
ŅŃÖŖ¶žŃõ»ÆĮņæÉŹ¹ĖįŠŌøßĆĢĖį¼ŲČÜŅŗĶŹÉ«£¬»Æѧ·“Ó¦·½³ĢŹ½ĪŖ£ŗ
5SO2 + 2KMnO4 + 2H2O £½ K2SO4 + 2MnSO4 + 2H2SO4
ÓĆĻĀĶ¼×°ÖĆĄ“ŃéÖ¤ÅØĮņĖįÓėľĢæŌŚ¼ÓČČĢõ¼žĻĀ·“Ó¦µÄ²śĪļÖŠŗ¬ÓŠSO2ŗĶCO2
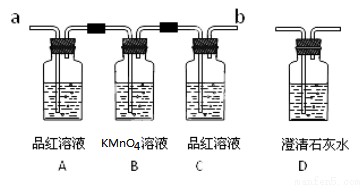
(1) Š“³öÅØĮņĖįŗĶľĢæŌŚ¼ÓČČĢõ¼žĻĀ·¢Éś·¢Ó¦µÄ»Æѧ·½³ĢŹ½£ŗ________________
(2) ŹµŃ鏱£¬·“Ó¦²śÉśµÄĘųĢåÓ¦“Ó_________¶ĖĶØČė£» “Ó________¶ĖĮ¬½ÓŹ¢ÓŠ³ĪĒåŹÆ»ŅĖ®µÄŹµŃé×°ÖĆ(ÓĆ ”°a”±»ņ”°b”± ĢīæÕ)”£
(3) BĘæµÄ×÷ÓĆŹĒ_______________”£
(4) Ö¤Ć÷»ģŗĻĘųĢåÖŠŗ¬ÓŠ¶žŃõ»ÆĢ¼µÄĻÖĻóŹĒ ____________”¢____________”£

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2016-2017ѧğ°²»ÕŹ”Āķ°°É½ŹŠø߶žÉĻŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
(1)ÓĆCH4“߻ƻ¹ŌNOxæÉŅŌĻū³żµŖŃõ»ÆĪļµÄĪŪČ¾”£ĄżČē£ŗ
CH4(g)£«4NO2(g)===4NO(g)£«CO2(g)£«2H2O(g)”” ¦¤H£½£574 kJ”¤mol£1
CH4(g)£«4NO(g)===2N2(g)£«CO2(g)£«2H2O(g) ¦¤H£½£1 160 kJ”¤mol£1
ČōÓƱź×¼×“æöĻĀ4.48 L CH4»¹ŌNO2Éś³ÉN2£¬·“Ó¦ÖŠ×ŖŅʵĵē×Ó×ÜŹżĪŖ________ (°¢·ü¼ÓµĀĀŽ³£ŹżÓĆNA±ķŹ¾)£¬·Å³öµÄČČĮæĪŖ________kJ”£
(2)ŅŃÖŖ£ŗC3H8(g) == CH4(g)£«HC”ŌCH(g)£«H2(g)””¦¤H1£½£«156.6 kJ”¤mol£1
CH3CH=CH2(g) == CH4(g)£«HC”ŌCH(g) ¦¤H2£½£«32.4 kJ”¤mol£1
ŌņĻąĶ¬Ģõ¼žĻĀ£¬·“Ó¦C3H8(g) === CH3CH=CH2(g)£«H2(g)µÄ¦¤H£½______kJ”¤mol£1”£
(3)¼×ĶéŌŚøßĪĀĻĀÓėĖ®ÕōĘų·“Ó¦µÄ·½³ĢŹ½ĪŖCH4(g)£«H2O(g) CO(g)£«3H2(g)”£²æ·ÖĪļÖŹµÄČ¼ÉÕČČŹż¾ŻČēĻĀ±ķ£ŗŅŃÖŖ1 mol H2O(g)×Ŗ±äĪŖ1 mol H2O(l)Ź±·Å³ö44.0 kJČČĮ攣Š“³öCH4ŗĶH2OŌŚøßĪĀĻĀ·“Ó¦µÄČČ»Æѧ·½³ĢŹ½£ŗ _______________________________”£
CO(g)£«3H2(g)”£²æ·ÖĪļÖŹµÄČ¼ÉÕČČŹż¾ŻČēĻĀ±ķ£ŗŅŃÖŖ1 mol H2O(g)×Ŗ±äĪŖ1 mol H2O(l)Ź±·Å³ö44.0 kJČČĮ攣Š“³öCH4ŗĶH2OŌŚøßĪĀĻĀ·“Ó¦µÄČČ»Æѧ·½³ĢŹ½£ŗ _______________________________”£
ĪļÖŹ | Č¼ÉÕČČ(kJ”¤mol£1) |
H2(g) | £285.8 |
CO(g) | £283.0 |
CH4(g) | £890.3 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2016-2017ѧğŗŚĮś½Ź”ĵµ¤½ŹŠøßŅ»ĻĀѧʌæŖѧ¼ģ²ā»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
£Ø1£©ŹµŃéŹŅÉŁĮæµÄ½šŹōÄʱ£“ęŌŚ___________ÖŠ£¬Č”ÓĆŹ±ÓƵ½µÄŅĒĘ÷ŗĶÓĆĘ·ÓŠŠ”µ¶”¢²£Į§Ę¬”¢ĀĖÖ½ŗĶ____________, Ź£ÓąµÄÄĘÓ¦_______________£»½«Ņ»Š”æéÄĘĶ¶Čėµ½ĮņĖįĶČÜŅŗÖŠ£¬·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ______________________________________£¬¹Ū²ģµ½µÄĻÖĻóĪŖ_____________(ĢīŠ“±źŗÅ)”£
a£®ÄĘø”ŌŚŅŗĆęÉĻĖÄ“¦ÓĪ¶Æ b£®ÄĘČŚ³ÉĮĖŅ»øöÉĮĮĮµÄŠ”Ēņ
c£®ČÜŅŗÖŠÓŠĄ¶É«³ĮµķÉś³É d£®ČÜŅŗÖŠÓŠŗģÉ«¹ĢĢåĪö³ö
£Ø2£©ŹµŃéŹŅÖĘĀČĘųµÄ»Æѧ·“Ó¦·½³ĢŹ½ĪŖ_________________________________£»ĀČĘųĪŖÓŠ¶¾ĘųĢ壬ŠčÓĆĒāŃõ»ÆÄĘČÜŅŗ½ųŠŠĪ²Ęų“¦Ąķ£¬ĒėĶź³ÉøĆĄė×Ó·½³ĢŹ½____________£¬ øĆŌĄķ»¹æÉÓĆÓŚ¹¤ŅµÖĘ______________”£
£Ø3£©ĻĀĮŠĪļÖŹ¼ČÄÜÓėŃĪĖį·“Ó¦£¬ÓÖÄÜÓėÉÕ¼īČÜŅŗ·“Ó¦µÄŹĒ________________________________
a. Al b. Mg c. CH3COONH4 d. NaHCO3 e. Al2O3
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2016-2017ѧğŗŚĮś½Ź”ĵµ¤½ŹŠøßŅ»ĻĀѧʌæŖѧ¼ģ²ā»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
100ml 1mol/LµÄAlCl3ČÜŅŗÓė100ml 3.5mol/LµÄNaOHČÜŅŗ»ģŗĻ£¬µĆµ½³ĮµķĪŖ( )
A. 7.8g B. 0g C. 9.1 g D. 3.9g
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2016-2017ѧğŗŚĮś½Ź”ĵµ¤½ŹŠøßŅ»ĻĀѧʌæŖѧ¼ģ²ā»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ĻĀĮŠø÷×éĪļÖŹÓėĘäÓĆĶ¾µÄ¹ŲĻµ²»ÕżČ·µÄŹĒ( )
A. ¹żŃõ»ÆÄĘ£ŗ¹©Ńõ¼Į B. ÉÕ¼ī£ŗ ÖĪĮĘĪøĖį¹ż¶ąµÄŅ»ÖÖŅ©¼Į
C. Š”ĖÕ“ņ£ŗ ·¢½Ķ·ŪÖ÷ŅŖ³É·Ö D. Ć÷·Æ£ŗ ¾»Ė®¼Į
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2016-2017ѧğ°²»ÕŹ”°²ĒģŹŠøßŅ»ÉĻŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ĻĀĮŠĄØŗÅÄŚĪŖŌÓÖŹ£¬Ń”ĻīÖŠ³żŌÓÖŹµÄ·½·ØÕżČ·µÄŹĒ
A. Cu £ØCuO£© ¼ÓČėĻ”ĻõĖįČÜŅŗ£¬Č»ŗó¹żĀĖĻ“µÓøÉŌļ
B. CO2 ( SO2 ) Ķعż±„ŗĶNa2 CO3 ČÜŅŗ
C. CO2 £ØO2 £© Ķعż×ĘČȵÄĶĶų
D. NO2 £ØNO£© ĶعżĖ®Ļ“
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2016-2017ѧğ°²»ÕŹ”°²ĒģŹŠøßŅ»ÉĻŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ÓŠ¹ŲSiO2»ņ¹čĖįŃĪµÄĖµ·ØÕżČ·µÄŹĒ
A. Ė®Äą”¢ŹÆÓ¢²£Į§”¢ĢՓɾłŹĒ¹čĖįŃĪ²śĘ·
B. ¹čĢ«ŃōÄܵē³Ų°åĖłÓƵďĒøß“æ¶ČSiO2
C. øֻƲ£Į§ÓėĘÕĶز£Į§³É·ÖĻąĶ¬
D. SiO2¼ČÄÜÓėHFĖį·“Ó¦ÓÖÄÜÓėNaOH·“Ó¦£¬¹ŹSiO2ĪŖĮ½ŠŌŃõ»ÆĪļ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗŗžÄĻŹ”³¤É³ŹŠ2016-2017ѧğøßŅ»ÉĻŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌ¾ķ ĢāŠĶ£ŗŃ”ŌńĢā
ĻĀĮŠĄė×Ó·½³ĢŹ½ÕżČ·µÄŹĒ
A. ½«ĀČĘųČÜÓŚĖ®Öʱø“ĪĀČĖį£ŗCl2+H2OØT2H++Cl-+ClO-
B. Ģ¼ĖįøĘÓėĻ”ŃĪĖį·“Ó¦£ŗCO32-+2H+=H2O+CO2”ü
C. ĮņĖįĀĮŗĶ°±Ė®·“Ó¦£ŗAl3++3OH-£½Al(OH)3”ż
D. NaÓėH2O·“Ó¦£ŗ2Na+2H2O=2Na++2OH-+H2”ü
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2017½ģø£½ØŹ”½ś½ŹŠµČĖÄŠ£øßČżµŚ¶ž“ĪĮŖŗĻæ¼ŹŌĄķ×Ū»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
Īż(Sn)ŹĒŅ»ÖÖÖŲŅŖµÄ½šŹō£¬“æĪżæÉÓĆÓŚ×÷³¬µ¼²ÄĮĻ”¢°ėµ¼Ģ唣ĪżŌŖĖŲĪ»ÓŚÖÜĘŚ±ķµŚ5ÖÜĘŚ£¬µŚIVA×唣
¢ÅĪżŌŚ»ÆŗĻĪļÖŠ³£¼ūµÄ»ÆŗĻ¼ŪÓŠĮ½ÖÖ£ŗ+2ŗĶ_________”£
¢ĘĪż¼ČÄÜÓėŃĪĖįŅ²ÄÜÓėNaOHČÜŅŗ·“Ó¦£¬Éś³ÉµÄŃĪ¶¼ŹĒŗ¬+2¼ŪµÄĪż£¬Š“³öĪżÓėNaOHČÜŅŗ·“Ó¦µÄĄė×Ó·½³ĢŹ½_____________________________”£
¢ĒČēĶ¼ŹĒ¹¤ŅµŅ±Į¶ĪżµÄ»ł±¾Į÷³Ģ£ŗ
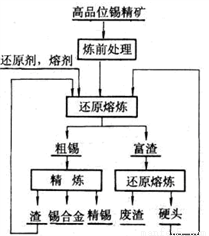
¢ŁĮ¶Ē°“¦Ąķ°üŗ¬Īż¾«æó£ØÖ÷ŅŖŗ¬Īż”¢Ģś”¢Ķ”¢Ē¦µÄĮņ»ÆĪļŗĶÉé»ÆĪļ£©ŌŚæÕĘųÖŠ±ŗÉÕ¼°Į÷³Ģ£¬Ź¹ĮņŗĶÉéŌŖĖŲ×Ŗ»ÆĪŖŅ×»Ó·¢µÄSO2ŗĶAs2O3£¬ĒėŠ“³öÉé“Å»ĘĢśæó£ØFeAsS2£©±ŗÉÕÉś³Éŗģ×ŲÉ«¹ĢĢåµÄ»Æѧ·½³ĢŹ½______________£¬Éś³ÉµÄĘųĢåÓĆ¹żĮæµÄNaOHČÜŅŗĪüŹÕ£¬ŌņČÜŅŗÖŠµÄÅضČ×ī“óµÄŗ¬ŃõĖįøłĄė×ÓŹĒ_______
¢Ś»¹ŌČŪĮ¶µÄ·“Ó¦ŌĄķĪŖ£ŗSnO2(s)+2CO(g) Sn(s)+2CO2(g)£¬Ņ»¶ØĪĀ¶ČĻĀ£¬ŌŚ¹Ģ¶ØČŻ»żµÄĆܱÕČŻĘ÷ÖŠ£¬SnO2ÓėCO·“Ó¦£¬Ę½ŗāŗóČŻĘ÷ÄŚĘųĢåµÄĘ½¾łÄ¦¶ūÖŹĮæĪŖ37.6£¬øĆĪĀ¶ČĻĀµÄĘ½ŗā³£ŹżK=____________
Sn(s)+2CO2(g)£¬Ņ»¶ØĪĀ¶ČĻĀ£¬ŌŚ¹Ģ¶ØČŻ»żµÄĆܱÕČŻĘ÷ÖŠ£¬SnO2ÓėCO·“Ó¦£¬Ę½ŗāŗóČŻĘ÷ÄŚĘųĢåµÄĘ½¾łÄ¦¶ūÖŹĮæĪŖ37.6£¬øĆĪĀ¶ČĻĀµÄĘ½ŗā³£ŹżK=____________
¢Ū“ÖĪżÖŠÖ÷ŅŖŗ¬ÓŠFe”¢Cu”¢Pb”¢Sb½šŹō£¬³£²ÉÓƵē½ā¾«Į¶·½·Ø»ńµĆ¾«Īż”£ĘäÖŠSnSO4ČÜŅŗ×÷µē½āŅŗ£¬¾«ĪżĮ¬½ÓµēŌ“µÄ______¼«£¬¼ģ²āæÉÖŖµē½āŗóŃō¼«ÄąÖŠŗ¬ÓŠSb”¢Cu”¢PbSO4µČ£¬Ōņµē½ā¹ż³ĢÖŠŃō¼«µē¼«·“Ó¦Ź½ĪŖ________________________________________”£
¢Ü¾«Į¶ŗó²śÉśµÄŌü¼°»¹ŌČŪĮ¶²śÉśµÄÓ²Ķ·ÖŠŅ»¶Øŗ¬ÓŠµÄŌŖĖŲŹĒ________”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com