
�����й�˵����ȷ����(����)
A�����ں�������ϸ���һЩͭ�飬����Լ���������ǵĸ�ʴ
B��2NO(g)��2CO(g)===N2(g)��2CO2(g)�ڳ��������Է����У���÷�Ӧ�Ħ�H>0
C������0.1 mol��L��1Na2CO3��Һ��CO ��ˮ��̶Ⱥ���Һ��pH������
��ˮ��̶Ⱥ���Һ��pH������
D�������������Ҵ���������Ӧ(��H<0)����������Ũ���Ტ���ȣ��÷�Ӧ�ķ�Ӧ���ʺ�ƽ�ⳣ��������
C��[����] ���ֵ�����Ǹ�����������ͭ���ã���������ϸ���ͭ����γ�ԭ��أ���������������ٺ�����ǵĸ�ʴ��A�����B��ӦΪ���������С�ķ�Ӧ����Ӧ��S��0������Ӧ���Է����У���H��T��S��0���ʷ�Ӧ�Ħ�H��0��B��������������ˮ�ⷴӦΪ���ȷ�Ӧ�������¶�������CO ��ˮ��̶ȣ���Һ��c(OH��)������ҺpH����C����ȷ��Ũ������������Ӧ�Ĵ�������������Ũ����ʹ��Ӧ�������÷�ӦΪ���ȷ�Ӧ��һ��ʱ�������ϵ�¶����ߣ�ƽ�������ƶ���ƽ�ⳣ����С��D�����
��ˮ��̶ȣ���Һ��c(OH��)������ҺpH����C����ȷ��Ũ������������Ӧ�Ĵ�������������Ũ����ʹ��Ӧ�������÷�ӦΪ���ȷ�Ӧ��һ��ʱ�������ϵ�¶����ߣ�ƽ�������ƶ���ƽ�ⳣ����С��D�����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���������(Na2S2O3)�����������Լ������ﻹԭ���������ȡ������ֽ⡣��ҵ�Ͽ��÷�Ӧ��2Na2S��Na2CO3��4SO2===3Na2S2O3��CO2�Ƶá�ʵ����ģ��ù�ҵ���̵�װ����ͼ��ʾ��
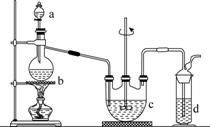
�ش��������⣺
(1)b�з�Ӧ�����ӷ���ʽΪ________________��c���Լ�Ϊ________��
(2)��Ӧ��ʼ��c�����л��Dz��������ֱ���塣�˻�������________��
(3)d�е��Լ�Ϊ________��
(4)ʵ����Ҫ����SO2�������ʣ����Բ�ȡ�Ĵ�ʩ��______________________________________(д������)��
(5)Ϊ�˱�֤��������ƵIJ�����ʵ����ͨ���SO2���ܹ�����ԭ����______________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
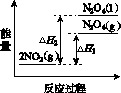
���ܱ������г���һ������NO2��������Ӧ2NO2(g) N2O4(g)����H����57 kJ��mol��1�����¶�ΪT1��T2ʱ��ƽ����ϵ��NO2�����������ѹǿ�仯��������ͼK203��ʾ������˵����ȷ����(����)
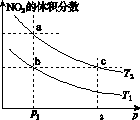
ͼK203
A��a��c����ķ�Ӧ���ʣ�a��c
B��a��b����NO2��ת���ʣ�a��b
C��a��c�����������ɫ��a�cdz
D����a�㵽b�㣬�����ü��ȵķ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
Ԫ�ص��ʼ��仯�����й㷺��;����������ڱ��е�������Ԫ�����֪ʶ�ش��������⣺
(1)��ԭ������������˳��(ϡ���������)������˵����ȷ����________��
a��ԭ�Ӱ뾶�����Ӱ뾶����С
b�������Լ������ǽ�������ǿ
c���������Ӧ��ˮ������Լ�����������ǿ
d�����ʵ��۵㽵��
(2)ԭ�������������������������ͬ��Ԫ������Ϊ________�������������ļ���������________��
(3)��֪��
| ������ | MgO | Al2O3 | MgCl2 | AlCl3 |
| ���� | ���ӻ����� | ���ӻ����� | ���ӻ����� | ���ۻ����� |
| �۵�/�� | 2800 | 2050 | 714 | 191 |
��ҵ��þʱ�����MgCl2�������MgO��ԭ����__________________________________��
����ʱ�����Al2O3�������AlCl3��ԭ����______________________________��
(4)�����(�۵�1410 ��)�����õİ뵼����ϡ��ɴֹ��ƴ���������£�
Si(��)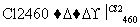 SiCl4
SiCl4 SiCl4(��)
SiCl4(��)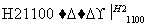 Si(��)
Si(��)
д��SiCl4�ĵ���ʽ��________________����������SiCl4�ƴ���ķ�Ӧ�У����ÿ����1.12 kg����������a kJ������д���÷�Ӧ���Ȼ�ѧ����ʽ��________________________________________________________________________
________________________________________________________________________��
(5)P2O5�Ƿ������Ը�������������岻����Ũ����������P2O5�������________��
a��NH3 ��b��HI c��SO2 d��CO2
(6)KClO3������ʵ������O2�������Ӵ�����400 ��ʱ�ֽ�ֻ���������Σ�����һ�����������Σ���һ���ε��������Ӹ�����Ϊ1��1��д���÷�Ӧ�Ļ�ѧ����ʽ��________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����£���1 mol��CuSO4��5H2O(s)����ˮ��ʹ��Һ�¶Ƚ��ͣ���ЧӦΪ��H1����1 mol��CuSO4(s)����ˮ��ʹ��Һ�¶����ߣ���ЧӦΪ��H2��CuSO4��5H2O���ȷֽ�Ļ�ѧ����ʽΪCuSO4��5H2O(s) CuSO4(s)��5H2O(l)����ЧӦΪ��H3���������ж���ȷ����(����)
CuSO4(s)��5H2O(l)����ЧӦΪ��H3���������ж���ȷ����(����)
A����H2����H3
B����H1����H3
C����H1����H3����H2
D����H1����H2����H3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���е�ع���ʱ��O2�������ŵ����(����)
|
|
|
|
|
| A.п�̵�� | B.��ȼ�ϵ�� | C.Ǧ���� | D.���ӵ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����˵������ȷ����(����)
A�������ԭˮ����ȵ��ˮ��������ܻ�����������
B��������ˮ(��NH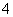 ��NH3)���û�ѧ��������绯ѧ����������
��NH3)���û�ѧ��������绯ѧ����������
C��ij�ֻ�ѧ��⼼�����м��ߵ������ȣ��ɼ�����ϸ��(V��10��12 L)�ڵ�����Ŀ����ӣ��ݴ˿�����ü�⼼���ܲ���ϸ����Ũ��ԼΪ10��12��10��11mol��L��1��Ŀ�����
D�������������Ӽ״��û��ȼ�ϵ���ֵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
NH3��һϵ�з�Ӧ���Եõ�HNO3��NH4NO3������ͼ��ʾ��
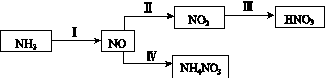
(1)���У�NH3��O2�ڴ��������·�Ӧ���仯ѧ����ʽ��_________________________��
(2)����2NO(g)��O2(g)  2NO2(g)��������������
2NO2(g)��������������
ͬʱ���ֱ���NO��ƽ��ת�����ڲ�ͬѹǿ(p1��p2)�����¶ȱ仯������(��ͼ)��

�ٱȽ�p1��p2�Ĵ�С��ϵ��________��
�����¶����ߣ��÷�Ӧƽ�ⳣ���仯��������________��
(3)���У������¶ȣ���NO2(g)ת��ΪN2O4(l)�����Ʊ�Ũ���ᡣ
����֪��2NO2(g)  N2O4(g)����H1 2NO2(g)
N2O4(g)����H1 2NO2(g) N2O4(l)����H2
N2O4(l)����H2
���������仯ʾ��ͼ�У���ȷ����(ѡ����ĸ)________��
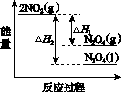 ��
��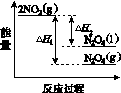
������A������������������B C
��N2O4��O2��H2O���ϵĻ�ѧ����ʽ��________________________________________��
(4)���У����NO�Ʊ�NH4NO3���乤��ԭ����ͼ��ʾ��Ϊʹ������ȫ��ת��ΪNH4NO3���貹��A��A��________��˵�����ɣ�________________________________________��
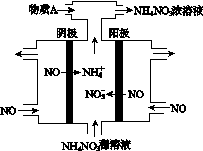
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�±��ж�Ӧ��ϵ��ȷ����(����)
��������������������������������
| A | CH3CH3��Cl2 CH2===CH2��HCl�D��CH3CH2Cl | ��Ϊȡ����Ӧ |
| B | ����֬�õ����� �ɵ��۵õ������� | ��������ˮ�ⷴӦ |
| C | Cl2��2Br��===2Cl����Br2 Zn��Cu2��===Zn2����Cu | ��Ϊ���ʱ���ԭ���û���Ӧ |
| D | 2Na2O2��2H2O===4NaOH��O2�� Cl2��H2O===HCl��HClO | ��Ϊˮ����ԭ����������ԭ��Ӧ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com