

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
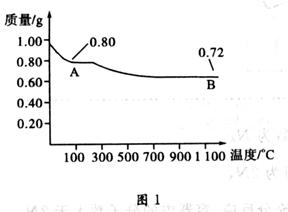
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��1��12g | B��6��42g | C��2��24g | D��3��24g |
�鿴�𰸺ͽ���>>
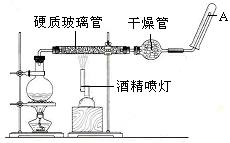
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
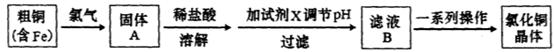
| A��NaOH | B��NH3.H2O | C��CuO | D��Cu(OH)2E. CuSO4 |
�鿴�𰸺ͽ���>>
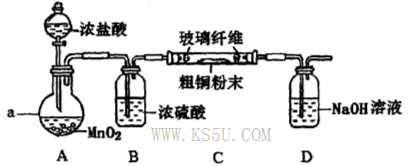
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
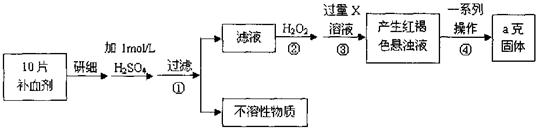
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ֱ�ӼӼ������ҺpH��9.6 |
| B���Ӵ�ͭ�ۣ���Fe2����ԭ���� |
| C���Ƚ�Fe2��������Fe3�����ٵ���pH��3��4 |
| D��ͨ�����⣬ʹFe2��ֱ�ӳ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

| ʵ����� | ʵ����������� |
| | |
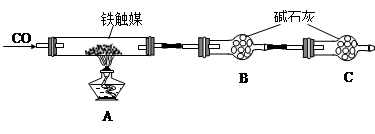
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| ʵ����� | ʵ������ | ���� |
| ��1mL 0.1mol/L��FeCl2��Һ�м�2��KSCN��Һ | ��Һ����� | Fe2+��SCN-����� |
| ����ٵ���Һ�м�3%��H2O21�β��� | ��������Ѫ��ɫ ����ɫ | ����H2O2����Һ�� ������ (���ӷ���) |
| ����ڵ���Һ�м�����H2O2��Һ | ��Һ�г��ִ������� Ѫ��ɫ��ȥ | |
| ���ô����ǵ�ľ��������е����� | ľ����ȼ | ����� ���� |
| ʵ����� | ʵ������ | ���� |
| ��ȡ2ml KSCN��Һ�����м��뼸��BaCl2��Һ��ϡ���� | ���������� | |
| ��������õ���Һ�еμ�3%��H2O2 | ��Һ�г��ְ�ɫ���������������� | ��ɫ����ΪBaSO4 |
| �۽�6%��H2O2��Һ����KSCN�����У����ɵ���������ͨ��Ʒ����Һ������KMnO4��Һ�ͳ����ʯ��ˮ | | KSCN��H2O2����������SO2��CO2���� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com