
ЈЁ6·ЦЈ©ОЄБЛСРѕїНвЅзМхјю¶Ф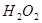 ·ЦЅв·ґУ¦ЛЩВКµДУ°ПмЈ¬ДіН¬С§ФЪЛДЦ§КФ№ЬЦР·Ц±рјУИл
·ЦЅв·ґУ¦ЛЩВКµДУ°ПмЈ¬ДіН¬С§ФЪЛДЦ§КФ№ЬЦР·Ц±рјУИл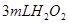 ИЬТєЈ¬ІўІвБїКХјЇ
ИЬТєЈ¬ІўІвБїКХјЇ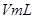 ЖшМеЛщРиµДК±јдЈ¬КµСйјЗВјИзПВЈє
ЖшМеЛщРиµДК±јдЈ¬КµСйјЗВјИзПВЈє
| КµСйРтєЕ | 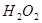 ИЬТєЕЁ¶И ИЬТєЕЁ¶И | 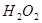 ИЬТєОВ¶И ИЬТєОВ¶И | ґЯ»ЇјБ | ЛщУГК±јд |
| ўЩ | 5% | 20Ўж | 2µО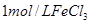 | 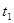 |
| ўЪ | 5% | 40Ўж | 2µО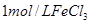 | 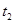 |
| ўЫ | 10% | 20Ўж | 2µО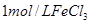 | 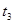 |
| ўЬ | 5% | 20Ўж | І»К№УГ | 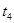 |
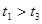 Ј¬ФТтКЗ__________ЎЈ
Ј¬ФТтКЗ__________ЎЈ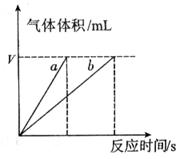

| Дкј¶ | ёЯЦРїОіМ | Дкј¶ | іхЦРїОіМ |
| ёЯТ» | ёЯТ»Гв·СїОіМНЖјцЈЎ | іхТ» | іхТ»Гв·СїОіМНЖјцЈЎ |
| ёЯ¶ю | ёЯ¶юГв·СїОіМНЖјцЈЎ | іх¶ю | іх¶юГв·СїОіМНЖјцЈЎ |
| ёЯИэ | ёЯИэГв·СїОіМНЖјцЈЎ | іхИэ | іхИэГв·СїОіМНЖјцЈЎ |
їЖДїЈєёЯЦР»ЇС§ АґФґЈє МвРНЈєФД¶БАнЅв
 ОЄБЛСРѕїНвЅзМхјю¶Ф№эСх»ЇЗв·ЦЅвЛЩВКµДУ°ПмЈ¬ДіН¬С§ЧцБЛТФПВКµСйЈ¬Зл»ШґрПВБРОКМвЈ®
ОЄБЛСРѕїНвЅзМхјю¶Ф№эСх»ЇЗв·ЦЅвЛЩВКµДУ°ПмЈ¬ДіН¬С§ЧцБЛТФПВКµСйЈ¬Зл»ШґрПВБРОКМвЈ®| ±аєЕ | ІЩ Чч | КµСйПЦПу |
| ўЩ | ·Ц±рФЪКФ№ЬAЎўBЦРјУИл 5mL 5% H2O2ИЬТєЈ¬ёчµОИл1Ў«2 µО1mol/L FeCl3ИЬТєЈ®ґэКФ№ЬЦРѕщУРККБїЖшЕЭіцПЦК±Ј¬Ѕ«КФ№ЬA·ЕИлКўУР5ЎжЧуУТАдЛ®µДЙХ±ЦРЈ»Ѕ«КФ№ЬB·ЕИлКўУР40ЎжЧуУТИИЛ®µДЙХ±ЦРЈ® | КФ№ЬAЦРІ»ФЩІъЙъЖшЕЭЈ¬КФ№ЬBЦРІъЙъµДЖшЕЭБїФцґу |
| ўЪ | БнИЎБЅЦ§КФ№Ь·Ц±рјУИл 5mL 5%H2O2ИЬТєєН 5mL 10%H2O2ИЬТєЈ® |
КФ№ЬAЎўBЦРѕщОґјыЖшЕЭІъЙъ |
| ||
| ||
Ійїґґр°ёєНЅвОц>>
їЖДїЈєёЯЦР»ЇС§ АґФґЈє МвРНЈєФД¶БАнЅв
| ±аєЕ | ІЩЧч | КµСйПЦПу |
| ўЩ | ·Ц±рФЪКФ№ЬAЎўBЦРјУИл5mL 5% H2O2ИЬТєЈ¬ёчµОИл2µО1mol/L FeCl3ИЬТєЈ®ґэКФ№ЬЦРѕщУРККБїЖшЕЭіцПЦК±Ј¬Ѕ«КФ№ЬA·ЕИлКўУР5ЎжЧуУТАдЛ®µДЙХ±ЦРЅюЕЭЈ»Ѕ«КФ№ЬB·ЕИлКўУР40ЎжЧуУТИИЛ®µДЙХ±ЦРЅюЕЭЈ® | КФ№ЬAЦРІ»ФЩІъЙъЖшЕЭЈ» КФ№ЬBЦРІъЙъµДЖшЕЭБїФцґуЈ® |
| ўЪ | БнИЎБЅЦ§КФ№Ь·Ц±рјУИл5mL 5% H2O2ИЬТєєН5mL 10% H2O2ИЬТє | КФ№ЬAЎўBЦРѕщОґГчПФјыµЅУРЖшЕЭІъЙъЈ® |
| ||
| ||

Ійїґґр°ёєНЅвОц>>
їЖДїЈєёЯЦР»ЇС§ АґФґЈє2014Ѕм±±ѕ©КРіЇСфЗшёЯТ»ПВС§ЖЪЖЪД©НіТ»їјКФ»ЇС§КФѕнЈЁЅвОц°жЈ© МвРНЈєМоїХМв
ЈЁ6·ЦЈ©ОЄБЛСРѕїНвЅзМхјю¶Ф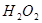 ·ЦЅв·ґУ¦ЛЩВКµДУ°ПмЈ¬ДіН¬С§ФЪЛДЦ§КФ№ЬЦР·Ц±рјУИл
·ЦЅв·ґУ¦ЛЩВКµДУ°ПмЈ¬ДіН¬С§ФЪЛДЦ§КФ№ЬЦР·Ц±рјУИл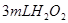 ИЬТєЈ¬ІўІвБїКХјЇ
ИЬТєЈ¬ІўІвБїКХјЇ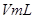 ЖшМеЛщРиµДК±јдЈ¬КµСйјЗВјИзПВЈє
ЖшМеЛщРиµДК±јдЈ¬КµСйјЗВјИзПВЈє
|
КµСйРтєЕ |
|
|
ґЯ»ЇјБ |
ЛщУГК±јд |
|
ўЩ |
5% |
20Ўж |
2µО |
|
|
ўЪ |
5% |
40Ўж |
2µО |
|
|
ўЫ |
10% |
20Ўж |
2µО |
|
|
ўЬ |
5% |
20Ўж |
І»К№УГ |
|
Зл»ШґрЈє
ЈЁ1Ј©№эСх»ЇЗв·ЦЅвµД»ЇС§·ЅіМКЅКЗ__________ЎЈ
ЈЁ2Ј©КµСйўЩўЪКЗСРѕї__________¶Ф·ґУ¦ЛЩВКµДУ°ПмЎЈ
ЈЁ3Ј©КµСйІвµГ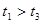 Ј¬ФТтКЗ__________ЎЈ
Ј¬ФТтКЗ__________ЎЈ
ЈЁ4Ј©КµСйўЩўЬµДІв¶Ё№эіМИзПВНјЈ¬ЗъПЯa¶ФУ¦µДКµСйРтєЕКЗ__________ЈЁМоЎ°ўЩЎ±»тЎ°ўЬЎ±Ј©ЎЈ
Ійїґґр°ёєНЅвОц>>
їЖДїЈєёЯЦР»ЇС§ АґФґЈє МвРНЈє
ОЄБЛСРѕїНвЅзМхјю¶Ф![]() ·ЦЅв·ґУ¦ЛЩВКµДУ°ПмЈ¬ДіН¬С§ФЪЛДЦ§КФ№ЬЦР·Ц±рјУИл
·ЦЅв·ґУ¦ЛЩВКµДУ°ПмЈ¬ДіН¬С§ФЪЛДЦ§КФ№ЬЦР·Ц±рјУИл![]() ИЬТєЈ¬ІўІвБїКХјЇ
ИЬТєЈ¬ІўІвБїКХјЇ![]() ЖшМеЛщРиµДК±јдЈ¬КµСйјЗВјИзПВЈє
ЖшМеЛщРиµДК±јдЈ¬КµСйјЗВјИзПВЈє
| КµСйРтєЕ |
|
| ґЯ»ЇјБ | ЛщУГК±јд |
| ўЩ | 5% | 20Ўж | 2µО |
|
| ўЪ | 5% | 40Ўж | 2µО |
|
| ўЫ | 10% | 20Ўж | 2µО |
|
| ўЬ | 5% | 20Ўж | І»К№УГ |
|
Зл»ШґрЈє
ЈЁ1Ј©№эСх»ЇЗв·ЦЅвµД»ЇС§·ЅіМКЅКЗ__________ЎЈ
ЈЁ2Ј©КµСйўЩўЪКЗСРѕї__________¶Ф·ґУ¦ЛЩВКµДУ°ПмЎЈ
ЈЁ3Ј©КµСйІвµГ![]() Ј¬ФТтКЗ__________ЎЈ
Ј¬ФТтКЗ__________ЎЈ
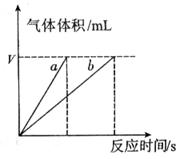
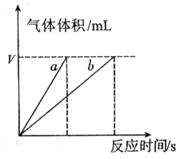
ЈЁ4Ј©КµСйўЩўЬµДІв¶Ё№эіМИзПВНјЈ¬ЗъПЯa¶ФУ¦µДКµСйРтєЕКЗ__________ЈЁМоЎ°ўЩЎ±»тЎ°ўЬЎ±Ј©ЎЈ
Ійїґґр°ёєНЅвОц>>
°Щ¶ИЦВРЕ - Б·П°ІбБР±н - КФМвБР±н
єю±±КЎ»ҐБЄНшОҐ·ЁєНІ»БјРЕПўѕЩ±ЁЖЅМЁ | НшЙПУРє¦РЕПўѕЩ±ЁЧЁЗш | µзРЕХ©ЖѕЩ±ЁЧЁЗш | ЙжАъК·РйОЮЦчТеУРє¦РЕПўѕЩ±ЁЧЁЗш | ЙжЖуЗЦИЁѕЩ±ЁЧЁЗш
ОҐ·ЁєНІ»БјРЕПўѕЩ±Ёµз»°Јє027-86699610 ѕЩ±ЁУКПдЈє58377363@163.com