
½ńÓŠ¢ŁCH3COOH ¢ŚHCl ¢ŪH2SO4 ČżÖÖČÜŅŗ”£øł¾ŻŅŖĒó»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©µ±ĖüĆĒpHĻąĶ¬Ź±£¬ĘäĪļÖŹµÄĮæÅضČ×ī“óµÄŹĒ________________£ØĢīŠņŗÅ£©”£
£Ø2£©µ±ĖüĆĒµÄĪļÖŹµÄĮæÅضČĻąĶ¬Ź±£¬ĘäpH×īŠ”µÄŹĒ____________£ØĢīŠņŗÅ£©”£
£Ø3£©½«c(H£«)ĻąĶ¬µÄČżÖÖĖį¾ł¼ÓĖ®Ļ”ŹĶÖĮŌĄ“µÄ10±¶£¬c(H£«)Óɓ󵽊”µÄĖ³ŠņĪŖ_________________”££ØĢīŠņŗÅ£©
£Ø4£©Ģå»żŗĶĪļÖŹµÄĮæÅØ¶Č¾łĻąĶ¬µÄ¢Ł¢Ś¢ŪČżÖÖĖįČÜŅŗ£¬·Ö±šÓėĻąĶ¬ÅØ¶ČµÄÉÕ¼īČÜŅŗĒ”ŗĆĶźČ«·“Ó¦£¬ĖłŠčÉÕ¼īµÄĢå»ż±ČĪŖ_____________________£»“ĖŹ±ĖłµĆµ½µÄČżÖÖČÜŅŗµÄpH“󊔹ŲĻµĪŖ______________________£ØĢīŠņŗÅ£©”£
 ¶į¹Ś½š¾ķČ«ÄÜĮ·æ¼ĻµĮŠ“š°ø
¶į¹Ś½š¾ķČ«ÄÜĮ·æ¼ĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2015-2016ѧğø£½ØŹ”øßŅ»ÉĻĘŚÖŠ²āŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ĻĀĮŠŹµŃé²Ł×÷¾łŅŖÓĆ²£Į§°ō£¬ĘäÖŠ²£Į§°ō×÷ÓĆĻąĶ¬µÄŹĒ
¢Ł¹żĀĖ ¢ŚÕō·¢ ¢ŪČܽā ¢ÜĻņČŻĮæĘæ×ŖŅĘŅŗĢå
A£®¢ŁŗĶ¢Ś B£®¢ŁŗĶ¢Ū C£®¢ŪŗĶ¢Ü D£®¢ŁŗĶ¢Ü
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2015-2016ѧğøŹĖąĢģĖ®Ņ»ÖŠøßŅ»ÉĻµŚ¶žŃ§¶Ī¶ĪÖŠ²ā»Æѧ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
ÓŠŅ»ĶøĆ÷ČÜŅŗ£¬ŅŃÖŖĘäÖŠæÉÄÜŗ¬ÓŠFe3£«”¢Mg2£«”¢Cu2£«”¢Al3£«”¢NH4£«£¬¼ÓČėŅ»ÖÖµ»ĘÉ«·ŪÄ©¹ĢĢåŹ±£¬¼ÓČČÓŠ“Ģ¼¤ŠŌĘųĪ¶µÄ»ģŗĻĘųĢå·Å³ö£¬Ķ¬Ź±Éś³É°×É«³Įµķ”£µ±¼ÓČė0.4 molµ»ĘÉ«·ŪÄ©Ź±£¬²śÉśĘųĢå0.3 mol£¬¼ĢŠų¼ÓČėµ»ĘÉ«·ŪÄ©Ź±£¬²śÉśĪŽ“Ģ¼¤ŠŌĘųĪ¶µÄĘųĢ壬ĒŅ¼ÓČėµ»ĘÉ«·ŪÄ©Ź±²śÉś°×É«³ĮµķµÄĮæČēĻĀĶ¼ĖłŹ¾”£(ŅŃÖŖ£ŗNH4£«£«OH£ NH3”ü£«H2O£©
NH3”ü£«H2O£©
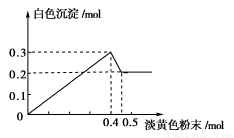
øł¾ŻĢāŅā»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©µ»ĘÉ«·ŪÄ©ĪŖ____________(ĢīĆū³Ę)”£
£Ø2£©ČÜŅŗÖŠæĻ¶ØÓŠ______________Ąė×Ó£¬æĻ¶Øƻӊ__________Ąė×Ó”£
£Ø3£©ČÜŅŗÖŠĄė×ÓµÄĪļÖŹµÄĮæÖ®±ČĪŖ______________________________”£
£Ø4£©Š“³ö³Įµķ²æ·Ö¼õÉŁŹ±µÄĄė×Ó·½³ĢŹ½£ŗ__________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2015-2016ѧğøŹĖąĢģĖ®Ņ»ÖŠøßŅ»ÉĻµŚ¶žŃ§¶Ī¶ĪÖŠ²ā»Æѧ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
“Ó½šŹōĄūÓƵĥśŹ·Ą“æ“£¬ĻČŹĒĒąĶĘ÷Ź±“ś£¬¶ųŗóŹĒĢśĘ÷Ź±“ś£¬ĀĮµÄĄūÓĆŹĒ½ü°ŁÄźµÄŹĀ”£ÕāøöĻČŗóĖ³ŠņøśĻĀĮŠÓŠ¹ŲµÄŹĒ£ŗ¢ŁµŲæĒÖŠµÄ½šŹōŌŖĖŲµÄŗ¬Į棻¢Ś½šŹō»ī¶ÆŠŌĖ³Šņ£»¢Ū½šŹōµÄµ¼µēŠŌ£»¢Ü½šŹōŅ±Į¶µÄÄŃŅ׳Ģ¶Č£»¢Ż½šŹōµÄŃÓÕ¹ŠŌ£»
A£®¢Ł¢Ū B£®¢Ś¢Ż C£®¢Ū¢Ż D£®¢Ś¢Ü
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2015-2016ѧğ¹ćĪ÷Ź”øßŅ»ÉĻĘŚÖŠ¶Īæ¼»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ĻĀĮŠ·“Ó¦µÄĄė×Ó·½³ĢŹ½ŹéŠ“ÕżČ·µÄŹĒ
A£®Ģ¼ĖįĒāÄĘČÜŅŗÓėĒāŃõ»ÆÄĘČÜŅŗ»ģŗĻ£ŗHCO3££«OH£===H2O£«CO32£
B£®Ļ” H2SO4ÓėĢś·Ū·“Ó¦£ŗ2Fe+6H+=2Fe3++3H2”ü
C£®ĒāŃõ»Æ±µČÜŅŗÓėĻ” H2SO4·“Ó¦£ŗBa2++SO42£=BaSO4”ż
D£®Ģ¼ĖįøĘÓėŃĪĖį·“Ó¦£ŗCO32£+2H+=H2O+CO2”ü
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2015-2016ѧğŗ£ÄĻŹ”ø߶žÉĻĘŚÖŠ£ØĄķ£©»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ĻĀĮŠĄė×Ó·½³ĢŹ½ÖŠ£¬ŹōÓŚĖ®½ā·“Ó¦µÄŹĒ
A£®CH3COOH +H2O 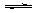 CH3COO- + H3O+ B£®SO2 + H2O
CH3COO- + H3O+ B£®SO2 + H2O 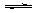 HSO3- + H+
HSO3- + H+
C£®HS- + H2O 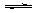 S2- + H3O+ D£®CO32- + H2O
S2- + H3O+ D£®CO32- + H2O  HCO3- + OH-
HCO3- + OH-
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2015-2016ѧğø£½ØŹ”ø߶žÉĻĘŚÖŠ²āŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
£Ø1£©ŹµŃé²āµĆ£¬8g¼×ĶéŌŚŃõĘųÖŠ³ä·ÖČ¼ÉÕÉś³É¶žŃõ»ÆĢ¼ĘųĢåŗĶŅŗĢ¬Ė®Ź±ŹĶ·Å³ö113kJµÄČČĮ棬ŹŌŠ“³ö¼×ĶéČ¼ÉÕµÄČČ»Æѧ·½³ĢŹ½_________________________
£Ø2£©ŌŚŃ¹ĒæĪŖ0.1MPaĢõ¼žĻĀ£¬½«amolCOÓė3amolH2µÄ»ģŗĻĘųĢåŌŚ“߻ƼĮ×÷ÓĆĻĀÄÜ×Ō·¢·“Ӧɜ³É¼×“¼£ŗ
CO£Øg£©+2H2£Øg£© CH30H£Øg£©
CH30H£Øg£©
¢ŁøĆ·“Ó¦µÄ”÷H_____0£¬___0£ØĢī”°>”±”¢”°<”±»ņ”°=”±£©
¢ŚČōČŻĘ÷ČŻ»ż²»±ä£¬ĻĀĮŠ“ėŹ©æÉŌö¼Ó¼×“¼²śĀŹµÄŹĒ____________
A£®ÉżøßĪĀ¶Č
B£®½«CH3OH£Øg£©“ÓĢåĻµÖŠ·ÖĄė
C£®³äČėHe£¬Ź¹ĢåĻµ×ÜŃ¹ĒæŌö“ó
D£®ŌŁ³äČĖlmolCOŗĶ3mol H2
£Ø3£©ŅŌCH4ŗĶH2OĪŖŌĮĻ£¬ĶعżĻĀĮŠ·“Ó¦Ą“Öʱø¼×“¼”£
¢ń£ŗCH4£Øg£©+H2O£Øg£©=CO£Øg£©+3H2£Øg£©”÷H=+206.0kJ•mol-1
¢ņ£ŗCO£Øg£©+2H2£Øg£©=CH3OH£Øg£©”÷H=-129.0kJ•mol-1
CH4£Øg£©ÓėH2O£Øg£©·“Ӧɜ³ÉCH3OH£Øg£©ŗĶH2£Øg£©µÄČČ»Æѧ·½³ĢŹ½ĪŖ____________
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2015-2016ѧğø£½ØŹ”øßŅ»ÉĻĘŚÖŠ²āŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ
A£®0.5molH2ĖłÕ¼Ģå»żŌ¼ĪŖ11.2L
B£®±ź×¼×“æöĻĀ£¬6.02”Į1023øö·Ö×ÓĖłÕ¼µÄĢå»żŌ¼ĪŖ22.4L
C£®H2µÄĦ¶ūÖŹĮæĪŖ2g/mol
D£®°¢·ü¼ÓµĀĀŽ³£Źż¾ĶŹĒ6.02”Į1023£¬Ć»ÓŠµ„Ī»
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2015-2016ѧğŗ£ÄĻŹ”øßŅ»ÉĻĘŚÖŠ²āŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
½ńÓŠŹ®ÖÖĪļÖŹ£ŗ¢ŁĀĮĻß ¢ŚŹÆÄ« ¢ŪCO2 ¢ÜBaSO4¾§Ģå ¢Ż“æĮņĖį ¢Ž½šøÕŹÆ ¢ßŹÆ»ŅĖ® ¢ąŅŅ“¼ ¢įČŪ»ÆµÄKNO3 ¢āHCl£ØĢīŠņŗÅ£©
ĘäÖŠ£¬Äܵ¼µēµÄÓŠ £¬ŹōÓŚµē½āÖŹµÄÓŠ £¬ŹōÓŚ·Ēµē½āÖŹµÄÓŠ ”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com