
��֪A��B��C��D����ѧ��ѧ�г��������ֲ�ͬ���ӣ�����֮�������ͼ��ʾ��ת����ϵ(��Ӧ�����Ѿ���ȥ)��

(1)���A��B��C��D����10���ӵ����ӣ���д��A��D�ĵ���ʽ��A____________��D____________��
(2)���A��C��18���ӵ����ӣ�B��D��10���ӵ����ӣ���д����
��A��B����Һ�з�Ӧ�����ӷ���ʽΪ__________________________________________��
�ڸ����������ӷ���ʽ�������ж�C��B������ӵ�������С��________________________________________________________________________
________________________________________________________________________
(�û�ѧʽ�����ӷ��ű�ʾ)��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ͼ��ij��������Ķ��۷��ӣ��÷�����A��B����Ԫ�ض��ǵ�3���ڵ�Ԫ�أ�����������ԭ�ӵ��������������ﵽ8�����ӵ��ȶ��ṹ������˵������ȷ����(����)

A���û�����Ļ�ѧʽ��Al2Cl6 B���û����������ӻ����������״̬���ܵ���
C���û������ڹ�̬ʱ���γɵľ����Ƿ��Ӿ��� D���û������в��������Ӽ���Ҳ�����зǼ��Թ��ۼ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����������һ����Ҫ�Ļ�����Ʒ��ij��ȤС�����Ʊ���������ƾ���(Na2S2O3·5H2O)��
��.[��������]
(1)Na2S2O3·5H2O����ɫ�����壬������ˮ����ϡ��Һ��BaCl2��Һ����������ɡ�
(2)��Na2CO3��Na2S�����Һ��ͨ��SO2���Ƶ�Na2S2O3�����ò�Ʒ����������Na2SO3��Na2SO4��
(3)Na2SO3�ױ�������BaSO3������ˮ��������ϡHCl��
��.[�Ʊ���Ʒ]
ʵ��װ����ͼ��ʾ(ʡ�Լг�װ��)��
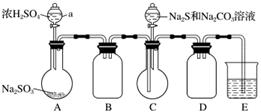
ʵ�鲽�裺
(1)���װ�������ԣ���ͼʾ�����Լ���
����a��������________��E�е��Լ���________(ѡ��������ĸ���)��
A��ϡH2SO4
B��NaOH��Һ
C������NaHSO3��Һ
(2)����C����ƿ����Na2S��Na2CO3�����Һ������A����ƿ�μ�ŨH2SO4��
(3)��Na2S��Na2CO3��ȫ���ĺ�����Ӧ������C�л�����Һ��__________(��д��������)���ᾧ�����ˡ�ϴ�ӡ�����õ���Ʒ��
��.[̽���뷴˼]
(1)Ϊ��֤��Ʒ�к���Na2SO3��Na2SO4����С�����������ʵ�鷽�����뽫��������������
(�����Լ���ϡHNO3��ϡH2SO4��ϡHCl������ˮ��ѡ��)
ȡ������Ʒ���ϡ��Һ���μ�����BaCl2��Һ���а�ɫ�������ɣ�________________��������δ��ȫ�ܽ⣬���д̼�����ζ��������������ȷ����Ʒ�к���Na2SO3��Na2SO4��
(2)Ϊ����װ��C������Na2SO4�������ڲ��ı�ԭ��װ�õĻ����϶�ʵ�鲽��(2)�����˸Ľ����Ľ���IJ�����
________________________________________________________________________��
(3)Na2S2O3·5H2O���ܽ�����¶����������������ò�Ʒͨ��________________�����ᴿ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ijԪ�ص�һ��ͬλ��X��ԭ��������ΪA����N�����ӣ�����1Hԭ�����HmX���ӣ���a g HmX���������ӵ����ʵ����� (����)
A.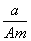 (A��N��m)mol B.
(A��N��m)mol B.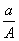 (A��N)mol
(A��N)mol
C.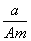 (A��N)mol D.
(A��N)mol D.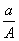 (A��N��m)mol
(A��N��m)mol
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�ڵ�n���Ӳ��У�������Ϊԭ�ӵ������ʱ�����ɵ����������(n��1)����ͬ��������Ϊԭ�ӵĴ����ʱ�����������(n��1)���10������˵��Ӳ���(����)
A��K�� B��L�� C��M�� D��N��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ˮ(D2O)����Ҫ�ĺ˹�ҵԭ�ϣ�����˵���������(����)
A���(D)ԭ�Ӻ�����1������
B��1H��D����ͬλ��
C��H2O��D2O����ͬ��������
D��1H O��D
O��D O����Է���������ͬ
O����Է���������ͬ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ԭ������ͬ������������ͬ�����ӣ�����Ϊ�ȵ����塣��Ԫ�ؿ��γ������ȵ����������ӣ�BO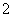 ��BC
��BC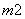 ��BN
��BN ����m��nֵΪ(����)
����m��nֵΪ(����)
A��5��3 B��2��4 C��3��1 D��1��2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����ʵ������д������(����)
A����Һʱ����Һ©���²�Һ����¿ڷų����ϲ�Һ����Ͽڵ���
B���������ʱ��Ӧʹ�¶ȼ�ˮ����������ƿ��֧�ܿڴ�
C�������У���ȴˮӦ�������ܵ��Ͽ�ͨ�룬�¿�����
D����������ʱ��Ӧʹ������е�ˮ����ȫ���ɺ���ֹͣ����
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com