
��1����֪298Kʱ��Fe(OH)3���ܶȻ�����![]() =2.6��10-39�� Mg(OH)2���ܶȻ�����
=2.6��10-39�� Mg(OH)2���ܶȻ�����![]() =5.6��
=5.6��![]() ��ȡ����Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��FeCl3���Һ����HCl��������һ������MgCO3�ﵽ������Һƽ�⣬���pH=4.00������¶��²�������Һ�е�c(Fe3+)=______________��
��ȡ����Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��FeCl3���Һ����HCl��������һ������MgCO3�ﵽ������Һƽ�⣬���pH=4.00������¶��²�������Һ�е�c(Fe3+)=______________��
��û��Mg(OH)2��������______________��С����ޡ�����������________________________________��
��2����HnA���B(OH)m��ȫ��Ӧ��������.
����HnAΪHCl���Ҹ�����Һ��pH��7�������ӷ���ʽ˵��ԭ��
������0.4mol��L��1��NaOH��Һ��0.2mol��L��1��HnA��Һ�������Ϻ�pH=10��
��HnAΪ ������ţ�.
a.һԪǿ�� b. һԪ���� c. ��Ԫǿ�� d. ��Ԫ����
��3��ijѧУ������ȤС��Ӻ�ˮɹ�κ����±(��Ҫ��Na+��Mg2+��Cl����Br����)��ģ�ҵ��������ȡþ����Ҫ�������£��ش��������⣺
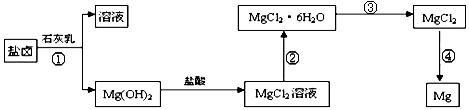
��.�ӹ��̢ٵõ���Mg(OH)2�����л���������Ca(OH)2 ����ȥ����Ca(OH)2�ķ������Ƚ��������뵽ʢ�� ��Һ���ձ��У���ֽ�����ˡ�ϴ�ӿɵô�����Mg(OH)2��
��.���̢۵�ת����Ҫ��HCl�����м��ȣ�HCl��������
��.д�����̢��з�����Ӧ�Ļ�ѧ����ʽ
 �����ߴ���ϵ�д�
�����ߴ���ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�꽭��ʡ�ϲ��еڶ���ѧ�߶���һ���¿���ѧ�Ծ����������� ���ͣ�������
�����10�֣����ס�������������ͬ���칹�壬�ڿ�����ȼ�յõ����������������ʱ����P4O6����������������P4O10��
��1����֪298Kʱ���ס�������ȫȼ�յ��Ȼ�ѧ����ʽ�ֱ�Ϊ��
P4(s,����)+5O2(g)=P4O10(s) ��H1=" -2983.2" kJ?mol-1,
P(s������)+ 5/4O2(g)="1/4" P4O10(s) ��H2=" -738.5" kJ?mol-1
����¶��°���ת��Ϊ�����Ȼ�ѧ����ʽΪ ��
��2����֪298Kʱ���ײ���ȫȼ�յ��Ȼ�ѧ����ʽΪP4(s,����)+3O2(g)=P4O6(s) ��H= -1638kJ?mol-1����ij�ܱ������м���62g����50.4L��������״���£�����������ʹ֮ǡ����ȫ��Ӧ�������õ���P4O10��P4O6�����ʵ���֮��Ϊ ����Ӧ�����зų�������ΪΪ ��
��3����֪����PCl3�ķ��ӽṹ��ͼ��ʾ�����ṩ���µĻ�ѧ���ļ��ܣ�KJ��mol��:P-P 198��Cl-Cl 243��P-Cl 331��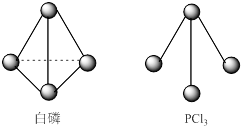
��ӦP4(s,����)+6Cl2(g)=4PCl3(s)�ķ�Ӧ�Ȧ�H = ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014�콭��ʡ�ϲ��и߶���һ���¿���ѧ�Ծ��������棩 ���ͣ�������
�����10�֣����ס�������������ͬ���칹�壬�ڿ�����ȼ�յõ����������������ʱ����P4O6����������������P4O10��
��1����֪298Kʱ���ס�������ȫȼ�յ��Ȼ�ѧ����ʽ�ֱ�Ϊ��
P4(s,����)+5O2(g)=P4O10(s) ��H1= -2983.2 kJ•mol-1,
P(s������)+ 5/4O2(g)=1/4 P4O10(s) ��H2= -738.5 kJ•mol-1
����¶��°���ת��Ϊ�����Ȼ�ѧ����ʽΪ ��
��2����֪298Kʱ���ײ���ȫȼ�յ��Ȼ�ѧ����ʽΪP4(s,����)+3O2(g)=P4O6(s) ��H= -1638kJ•mol-1����ij�ܱ������м���62g����50.4L��������״���£�����������ʹ֮ǡ����ȫ��Ӧ�������õ���P4O10��P4O6�����ʵ���֮��Ϊ ����Ӧ�����зų�������ΪΪ ��
��3����֪����PCl3�ķ��ӽṹ��ͼ��ʾ�����ṩ���µĻ�ѧ���ļ��ܣ�KJ��mol��:P-P 198��Cl-Cl 243��P-Cl 331��

��ӦP4(s,����)+6Cl2(g)=4PCl3(s)�ķ�Ӧ�Ȧ�H = ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010-2011�긣����Ϫһ�С��ݰ�һ�С�������ѧ�߶���ѧ����ĩ������ѧ�� ���ͣ������
��16�֣�
��1����֪298Kʱ��Fe(OH)3���ܶȻ����� =2.6��10-39�� Mg(OH)2���ܶȻ�����
=2.6��10-39�� Mg(OH)2���ܶȻ����� =5.6��
=5.6�� ��ȡ����Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��FeCl3���Һ����HCl��������һ������MgCO3�ﵽ������Һƽ�⣬���pH=4.00������¶��²�������Һ�е�c(Fe3+)=______________��
��ȡ����Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��FeCl3���Һ����HCl��������һ������MgCO3�ﵽ������Һƽ�⣬���pH=4.00������¶��²�������Һ�е�c(Fe3+)=______________��
��û��Mg(OH)2��������______________��С����ޡ�����������________________________________��
��2����HnA���B(OH)m��ȫ��Ӧ��������.
����HnAΪHCl���Ҹ�����Һ��pH��7�������ӷ���ʽ˵��ԭ��
������0.4mol��L��1��NaOH��Һ��0.2mol��L��1��HnA��Һ�������Ϻ�pH=10��
��HnAΪ ������ţ�.
a.һԪǿ�� b. һԪ���� c. ��Ԫǿ�� d. ��Ԫ����
��3��ijѧУ������ȤС��Ӻ�ˮɹ�κ����±(��Ҫ��Na+��Mg2+��Cl����Br����)��ģ�ҵ��������ȡþ����Ҫ�������£��ش��������⣺
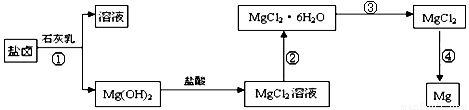
��.�ӹ��̢ٵõ���Mg(OH)2�����л���������Ca(OH)2 ����ȥ����Ca(OH)2�ķ������Ƚ��������뵽ʢ�� ��Һ���ձ��У���ֽ�����ˡ�ϴ�ӿɵô�����Mg(OH)2��
��.���̢۵�ת����Ҫ��HCl�����м��ȣ�HCl��������
��.д�����̢��з�����Ӧ�Ļ�ѧ����ʽ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
I.����ͼ��ʾ����2molA�����1molB�������һ�ݻ��ɱ���ܱ������С�
������Ӧ��2A(g)+B(g)![]() 2C(g)����Ӧ��ʼʱ�ɻ����Ļ�����λ����ͼ1��ʾ������Ӧ�ﵽƽ��ʱ������λ����ͼ2��ʾ.��ﵽƽ��ʱ��A��ת����Ϊ________________;
2C(g)����Ӧ��ʼʱ�ɻ����Ļ�����λ����ͼ1��ʾ������Ӧ�ﵽƽ��ʱ������λ����ͼ2��ʾ.��ﵽƽ��ʱ��A��ת����Ϊ________________;
�������·�Ӧ��ƽ�ⳣ��Ϊ_______________________________��
|
|

|
|
|
|
|
��.��1����֪298Kʱ��1molC2H6����������ȫȼ�����ɶ�����̼��Һ̬ˮ���ų�����1558.3KJ��д���÷�Ӧ�Ļ�ѧ����ʽ__________________________________________.
 ��20���ø÷�Ӧ���һ��ȼ�ϵ��:������������Һ����� ����Һ���ö��ʯī���缫���ڵ缫�Ϸֱ���������������д�������ĵ缫��Ӧʽ____________________________
��20���ø÷�Ӧ���һ��ȼ�ϵ��:������������Һ����� ����Һ���ö��ʯī���缫���ڵ缫�Ϸֱ���������������д�������ĵ缫��Ӧʽ____________________________
![]() (3)�����һ�ѧʵ��װ��ͼ��
(3)�����һ�ѧʵ��װ��ͼ��
ʯī���ϵĵ缫��ӦʽΪ_______________________________;
�����ʼʱʢ��1000mLpH=5������ͭ��Һ��25�棩��CuSO4 ��������һ��ʱ�����Һ��pH��Ϊ1����Ҫʹ��Һ�ָ�����ʼŨ�ȣ�������Һ����ı仯����������Һ�м���________(����������)��������Ϊ_______________.
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com