
����Ŀ��ij̼������Ʒ�п��ܺ����������ơ�̼��ơ���ʯ�ҡ��Ȼ��ơ�����ͭ���������е����֡��ֽ�������ʵ�飺
�ٳ�ȡ4.7g ��Ʒ����������ˮ����Ʒ�����ܽ⡣
���������������Һ�м���100mL1 mol/LHCl�����յõ���ɫ������Һ�� �˹����й�����0.04 mol���塣
������з�Ӧ����Һ�м�����������������ϡ���ᣬ�õ�15.8g��ɫ������
�ɴ˿�֪�����У� ��
A.һ����NaCl��һ������CuSO4B.���ܺ�CaO��NaOH
C.һ����CaCO3�����ܺ�NaOHD.���ܺ�CaO������CaCO3
���𰸡�AC
��������
�������������Һ�м���100mL1 mol/LHCl�����յõ���ɫ������Һ����֪��Ʒ�в���CuSO4���˹����й�����0.04 mol���壬��CO2����CO32-+2H+=CO2+H2O��֪��Ӧ������n(HCl)=2n(CO2)=0.08mol����HCl��ʣ�࣬CO32-��ȫ��Ӧ����ȡ4.7g ��Ʒ����������ˮ����Ʒ�����ܽ⣬��֪��Ʒ�к���CaCO3��CaCO3��CaO��CaO�����������һ�֣�����з�Ӧ����Һ�м�����������������ϡ���ᣬ�õ�15.8g��ɫ��������AgCl��n(AgCl)=![]() >0.1mol����ԭ��Ʒ��һ����NaCl������Ϊ(
>0.1mol����ԭ��Ʒ��һ����NaCl������Ϊ(![]() -0.1mol)��58.5g/mol
-0.1mol)��58.5g/mol![]() 0.59g����ԭ��Ʒ�������ɷ�������Ϊ4.7g-0.59g=4.11g��������CaCO3����̼Ԫ�����ʵ���<
0.59g����ԭ��Ʒ�������ɷ�������Ϊ4.7g-0.59g=4.11g��������CaCO3����̼Ԫ�����ʵ���<![]() �����������⣬��ԭ��Ʒ��һ������CaCO3������ԭ��Ʒֻ���������ʣ���CaO��NaOHֻ������һ�֡�
�����������⣬��ԭ��Ʒ��һ������CaCO3������ԭ��Ʒֻ���������ʣ���CaO��NaOHֻ������һ�֡�
�������������Һ�м���100mL1 mol/LHCl�����յõ���ɫ������Һ����֪��Ʒ�в���CuSO4���˹����й�����0.04 mol���壬��CO2����CO32-+2H+=CO2+H2O��֪��Ӧ������n(HCl)=2n(CO2)=0.08mol����HCl��ʣ�࣬CO32-��ȫ��Ӧ����ȡ4.7g ��Ʒ����������ˮ����Ʒ�����ܽ⣬��֪��Ʒ�к���CaCO3��CaCO3��CaO��CaO�����������һ�֣�����з�Ӧ����Һ�м�����������������ϡ���ᣬ�õ�15.8g��ɫ��������AgCl��n(AgCl)=![]() >0.1mol����ԭ��Ʒ��һ����NaCl������Ϊ(
>0.1mol����ԭ��Ʒ��һ����NaCl������Ϊ(![]() -0.1mol)��58.5g/mol
-0.1mol)��58.5g/mol![]() 0.59g����ԭ��Ʒ�������ɷ�������Ϊ4.7g-0.59g=4.11g��������CaCO3��̼Ԫ�����ʵ���<
0.59g����ԭ��Ʒ�������ɷ�������Ϊ4.7g-0.59g=4.11g��������CaCO3��̼Ԫ�����ʵ���<![]() �����������⣬��ԭ��Ʒ��һ������CaCO3������ԭ��Ʒֻ���������ʣ���CaO��NaOHֻ������һ�֣�
�����������⣬��ԭ��Ʒ��һ������CaCO3������ԭ��Ʒֻ���������ʣ���CaO��NaOHֻ������һ�֣�
A���ɷ�����֪��ԭ��Ʒ��һ����NaCl��һ������CuSO4����A��ȷ��
B���ɷ�����֪��ԭ��Ʒ�в�����ͬʱ��CaO��NaOH����B����
C���ɷ�����֪��ԭ��Ʒ��һ����CaCO3�����ܺ�NaOH����C��ȷ��
D���ɷ�����֪��ԭ��Ʒ�п��ܺ�CaO��һ������CaCO3����D����
�ʴ�ѡAC��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ij��ѧС���������������������װ�ã���ͼ�����û������ƻ���ϩ��
��֪��![]()
�ܶȣ� | �۵�� | �е�� | �ܽ��� | |
������ | 0.096 | 25 | 161 | ������ˮ |
����ϩ | 0.081 | -103 | 83 | ������ˮ |
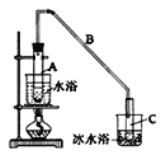
�Ʊ���Ʒ��
��12.5mL�����������Թ�A�У��ټ���lmLŨ���ᣬҡ�Ⱥ�������Ƭ��������������Ӧ��ȫ�����Թ�C�ڵõ�����ϩ��Ʒ��
��1��A�д�Ƭ��������______������B���˵�������е�������______��
��2���Թ�C���ڱ�ˮԡ�е�Ŀ����__________________________��
�Ʊ���Ʒ��
��3������ϩ��Ʒ�к��л������������������ʵȡ���������ʳ��ˮ�������á��ֲ㣬����ϩ��_______�㣨����������������������Һ����_________�������ţ�ϴ�ӣ�
a��KMnO4��Һ b��ϡH2SO4 c��Na2CO3��Һ
��4���ٽ�����ϩ��ͼ2װ��������ȴˮ��_______����f��g���ڽ��룬����ʱҪ������ʯ�ң�Ŀ����______________���ռ���Ʒʱ���¶�Ӧ������_____���ҡ�
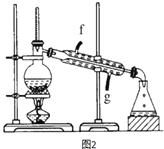
��5���������ֻ���ϩ��Ʒ���Ʒ�ķ�������������_________��
a�������Ը��������Һ b���ý����� c���ⶨ�е�
��������װ���Ʊ������������ش��������⡣
��6���÷�Һ©���ᴿ�Թ�C�е������������ڲ���ʱҪע����ϴ�Ӻ��÷�Һǰ����������������������__________�����ţ�
a�������������ڼ��ӻ���Ȧ�ϴ��ϲ�����
b�������������ڼ��ӻ���Ȧ�ϴ���
c�������ĵ�ס�����ϲ����ӵ��ú����
d����������ƽ����ʵ��̨�ϴ���
��7�����Ƶõ���Ӧ�Ӹ÷�Һ©����__________�����ţ�
a���²����� b���Ͽڵ��� c��������
��8�����ᴿ��������ʱ��ΪʲôҪʹ�ñ���̼������Һ��������NaOH��Һϴ�ӣ�
______________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����з�Ӧ�IJ�����һ��������ͬ���칹����ǣ� ��
A.CH2=CHCH3 ��HBrB.![]() ��NaOH �Ҵ���Һ����
��NaOH �Ҵ���Һ����
C.1��3������ϩ������ 1��1 ��ӦD.![]() ��NaHCO3 ��Һ
��NaHCO3 ��Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������֪Ũ�ȵ���ζ�δ֪Ũ�ȵļ�ʱ���ᵼ�´����Һ��Ũ��ƫ�͵IJ�����
����ʽ�ζ���������ˮϴ��δ�ñ�Һ��ϴ
����ʽ�ζ���������ˮϴ��δ�ô���Һ��ϴ
�����Ƽ�Һʱ�������Ĺ�������
���ζ�ǰ��ʽ�ζ��ܼ��첿��δ������Һ
���ζ��в�������ƿ��Һ��ҡ��������ƿ��
A.�٢ۢ�B.�ڢ�C.�ڢۢ�D.�٢�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������£��ס������ձ���ʢ��5mLpH=3�Ĵ�����Һ�������ձ���ˮϡ������ҺpH=4���������ձ���������Һ��������ȷ���ǣ� ��
A.��Һ�����10V��<V��
B.ˮ�������OH-Ũ�ȣ�10c��OH-���� < c��OH-����
C.���ֱ��õ����ʵ���Ũ�ȵ�NaOH��Һ��ȫ�кͣ�������Һ��pH����<��
D.���ֱ���5mLpH=11��NaOH��Һ��Ӧ��������Һ��pH����<��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������й�����߲�������Ҷ���ص�˵������ȷ���ǣ� ��
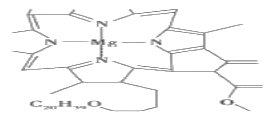
A.����������λ��Ϊ4
B.�û�����������Ԫ�صķǽ����ԣ�O>N>C>H>Mg
C.Mg���ʾ���ṹ����A3�����ܶѻ�
D.��������CԪ�ص��ӻ���ʽsp2��sp3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����֪X��Y��Z������ԭ��������������Ķ�����Ԫ�ء��ס��ҡ����ֱ�������Ԫ���γɵĵ��ʣ�A��B��C��D�ֱ���������Ԫ���е������γɵĻ������A��C�о�����10 �����ӡ�����֮��ת����ϵ����ͼ��ʾ������˵����ȷ����
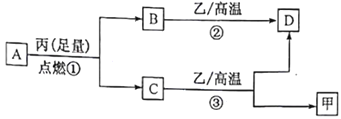
A. ԭ�Ӱ뾶��Z>Y>X
B. X��Y�γɵĻ�����ֻ�����Լ�
C. Y�ж���ͬ�������壬�Ҿ����и��۵㡢�߷е㡢Ӳ�ȴ������
D. ��̬�⻯����ȶ��ԣ�A<C
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʵ������ȼ�շ��ⶨij�ְ�����(CxHyOzNm)�ķ�����ɡ�ȡWg���ְ�������ڴ����г��ȼ�գ����ɶ�����̼��ˮ�͵���������ͼ��ʾװ�ý���ʵ�顣

�ش��������⣺
��1��ʵ�鿪ʼʱ������ͨ��һ��ʱ�����������������__________________��
��2������װ������Ҫ���ȵ�������_______ (��д��ĸ)������ʱӦ�ȵ�ȼ_____���ľƾ��ơ�
��3��Aװ���з�����Ӧ�Ļ�ѧ����ʽ��____________________________��
��4��Dװ�õ�������____________________________��
��5����ȡ���������ʱ��Ӧע����_________________����_________________��
��6��ʵ���в�õ��������ΪVmL(��״��)��Ϊȷ���˰�����ķ���ʽ������Ҫ���й�������____________________��
A�����ɶ�����̼���������
B������ˮ������
C��ͨ�����������
D�����������Է�������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ijͬѧ��ȡ9.00g��������ˮ���ⶨ���۵�ˮ��ٷ��ʣ���������£�
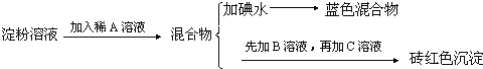
�Իش��������⣺
��1������������Լ�Ϊ��A_________��B_________��C_________��
��2������A��Һ��������B��Һ�Ƿ����_________����������_________��
��3��д������ˮ��ķ���ʽ_________��
��4��������1.44gש��ɫ����ʱ������ˮ������_________��[��֪��������Cu��OH��2��Ӧ�Ļ�ѧ����ʽΪ��CH2OH��CHOH��4CHO+2Cu��OH��2![]() CH2OH��CHOH��4COOH+Cu2O��+2H2O]��
CH2OH��CHOH��4COOH+Cu2O��+2H2O]��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com