
��1����ȡ3.50 g������������ˮ���100 mL��Һ������ȡ��10.0 mL��Һ����ƿ�У���2��ָʾ������0.100 mol��L-1������ζ����յ㣬�����ı�Һ20.0 mL(���ʲ����ᷴӦ�������������������������ʵ�����?
��2����ȡ5.25 g����������ʧȥȫ���ᾧˮ�����ʲ��ֽ⣩���Ƶ�����Ϊ3.09 g,��Ba(OH)2��nH2O�е�nֵ��?
��3��������Ba(OH)2��nH2O����������Ϊ���٣�??????
�������������ɷ���ʽ���3.5 g��Ʒ��Ba(OH)2�����ʵ����������ݼ���ʧˮ�����������ˮ�����ʵ�������nֵҲ����á�ֻҪ�������������Ϳ������Ʒ�Ĵ��ȡ�?
�𰸣���1��n��Ba(OH)2��=![]() n(HCl)��
n(HCl)��![]() =
=![]() ��0.100 mol��L-1��20��10-3 L��10=0.01 mol��?
��0.100 mol��L-1��20��10-3 L��10=0.01 mol��?
��2��5.25 g������Ba(OH)2�����ʵ���Ϊ![]() ��0.01 mol=0.015 mol,����H2O�����ʵ���Ϊ
��0.01 mol=0.015 mol,����H2O�����ʵ���Ϊ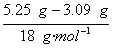 =0.12 mol��n��ֵΪ
=0.12 mol��n��ֵΪ![]() =8��?
=8��?
��3��3.50 g������Ba(OH)2��8H2O������Ϊ0.01 mol��315 g��mol-1=3.15 g���������У�Ba(OH)2��nH2O����������Ϊ![]() ��100%=90%��
��100%=90%��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com