
ij��ѧ����С��ͨ��ʵ��̽��NO2�����ʡ���֪��2NO2��2NaOH=NaNO3��NaNO2��H2O��
����1��������ͼ��ʾװ��̽��NO2�ܷ�NH3��ԭ(K1��K2��Ϊֹˮ�У��г�װ������ȥ)��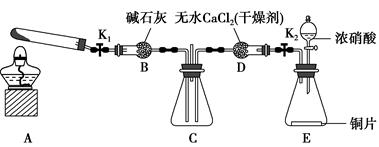
(1)Eװ������ȡNO2�Ļ�ѧ��Ӧ����ʽ��____________________________
____________________________________________��
(2)��NO2�ܹ���NH3��ԭ��Ԥ����Cװ���й۲쵽��������________________________________________________________________��
(3)ʵ������У�δ�ܹ۲쵽Cװ���е�Ԥ������С��ͬѧ�ӷ�Ӧԭ���ĽǶȷ�����ԭ����Ϊ���������ֿ��ܣ�
��NH3��ԭ�Խ��������ܽ�NO2��ԭ��
���ڴ������£�NO2��ת���ʼ��ͣ�
��______________________________________________________________��
(4)��ʵ��װ����һ�����Ե�ȱ����__________________________________��
����2��̽��NO2�ܷ���Na2O2����������ԭ��Ӧ��
(5)ʵ��ǰ����С��ͬѧ������ּ��衣
����1�����߲���Ӧ��
����2��NO2�ܱ�Na2O2������
����3��________________________________________________________��
(6)Ϊ����֤����2����С��ͬѧѡ������1�е�B��D��Eװ�ã���B�е�ҩƷ������Na2O2����ѡFװ��(��ͼ��ʾ)��������װ����ʵ�顣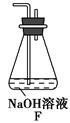
��װ�õĺ�������˳����(ijЩװ�ÿ����ظ�ʹ��)________��
��ʵ������У�Bװ���еķ�ĩ�ɵ���ɫ��ɰ�ɫ�������飬�ð�ɫ����Ϊ��������������������ɡ��Ʋ�Bװ���з�����Ӧ�Ļ�ѧ����ʽΪ__________________________________

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
(18��)��������ȼ�ջ����������������Ҫ�ɷ���SO2��CO2��N2��O2��ij�о���ѧϰС����ʵ������������װ���Ʊ�ģ��������������ģ������ͨ��ת����������Ч�ʡ�
�ش��������⣺
I��ģ���������Ʊ�
(1)��Aװ����SO2���仯ѧ��Ӧ����ʽΪ ��
(2)��Bװ����CO2��ʹ�ø�װ���ŵ���� ��
(3)���Ƶõ������������ֻ�ϣ����ģ���������ں���ʵ�顣
II���ⶨ������SO2���������
(4)������ģ����������ͨ��C��Dװ�ã�����C��D��ʢ�е�ҩƷ�ֱ��� �� ��(�����)
��KMnO4��Һ �ڱ���NaHSO3��Һ �۱���Na2CO3��Һ �ܱ���NaHCO3��Һ
(5)��ģ������������Ϊa mL/min����t1���Ӻ��������Һ�����ΪV mL(������Ϊ��״��)����SO2����������� ��
III������ģ������ͨ��ת����������Ч��(��ת����SO2ռԭ��SO2�İٷֱ�)
(6)��ģ������ͨ��ת����Eװ�ã�Eװ����ʢ��FeCl2��FeCl3�Ļ����Һ�������£����ɴ�SO2��O2�ķ�Ӧ���Դﵽ����Ŀ�ġ�д��SO2��FeCl3��Һ���뷴Ӧ���̵����ӷ���ʽ ��
(7)��ģ����������a mL/min������ͨ��ת����Eװ�ã�t2���Ӻ���Eװ���м����ữ��BaCl2��Һ�õ���ɫ����Һ���� ��ϴ�ӡ� ���������õ�mg���塣��ʵ����������������Ϊ��״�������ת����������Ч���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Ϊ��֤��һ����̼���л�ԭ�ԣ��������������ʵ�飺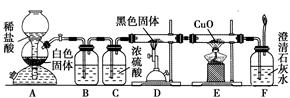
(1)װ��B�������˵��Լ��� ��
(2)װ��D�з�����Ӧ�Ļ�ѧ����ʽ�� �� ��
(3)������װ��C���ճ�ȥ������ˮ������������ �� ��
(4)������F��ʯ��ˮ����ǵ�����Ҳ��ȷ��CO���л�ԭ�ԣ�Ӧ����ͼ��װ�� �� ֮����������װ���е� (�����)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ѧ��ȤС������ʵ����̽�����������ʼ�ģ�ҵ��ȡƯ�ۣ����������װ�ý���ʵ�飨ʵ������ȡ�����ķ�ӦΪMnO2 + 4 HCI(Ũ�� MnCl2 + C12��+ 2 H2O����
MnCl2 + C12��+ 2 H2O����
�밴Ҫ��ش��������⣺
��1�����۵⻯����Һ�й۲쵽��������_________����Ӧ�����ӷ���ʽ____________��
��2�����������ɫ��������ɫ��ʪ�����ɫ������ɫ�������ʢ��________�������ƣ���
��3��C12��ʯ���鷴Ӧ��ȡƯ�۵Ļ�ѧ����ʽΪ___________________��
��4������ȤС����8.7g MnO2��������Ũ�����Ʊ��������������������Ƶñ�״���µ�Cl2______________L��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijʵ��С��ͬѧ̽��ͭ������ķ�Ӧ��
��1����Ũ�����ˮ���ղ�ͬ�������ɲ�ͬŨ�ȵ�������Һ����ȡ10 mL������Һ�ֱ���ͭƬ��Ӧ��ʵ���¼���£�
| ��� | Ũ������ˮ������� | ʵ������ |
| �� | 1:1 | ��Ӧ���ʿ죬��Һ�ܿ�����ɫ��ͭ˿�����д�������ð��������ʺ���ɫ |
| �� | 1:3 | ��Ӧ���ʽϿ죬��Һ�����ɫ��ͭ˿�����д�������ð����������ɫ |
| �� | 1:5 | ��Ӧ���������Ⱥ����ʼӿ죬��Һ�����ɫ��ͭ˿����������ð����������ɫ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
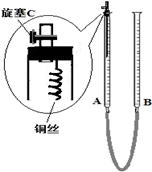
��ͼ1��ʾ��ʵ��������ȡ�����һ�ּ���װ�á�
��1��������������������Եķ��� ��
��2��������ͼ1��ʾװ����ȡ����ʱ�������õķ�Ӧ�����ͷ�Ӧ��״̬Ϊ________________________��
ijͬѧ�����ͼ2��ʾװ�ã��ô�������16.9%ϡ���ᷴӦ��ȡNO���岢̽�����������ļ�̬����ش��й����⡣
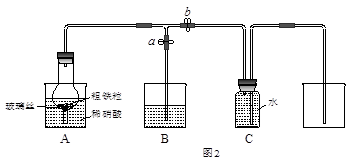
��3����֪16.9%ϡ������ܶ�Ϊ1.10g��cm-3���������ʵ���Ũ��Ϊ____________��������������λС����������63%����������16.9%ϡ����500mL������IJ�����������Ͳ�����������ձ��� ��
��4��ʵ�鿪ʼʱ���ȴ�ֹˮ��a���ر�ֹˮ��bʱ��Aװ�õĸ�����й۲쵽�������� ��Bװ���ձ���Һ���������_________________________����Aװ�������弸����ɫʱ����ֹˮ��b���ر�ֹˮ��a������Cװ���ռ�NO���塣
��5������������ҩƷ���Թܺͽ�ͷ�ιܣ�0.1mol��L-1KSCN��Һ��0.2mol��L-1����KMnO4��Һ��0.1mol��L-1KI��Һ����ˮ�ȡ��������һ����ʵ�飬̽��Aװ���ձ�����ȫ��Ӧ�������ܵļ�̬����д����ʵ�鱨�棺
| ʵ�鲽�� | ���� | ��������� |
| ��һ�� | ȡ����Һ��װ���Թܣ����Թ��е��뼸��KSCN��Һ�� | |
| �ڶ��� | | ����Һ��ɫ��ȥ����˵������Fe2+�� �������Ա仯����˵������Fe2+�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ѧʵ��Ҫ���ϡ���ɫ��ѧ�����ijʵ��С��ԡ�ͭ��Ũ���ᷴӦ��������̽����ʵ�顣�Իش���������:
(1)д��ͭ��Ũ���ᷴӦ�����ӷ���ʽ: ��
(2)��װ�â����,װ�â���ŵ��� ,װ�â������װ�â���ŵ���,���е��ŵ��� ��
(3)��װ�â���,��ʹNO2���������ƿ,Ӧ�ȹرյ��ɼ���������,�ٴ��ɼ���������;�����������ƿ��,��ͭ˿����,Ȼ���a��b��c���ر�,������ƿ���ڷ�ˮ��,������������������������
| A����ɫ |
| B������ |
| C�������ƽ����Է������� |
| D���ܶ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Ϊ��̽��AgNO3�������Ժ����ȶ��ԣ�ij��ѧ��ȤС�����������ʵ�顣
��.AgNO3��������
����������˿����AgNO3��Һ�У�һ��ʱ�����˿ȡ����Ϊ������Һ��Fe�������������Һ�е�Ag����������������ʵ�顣��ѡ�õ��Լ���KSCN��Һ����ˮ��
��1��������±���
| ���� | ���� | ���� |
| ȡ��������Ag�������Һ���Թ��У�����KSCN��Һ���� | ____ | ����Fe3�� |
| ʵ���� | ���� | ���� |
| a | ����������ˮ���� | ��ɫ���岻�ܽ� |
| b | ��������ϡ���ᣬ�� | ��ɫ�����ܽ⣬����������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�ס�����ͬѧ����ͼ��ʾװ�ý���ʵ��,̽������������Ӧ�IJ��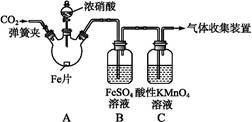
������:��.��Ũ��������ý�����Ӧ������,��������Ũ�ȵĽ���,�����ɵIJ�����+4��+2��-3�۵ȵ��Ļ����
��.FeSO4+NO Fe(NO)SO4(��ɫ)����H<0��
Fe(NO)SO4(��ɫ)����H<0��
��.NO2��NO���ܱ�KMnO4�������ա�
��ʵ������������¼����:
| ʵ����� | ʵ������ |
| ���ɼ�,ͨ��һ��ʱ��CO2,�رյ��ɼС� | |
| ��Һ©������,��Ũ���Ỻ��������ƿ��,�رջ����� | ���������� |
| ������ƿ,��Ӧ��ʼ��ֹͣ���ȡ� | ��A���к���ɫ�������,һ��ʱ���,������ɫ��dz;B����Һ����ɫ;C����Һ��ɫ��dz�� �ڷ�Ӧֹͣ��,A������ʣ�ࡣ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com