
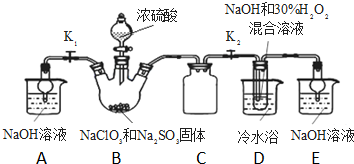
 �������ƣ�NaClO2������ҪƯ����̽��С�鿪չ����ʵ�飬�ش��������⣺
�������ƣ�NaClO2������ҪƯ����̽��С�鿪չ����ʵ�飬�ش��������⣺���� ��1����50%˫��ˮ����30%��H2O2��Һ����Ҫ����������Ͳ���ձ���������������ιܣ�
װ��D�з������巴Ӧ��װ����ѹǿ���ͣ�װ��C�������ǰ�ȫƿ����ֹDƿ��Һ������Bƿ�У�
��2��װ��B���Ʊ��õ�ClO2��װ��D��Ӧ�����Һ���NaClO2���壬װ��D������NaClO2��ClԪ�صĻ��ϼ۽��ͣ�˫��ˮӦ���ֻ�ԭ�ԣ����������ɣ����ԭ���غ��֪������ˮ���ɣ���ƽ��д����ʽ��
B�Ƶõ������к���SO2����װ��D�б������������ᣬ�������������ᱵ�ǰ�ɫ���������������
��3������Һ����ȡ���壬һ����������ᾧ�����ˡ�ϴ�ӡ�����ķ�����ע���¶ȿ��ƣ�
��4������Ŀ��Ϣ��֪��Ӧ�����¶�38�桫60�棬����60��ʱNaClO2�ֽ��NaClO3��NaCl��
��5��B�п��ܷ���Na2SO3+H2SO4��Ũ��=Na2SO4+SO2��+H2O��������SO2 ������D�У�SO2��H2O2 ��Ӧ���������ƣ�
��6���ٵ������۱���ɫ����Ӧ����ʱ���ⷴӦ��ȫ���μ����һ��Na2S2O3��Һʱ��Һ��ɫǡ����ȥ�Ұ�����ڲ���ԭ��˵������ζ��յ㣻
�ڸ��ݻ�ѧ��Ӧ�ɵù�ϵʽ��NaClO2��2I2��4S2O32-������Ʒ��NaClO2�����ʵ���x�����ݹ�ϵʽ���㣮
��� �⣺��1����50%˫��ˮ����30%��H2O2��Һ����Ҫ����������Ͳ���ձ���������������ιܣ����Ի���Ҫ��Ͳ��
װ��D�з������巴Ӧ��װ����ѹǿ���ͣ�װ��C�������ǰ�ȫƿ����ֹDƿ��Һ������Bƿ�У�
�ʴ�Ϊ����Ͳ����ֹDƿ��Һ������Bƿ�У�
��2��װ��B���Ʊ��õ�ClO2��װ��D��Ӧ�����Һ���NaClO2���壬װ��D������NaClO2��ClԪ�صĻ��ϼ۽��ͣ�˫��ˮӦ���ֻ�ԭ�ԣ����������ɣ����ԭ���غ��֪������ˮ���ɣ���ƽ��ʽΪ��2NaOH+2ClO2+H2O2=2NaClO2+2H2O+O2��
B�Ƶõ������к���SO2����װ��D�б������������ᣬ��Һ�п��ܴ���SO42-�����Ȼ�����Һ����SO42-�����������ȡ������Ӧ�����Һ���ȼ����������ᣬ�ټ�BaCl2��Һ����������ɫ��������˵������SO42-��
�ʴ�Ϊ��2NaOH+2ClO2+H2O2=2NaClO2+2H2O+O2��SO42-��ȡ������Ӧ�����Һ���ȼ����������ᣬ�ټ�BaCl2��Һ����������ɫ��������˵������SO42-��
��3������Һ����ȡ���壬һ����������ᾧ�����ˡ�ϴ�ӡ�����ķ�����Ϊ��ֹ��������NaClO2•3H2O��Ӧ���ȹ��ˣ�����Ŀ��Ϣ��֪��Ӧ�����¶�38�桫60�����ϴ�ӣ�����60����
�ʴ�Ϊ�����ȹ��ˣ���38�桫60����ˮϴ�ӣ�����60����
��4������Ŀ��Ϣ��֪��Ӧ�����¶�38�桫60�棬����60��ʱNaClO2�ֽ��NaClO3��NaCl�����������ȥD�е���ˮԡ�����ܵ��²�Ʒ�л��е�������NaClO3��NaCl��
�ʴ�Ϊ��NaClO3��NaCl��
��5��B�п��ܷ���Na2SO3+H2SO4��Ũ��=Na2SO4+SO2��+H2O��������SO2 ������D�У�SO2��H2O2 ��Ӧ���������ƣ�Ũ�����ѻӷ������������ѻӷ����Σ��������D����a��ȷ��b��c����ѡ��a��
��6���ٵ������۱���ɫ����Ӧ����ʱ���ⷴӦ��ȫ���μ����һ��Na2S2O3��Һʱ��Һ��ɫǡ����ȥ�Ұ�����ڲ���ԭ��˵������ζ��յ㣬
�ʴ�Ϊ���μ����һ��Na2S2O3��Һʱ����Һ��ɫǡ����ȥ�Ұ�����ڲ���ԭ��˵������ζ��յ㣻
������Ʒ��NaClO2�����ʵ���x����
NaClO2��2I2��4S2O32-��
1mol 4mol
0.25x c mol•L-1��V��10-3L
��x=c•V•10-3mol
�ʴ�Ϊ��c•V•10-3mol��
���� ���⿼�����������Ʊ�ʵ��Ļ����������������Ƶ����ʼ��к͵ζ���֪ʶ������ԭ���ǽ���Ĺؼ���ͬʱ����ѧ���������⡢���������������ѶȽϴ�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | FeO��Fe2O3��Na2O2��Ϊ���������� | B�� | ϡ���������ᡢ�Ȼ�����Һ��Ϊ���� | ||
| C�� | �ռ�����ᡢ���Ȼ�̼��Ϊ����� | D�� | ���ᡢˮ��������ˮ��Ϊ����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
| ʵ�� ��� | ʵ���¶� /�� | Na2S2O3 | H2SO4 | ����ˮ��� /mL | ||
| ���/mL | Ũ��/mol•L-1 | ���/mL | Ũ��/mol•L-1 | |||
| �� | 25 | 10 | 0.1 | 10 | 0.1 | 0 |
| �� | 25 | 5 | 0.1 | 10 | 0.1 | 5 |
| �� | 25 | 5 | 0.2 | 10 | 0.2 | 5 |
| �� | 50 | 5 | 0.1 | 10 | 0.1 | 5 |
| �� | 50 | 10 | 0.2 | 5 | 0.2 | 5 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ������ʹ����KMnO4��Һ��ɫ��������������ƣ���˱�Ϊ������ | |
| B�� | ���Ľṹ��ʽΪ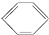 ������˫����������ˮ�����ӳɷ�Ӧ ������˫����������ˮ�����ӳɷ�Ӧ | |
| C�� | ����6��̼ԭ�Ӻ�6����ԭ����ͬһƽ���� | |
| D�� | ��1 mL����1 mLˮ��ֻ�Ϻ��ã������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| NaCl | MgCl2 | AlCl3 | SiCl4 | ����B | |
| �۵�/�� | 810 | 710 | 190 | -68 | 2 300 |
| �е�/�� | 1 465 | 1418 | 182.7 | 57 | 2 500 |
| A�� | SiCl4�Ƿ��Ӿ��� | B�� | ����B������ԭ�Ӿ��� | ||
| C�� | AlCl3���������� | D�� | KCl���۵����810�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com