¢ضئµأ´؟¾»µؤKNO3بـز؛£¬ؤ³ر§ةْةè¼ئبçدآتµرé·½°¸£؛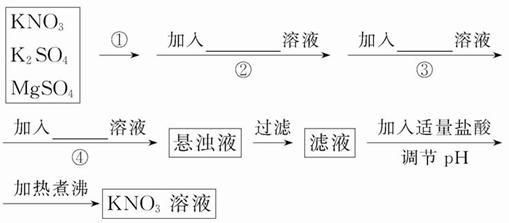
(1)²ظ×÷¢ظخھ___________________________،£
(2)²ظ×÷¢ع~¢ـ¼سبëµؤتش¼ءزہ´خ؟ةزشخھ£؛___________________________________________________،£
(3)بç؛خإذ¶دSO42-زر³¾،£؟____________________________،£
(4)تµرé¹³جضذ²ْةْµؤ¶à´خ³ءµي_______ (جî،°ذèزھ،±»ٍ،°²»ذèزھ،±)¶à´خ¹آث£¬ئنہيسةتا_______________________________________________________،£
(5)¸أح¬ر§µؤتµرéةè¼ئ·½°¸تا·ٌردأـ£؟اëثµأ÷ہيسة£؛___________________________________،£
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛¸كضذ»¯ر§ ہ´ش´£؛ جâذح£؛جî؟صجâ
رُ»¯ذ؟·غؤ©¹م·؛س¦سأسعدً½؛،¢ح؟ءد،¢جص´ة،¢»¯¹¤،¢ز½ز©،¢²£ء§µبذذزµ£¬خھ×غ؛دس¦سأ×تش´£¬³£´سز±ء¶ذ؟،¢ذ؟ضئئ·¼س¹¤ئَزµ»طتصµؤذ؟شü£¨؛¬سذZnO،¢FeO،¢Fe2O3،¢CuO،¢Al2O3µبشسضت£©ہ´ضئب،رُ»¯ذ؟£¬ء÷³جبçح¼£؛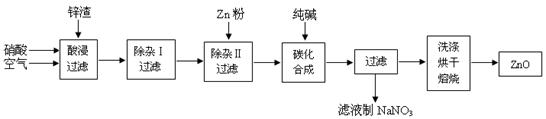
سذ¹طاâرُ»¯خï³ءµيحêب«µؤpH¼ûدآ±ي£؛
£¨1£©شعثل½¹³جضذ£¬³£½«ذ؟شüثé³ة·غؤ©²¢ح¨بë؟صئّ£¬ح¬ت±±£³ضثل¹ء؟£¬ہيسةتا ،£
£¨2£©ةدتِ¹¤زص¶à´¦ةو¼°،°¹آث،±£¬تµرéتزضذ¹آثذèزھت¹سأµؤ²£ء§زائ÷سذةص±،¢ ،£
£¨3£©¢ظشع،°³شس¢ٌ،±ضذ£¬³£سأثلذشKMnO4بـز؛£¬ئنؤ؟µؤتا £¬KMnO4تا¸أ·´س¦µؤ £¨رُ»¯¼ء،¢»¹ش¼ء£©،£½«بـز؛µؤpHµ÷ضء4£¬ؤ؟µؤتا ،£
¢عشع،°³شسII،±ضذ£¬¼سبëذ؟·غµؤؤ؟µؤتا ،£
£¨4£©شع،°ج¼»¯؛د³ة،±ضذ£¬²ْخïضذسذ¼îت½ج¼ثلذ؟£غZn2(OH)2CO3£ف؛حCO2µب£¬اëذ´³ِ¸أ·´س¦µؤ»¯ر§·½³جت½ ،£
£¨5£©±¾¹¤زصضذ£¬¹آث·ضہë´خت½د¶à£¬ئنأ÷دشµؤب±دفتا ،£
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛¸كضذ»¯ر§ ہ´ش´£؛ جâذح£؛تµرéجâ
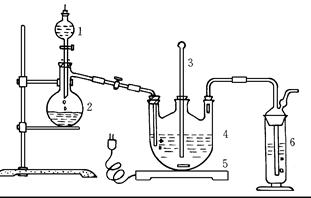
£¨15·ض£©S2Cl2³£سأسعدً½؛ءٍ»¯،¢سذ»ْخïآب»¯µؤتش¼ء£¬تµرéتز؟ةہûسأبçدآ×°ضأہ´ضئ±¸S2Cl2،££¨²؟·ض¼ذ³ضزائ÷زرآشب¥£©
زرضھ£؛¢ظضئ±¸µؤ·´س¦·½³جت½خھ£؛
¢ع·´س¦ةو¼°µؤ¼¸ضضخïضتذشضتبçدآ£؛
| ذشضت خïضت | بغµم | ·ذµم | ×إ»ًµم | ئنثûذشضت |
| µ¥ذ±ءٍ | 119.2،و | 446.6،و | 363،و | / |
| ذ±·½ءٍ | 112.8،و | 446.6،و | 363،و | / |
| S2Cl2 | -77،و | 137،و | / | سِث®ا؟ءز·ض½âةْ³ةS،¢SO2؛حHCl |
²é؟´´ً°¸؛ح½âخِ>>
°ظ¶بضآذإ - ء·د°²لءذ±ي - تشجâءذ±ي
؛±±ت،»¥ءھحّخ¥·¨؛ح²»ء¼ذإد¢¾ظ±¨ئ½ج¨ | حّةدسذ؛¦ذإد¢¾ظ±¨×¨اّ | µçذإص©ئ¾ظ±¨×¨اّ | ةوہْت·ذéخقض÷زهسذ؛¦ذإد¢¾ظ±¨×¨اّ | ةوئَاضب¨¾ظ±¨×¨اّ
خ¥·¨؛ح²»ء¼ذإد¢¾ظ±¨µç»°£؛027-86699610 ¾ظ±¨ستدن£؛58377363@163.com