
�������ѹ㷺Ӧ���ڸ���ṹ����Ϳ�ϡ�ֽ��Ϳ��ȣ��������ѻ�����Ϊ�Ʊ��ѵ��ʵ�ԭ�ϡ�
�������ѿ����������ַ����Ʊ���
����1��TiCl4ˮ������TiO2��xH2O�����ˡ�ˮϴ��ȥ���е�Cl�����ٺ�ɡ����ճ�ȥˮ�ֵõ�����TiO2���˷����Ʊ��õ��������������ѡ�
��1���� TiCl4ˮ������TiO2��x H2O�Ļ�ѧ����ʽΪ_______________________________��
�� ����TiO2��x H2O��Cl���Ƿ����ķ�����______________________________��
����2�����ú���Fe2O3����������Ҫ�ɷ�ΪFeTiO3������TiԪ�ػ��ϼ�Ϊ+4�ۣ���ȡ������Ҫ�������£�
��2��Fe2O3��H2SO4��Ӧ�����ӷ���ʽ�� ��
��3������Һ�г���TiO2+֮����еĽ����������� ��
��4����Fe�������� ��
��.�������ѿ�������ȡ�ѵ���
��5��TiO2��ȡ����Ti���漰���IJ������£�
��Ӧ�ڵķ���ʽ�� ���÷�Ӧ��Ҫ��Ar�����н��У������ԭ��_____________��
��1����TiCl4+��x+2��H2O==TiO2?xH2O��+4HCl��ȡ���һ��ϴ��Һ���������Թ��У��μ������ữ��AgNO3��Һ����������ɫ������˵��Cl-�ѳ���
��2��Fe2O3+6H+=2Fe3++3H2O
��3��Fe3+��Fe2+��
��4����Fe3+ת��ΪFe2+
��5��TiCl4+2Mg=2MgCl2+Ti�� ��ֹ������Mg��Ti��������е�O2����CO2��N2�����ã�
���������������1������TiCl4��ϵ��Ϊ1������Ԫ���غ㣬TiO2?xH2O��ϵ��Ϊ1��HCl��ϵ��Ϊ4���ٸ���OԪ���غ㣬��֪H2O��ϵ��Ϊ��2+x��������ʽΪTiCl4+��x+2��H2O?TiO2?xH2O��+4HCl���ڳ���������Һ�е�Cl-�����ݷ�����Ӧ��Cl-+Ag+�TAgCl����ȡ����ϴ��Һ��������Һ���ܽ�������Ƿ��ڣ�
��2�������������ᷴӦ������������ˮ�����ӷ���ʽΪ��Fe2O3+6H+=2Fe3++3H2O��
��3��Fe2O3+6H+=2Fe3++3H2O��FeTiO3+4H+=Fe2++TiO2++2H2O�����Ի����ڵ���������Fe3+��Fe2+
��4������Һ�к��������ӣ������л�ԭ�ԣ��ܽ������������������������Ҳ������µ����ʣ��������������ǣ���Fe3+ת��ΪFe2+
��5����800�������£����Ȼ��Ѻ�þ��Ӧ�����Ȼ�þ���ѣ���Ӧ����ʽΪ��TiCl4+2Mg==2MgCl2+Ti ��Mg�ǻ��ý�������������ж������ʷ�Ӧ����˿ɵó�Ar������Ϊ����������ֹMg�Ϳ��������ʷ�Ӧ
���㣺����ѧ���Թ����������⡢�Ķ���Ŀ��ȡ��Ϣ������������ԭ��Ӧ������ʽ����д�ȣ��Ѷ��еȣ�����������ǹؼ�����Ҫѧ�������Ķ���Ŀ��ȡ��Ϣ������������û���֪ʶ�������⡢�������������


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������˵������ȷ����____ ��
| A����������ijЩ���ַ����������� |
| B����������Ӳ�ȵ�ˮ��Ҫ�ü��ȵķ������������� |
| C�����Ṥҵ�У��ڽӴ��Ұ�װ�Ƚ�������Ϊ������S03ת��ΪH2S04ʱ�ų������� |
| D���ϳɰ���ҵԭ��������ʱ������̼�����Һ���ճ�ȥ������̼ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
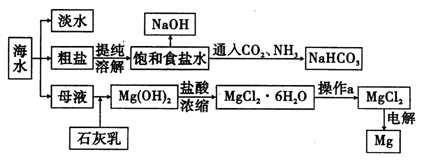
��������к���Cr(OH)3��Al2O3��ZnO��CuO��NiO������,��ҵ��ͨ�������±��ա���������������Na2Cr2O7�����ʡ�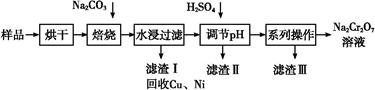
��֪:��Na2CrO4��Һ�к�������NaAlO2��Na2ZnO2������
(1)ˮ�������Һ������������(��ᡱ��������С�)��
(2)����������չ���������Na2CrO4�Ļ�ѧ����ʽ��
��������Cr(OH)3+��������Na2CO3+������������ ��������Na2CrO4+��������CO2+������������
��������Na2CrO4+��������CO2+������������
(3)���������Ҫ�ɷ���Zn(OH)2������������
(4)��ϵ�в�������Ϊ:��������H2SO4,��������,��ȴ�ᾧ,���ˡ���������H2SO4Ŀ������ ��
��֪:�ٳ�ȥ����II��,��Һ�д������·�Ӧ:
2CrO42��+2H+ Cr2O72��+H2O
Cr2O72��+H2O
��Na2Cr2O7��Na2CrO4�ڲ�ͬ�¶��µ��ܽ�����±�
| �¶� �ܽ�� ��ѧʽ | 20 �� | 60 �� | 100 �� |
| Na2SO4 | 19.5 | 45.3 | 42.5 |
| Na2Cr2O7 | 183 | 269 | 415 |
| Na2CrO4 | 84 | 115 | 126 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ�����÷���м(����������������������)������ʽ������[Fe(OH)SO4]�Ĺ����������£�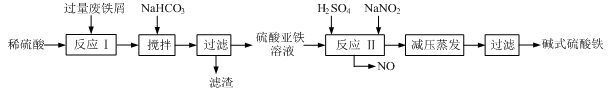
��֪������������������������ʽ����ʱ��Һ��pH���±���
| ������ | Fe(OH)3 | Fe(OH)2 | Al(OH)3 |
| ��ʼ���� | 2.3 | 7.5 | 3.4 |
| ��ȫ���� | 3.2 | 9.7 | 4.4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
̼��﮹㷺Ӧ�����մɺ�ҽҩ��������﮻�ʯ����Ҫ�ɷ�ΪLiAlSi2O6��Ϊԭ�����Ʊ�Li2CO3�Ĺ����������£�
��֪����2LiAlSi2O6��H2SO4(Ũ)  Li2SO4��Al2O3��4SiO2��H2O��
Li2SO4��Al2O3��4SiO2��H2O��
��Fe3+��Al3+��Fe2+��Mg2+������������ʽ��ȫ����ʱ����Һ��PH�ֱ�Ϊ3.2��4.7��9.0��11.1
��ijЩ���ʵ��ܽ�ȣ�S�����ұ�
��ش��������⣺
��1��﮻�ʯ��Ũ�����ȡ֮ǰҪ�����ϸ������Ŀ���� ��
��2����Һa�к���Li+��SO42-,������Fe3+��Al3+��Fe2+��Mg2+ ��Ca2+��Na+�����ʣ���������ڽ����¼���ʯ��ʯ�Ե�����Һ��pH��6.0��6.5����ʱ���������������� ��
��3�����������Һa�м���ij��Ӽ�����Ϊ������H2O2��Һ��ʯ�����Na2CO3��Һ��������Ӧ�����ӷ���ʽ�� ��
��4��������м��뱥��Na2CO3��Һ���˺���Ҫ����ˮϴ�ӵ�ԭ���� ��
��5������Һc�пɻ��յ���Ҫ������ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������ⷨ���Ѱ۲����ķ�Һ[���д���FeSO4��H2SO4������Fe2(SO4)3��TiOSO4]����������Ͳ�Ѫ�������������������������£�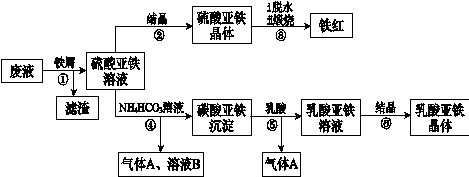
��֪��TiOSO4������ˮ����ˮ�п��Ե���ΪTiO2+��SO42������ش�
��1��������з�������������Һ�������IJ��������õIJ��������� ��
����ڵõ�������������IJ���Ϊ����Ũ���� ��
��1������ܵ����ӷ���ʽ�� ��
��1������ޱ������һ������նȣ�ԭ��������������ˮ�Լ� ��
��1�����������ڿ�������������������������÷�Ӧ���������ͻ�ԭ�������ʵ���֮��Ϊ ��
��1����ƽ���ƶ���ԭ�����Ͳ�����м������ܵõ�����������ԭ�� ��
��1��Ϊ�ⶨ����������þ�����FeSO4��7H2O������������ȡ������Ʒa g������ϡ�������100��00 mL��Һ��ȡ��20��00 mL��Һ����KMnO4��Һ�ζ���������KMnO4����Ӧ����������0��1000 mol?L-1 KMnO4��Һ20��00 mL�����þ�����FeSO4��7H2O����������Ϊ����a��ʾ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������أ�K2FeO4����һ�ּ�������������������һ������Ͷ��ˮ����������ҵ�ϳ�����NaClO�������������������������£�
��Ҫ��ӦΪ��3NaClO��2Fe(NO3)3��10NaOH �� 2Na2FeO4����3NaCl��6NaNO3��5H2O
Na2FeO4��2KOH��K2FeO4��2NaOH��
��1��д����Ӧ�ٵ����ӷ���ʽ�� ��
��2���ӡ������II���з����K2FeO4���и���Ʒ�����Ƕ�����Ҫ�Ļ�����Ʒ������һ���ڹ�ҵ�������ȼҵԭ�ϵ������� ��
��3����Ӧ���¶ȡ�ԭ�ϵ�Ũ�Ⱥ���ȶԸ�����صIJ��ʶ���Ӱ�졣ͼ��Ϊ��ͬ���¶��£�Fe(NO3)3��ͬ����Ũ�ȶ�K2FeO4�����ʵ�Ӱ�죻ͼ��Ϊһ���¶��£�Fe(NO3)3����Ũ�����ʱ��NaClOŨ�ȶ�K2FeO4�����ʵ�Ӱ�졣
ͼ�� ͼ��
������ͼ��������ҵ����������¶�Ϊ �棬��ʱFe(NO3)3��NaClO������Һ�������Ũ��֮��Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������������Ʒ�ӹ����ϣ�������GeO2����Ҫ;��������������ͼ��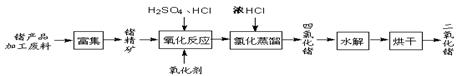
��1��Ge2+��������H2O2��Ӧ����Ge4+��д���÷�Ӧ�����ӷ���ʽ ��
��2������ɻ�GeCl4���ڴ˹����м���Ũ�����ԭ���� ��
��3��GeCl4ˮ������GeO2��nH2O����ѧ����ʽΪ ���¶ȶ�GeCl4��ˮ���ʲ�����Ӱ����ͼ1��ʾ����ԭ���Ǹ�ˮ�ⷴӦ��H 0����>����<������Ϊ������ѵķ�Ӧ�¶ȣ�ʵ��ʱ�ɲ�ȡ�Ĵ�ʩΪ ˮԡ��
A���ñ�ˮ����� B��49��ˮԡ C���ñ���ˮ
��4����֪Ge�ĵ��ʺͻ�����������Al�����ƣ��������ӷ���ʽ��ʾ��pH>8����Һ��GeO2���ܽ�ʱ�����ķ�Ӧ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
��ͼ��ʾ��һ���ۺϴ���SO2�����Ĺ������̣���ÿ������ȫ��Ӧ������˵����ȷ���� (����)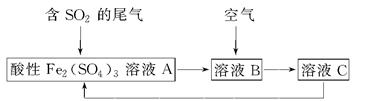
| A����ҺB�з����ķ�ӦΪ2SO2��O2=2SO3 |
| B���������Ը��������Һ������ҺC���Ƿ���Fe3�� |
| C�����������̿���֪�����ԣ�Fe3��>O2>SO42�� |
| D���˹��յ��ŵ�֮һ��������ѭ������ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com