
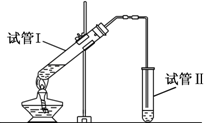
 ��ͼ���Թ�I���ȼ���3mL 95%���Ҵ�������ҡ���»�������2mLŨ���ᣬ���ҡ�ȣ���ȴ���ټ���2mL��ˮ���ᣬ�ò�������ֽ�����Թ̶ܹ�������̨�ϣ����Թܢ��м���5ml ���͵�̼������Һ����ͼ���Ӻ�װ�ã��þƾ��ƶ��Թ�IС�����3��5min���ô����ȣ����۲쵽�Թ�I������������ʱֹͣ���ȣ��Իش�
��ͼ���Թ�I���ȼ���3mL 95%���Ҵ�������ҡ���»�������2mLŨ���ᣬ���ҡ�ȣ���ȴ���ټ���2mL��ˮ���ᣬ�ò�������ֽ�����Թ̶ܹ�������̨�ϣ����Թܢ��м���5ml ���͵�̼������Һ����ͼ���Ӻ�װ�ã��þƾ��ƶ��Թ�IС�����3��5min���ô����ȣ����۲쵽�Թ�I������������ʱֹͣ���ȣ��Իش� CH3COOCH2CH3+H2O���÷�ӦΪ������Ӧ��
CH3COOCH2CH3+H2O���÷�ӦΪ������Ӧ�� CH3COOCH2CH3+H2O��������Ӧ��
CH3COOCH2CH3+H2O��������Ӧ��

 ��ѧȫ��������ѵ��ϵ�д�
��ѧȫ��������ѵ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ||
| ||
| ||
| ||
 Al��OH��3+OH-
Al��OH��3+OH- Al��OH��3+OH-
Al��OH��3+OH-�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
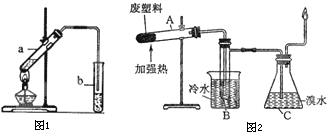
| ʵ�鲽�� | ʵ������ |
| I������Ϊ�٢ڢ۵�3֧�Թ��У��ֱ����1mL 20%��������Һ�����Թܢں͢��м���0.5mLϡ���ᣬ������3֧�Թ�ͬʱˮԡ����Լ5min | ������������ |
| II��ȡ�Թܢٺ͢ڣ���������������ͭ����Һ������������ | ������������ |
| III��ȡ�Թܢۣ��ȼ���NaOH��Һ����ҺpH�����ԣ��ټ�������������ͭ����Һ������������ | |
| ���ۣ�֤��������ϡ���������·�����ˮ�ⷴӦ | |
| ���� | ���� | ���� | ��ϩ | ��ϩ | �� | �ױ� | ̼ |
| ����������%�� | 12 | 24 | 12 | 16 | 20 | 10 | 6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ѧʵ����ȤС����ʵ���� ���������������е�ȼ��ʵ�顣
�������ͼ��ʾװ��I�Ʊ����ս����﴿���������Ա�����ʵ�飻

��ƿ�з�����Ӧ�����ӷ���ʽΪ ��
�����ʵ��Ŀ�Ľ�װ��I�����߿���װ�ò�����������������ע�������Լ���
����ͼ��ʾװ�â��У����Թ����ȼ���������ۣ��ٵ�������CaSO4��Һ��������һ����ϡ���ᣬ���Ͻ������۲쵽�д������ݲ�����

�ټ����ȡ����װ�������ԵIJ���������
�ڵ���ȼ�����������ʢ�������ļ���ƿ�У��۲쵽��������
������ֹͣȼ�պ�ȡ�����ܣ�ͬѧ�ǽ����˷���̽��ʵ�顣A��ͬѧ��������������Թ��л��н϶����ʣ�࣬ͬѧ�ǽ���������ͨ���Թ��У����ֹ���ȫ����ʧ����δ�۲쵽�����ݲ���������Ϳ���ԭ�������ӷ���ʽ��ʾ���� ��B��ͬѧ����ƿ��ע������ˮ��������������������Na2SO3��Һ��������Һ��û�г��ֻ�ɫ��������������Һ�е���ϡ�����ữ���Ȼ�����Һ�������˰�ɫ������[��֪Na2S2O3+H2SO4=Na2SO4+S��+SO2��+H2O]��ͬѧ�Ƿ��������ڼ���ƿ�л�����������������Ե�ʡ����Խ���ʵ��������������ӷ���ʽ�ǣ�
��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ѧʵ����ȤС����ʵ���ҽ��������������е�ȼ��ʵ��
��һ���������ͼ��ʾװ��I�Ʊ����ռ����﴿���������Ա�����ʵ�飺
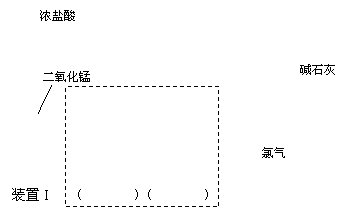

����ƿ�з�����Ӧ�����ӷ���ʽΪ�� ��
�������ʵ��Ŀ�Ľ�װ��I�����߿���װ�ò�����������������ע�������Լ���

 ����������ͼ��ʾװ��II�У����Թ����ȼ���������ۣ��ٵ�������CuSO4��Һ��������һ����ϡ���ᣬ���Ͻ������۲쵽�д������ݲ�����
����������ͼ��ʾװ��II�У����Թ����ȼ���������ۣ��ٵ�������CuSO4��Һ��������һ����ϡ���ᣬ���Ͻ������۲쵽�д������ݲ�����
�ټ����ȡ����װ�������ԵIJ���������
�ڵ���ȼ��������������ʢ�������ļ���ƿ�У��۲쵽��������
���Թ��м�������CuSO4��Һ�������� ����ԭ����
������������ֹͣȼ�պ�ȡ�����ܣ�ͬѧ�ǽ����˷���̽��ʵ�飬A��ͬѧ������ȡ��������Թ��л��н϶����ʣ�ࡣͬѧ�ǽ���������ͨ���Թ��У����ֹ���ȫ����ʧ����δ�۲쵽�����ݲ���������Ϳ���ԭ�������ӷ���ʽ��ʾ����
��B��ͬѧ����ƿ��ע������ˮ�������������е�������Na2S2O3��Һ��������Һ��û�г��ֻ�ɫ��������������Һ�е���ϡ�����ữ���Ȼ�����Һ�������˰�ɫ����������֪Na2S2O3+ H2SO4= Na2SO4+S��+SO2��+ H2O����ͬѧ�Ƿ��������ڼ���ƿ�л�����������������Ե�ʡ����Խ���ʵ��������������ӷ���ʽ�ǣ�
��
��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com