
���ǵؿ��к�����ߵĽ���Ԫ�أ��䵥�ʼ��Ͻ������������е�Ӧ�������㷺��
(1)���̼�Ȼ�ԭ-�Ȼ�����ʵ���������Ʊ�������������ص��Ȼ�ѧ����ʽ���£�
Al2O3(s)��AlCl3(g)��3C(s)=3AlCl(g)��3CO(g) ��H=a kJ��mol��1
3AlCl(g)=3Al(l)��AlCl3(g) ��H=b kJ��mol��1
�ٷ�ӦAl2O3(s)��3C(s)=2Al(l)��3CO(g)�ġ�H= kJ��mol��1(�ú�a��b�Ĵ���ʽ��ʾ)��
��Al4C3�Ƿ�Ӧ���̵��м���Al4C3�����ᷴӦ(����֮һ�Ǻ�������ߵ���)�Ļ�ѧ����ʽ ��
(2)þ���Ͻ�(Mg17Al12)��һ��DZ�ڵ�������ϣ�������������£���һ����ѧ�����ȵ�Mg��Al������һ���¶���������á��úϽ���һ����������ȫ����ķ�Ӧ����ʽΪ
Mg17Al12��17H2=17MgH2��12Al���õ��Ļ����Y(17MgH2��12Al)��һ���������ͷų�������
�������Ʊ�þ���Ͻ�(Mg17Al12)ʱͨ�������Ŀ���� ��
����6.0mol��L��1HCl��Һ�У������Y����ȫ�ͷų�H2��1 mol Mg17Al12��ȫ�����õ��Ļ����Y������������ȫ��Ӧ���ͷų�H2�����ʵ���Ϊ ��
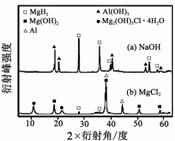 ����0.5 mol��L��1 NaOH��1.0 mol��L��1 MgCl2��Һ�У�
����0.5 mol��L��1 NaOH��1.0 mol��L��1 MgCl2��Һ�У�
�����Y��ֻ�ܲ��ַų���������Ӧ�������������X-����������ͼ����ͼ��ʾ(X-��������������ж�ij��̬�����Ƿ���ڣ���ͬ��̬���ʳ�������������Dz�ͬ)��������NaOH��Һ�У������Y�в�����������Ҫ������
(�ѧʽ)��
(3)�����������Խ��Al-AgO��ؿ�����ˮ��
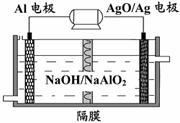 ������Դ����ԭ��������ͼ��ʾ���õ�ط�Ӧ
������Դ����ԭ��������ͼ��ʾ���õ�ط�Ӧ
�Ļ�ѧ����ʽΪ��
��
 �ƸԿ�����ҵ��ϵ�д�
�ƸԿ�����ҵ��ϵ�д� ��Ԫ����ĩ��ϰ�ȷ��ϵ�д�
��Ԫ����ĩ��ϰ�ȷ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����������ʹ��ˮ��ɫ��˵������������У� ��
A����ԭ�� B�������� C��Ư���� D������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ʯī�ڲ�����������ҪӦ�á�ij����ʯī�к�SiO2(7.8%)��Al2O3(5.1%)��Fe2O3(3.1%)��MgO(0.5%)�����ʡ���Ƶ��ᴿ���ۺ����ù������£�

(ע��SiCl4�ķе�Ϊ57.6 �棬�����Ȼ���ķе������150 ��)
(1)��Ӧ����ͨ��Cl2ǰ����ͨһ��ʱ��N2����ҪĿ����____________________��
(2)���·�Ӧ��ʯī�����������ʾ�ת��Ϊ��Ӧ���Ȼ��������е�̼��������ҪΪ________�����������ij��õ�ˮ�����Ļ�ѧ��Ӧ����ʽΪ____________________________________________��
(3)�����Ϊ�����衢________��������Һ���е���������________��
(4)����Һ�����ɳ��������ܷ�Ӧ�����ӷ���ʽΪ______________________________________________��100 kg����ʯī�����ܻ�â�������Ϊ______kg��
(5)ʯī��������Ȼˮ����ͭ���ĵ绯ѧ�����������ͼ����ʾ��ͼ��������Ӧ��ע��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
FeCl3���ִ���ҵ������Ӧ�ù㷺��ij��ѧ�о���ѧϰС��ģ�ҵ���������Ʊ���ˮFeCl3�����ø���ƷFeCl3��Һ�����ж���H2S��
I.���������ϵ�֪����ˮFeCl3�ڿ������׳��⣬����������������������Ʊ���ˮFeCl3��ʵ�鷽����װ��ʾ��ͼ�����ȼ��г�װ����ȥ���������������£�
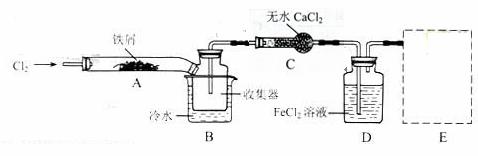
�ټ��װ�õ������ԣ�
��ͨ������Cl2���Ͼ�װ���еĿ�����
���þƾ�������м�·���������Ӧ���
�ܡ���
����ϵ��ȴ��ֹͣͨ��Cl2�����ø����N2�Ͼ�Cl2�����ռ����ܷ�
��ش��������⣺
װ��A�з�Ӧ�Ļ�ѧ����ʽΪ_____________________________________��
�ڢ۲����Ⱥ����ɵ���״FeCl3�ֽ����ռ��������������ڷ�Ӧ��A���Ҷˡ�Ҫʹ������FeCl3�����ռ������ڢܲ�������_______________________________________________��
���������У�Ϊ��ֹFeCl3��������ȡ�Ĵ�ʩ�У������ţ�_________________________��
װ��B�е���ˮԡ������Ϊ__________________��װ��C������Ϊ__________________��װ��D��FeCl2ȫ����Ӧ�����Ϊʧȥ����Cl2�����ö�ʧЧ��д������FeCl2�Ƿ�ʧЧ���Լ���___________��
�����߿��ڻ���β������װ��E��ע���Լ���
II.����ͬѧ��װ��D�еĸ���ƷFeCl3��Һ����H2S���õ��������˺�����ʯīΪ�缫����һ�������µ����Һ��
FeCl3��H2S��Ӧ�����ӷ���ʽΪ___________________________________________________��
������H+�������ŵ����H2�������ĵ缫��ӦΪ___________________________________��
�ۺϷ���ʵ��II��������Ӧ����֪��ʵ�������������ŵ㣺
��H2S��ԭ��������100%����____________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����þ��ҽҩ����������ҵӦ�ù㷺������þ��ԭ�Ƚ��Ʊ��ߴ�����þ��һ���µ�̽��������þ��(��Ҫ�ɷ�ΪMgCO3��������FeCO3)Ϊԭ���Ʊ��ߴ�����þ��ʵ���������£�

(1)MgCO3��ϡ���ᷴӦ�����ӷ���ʽΪ ��
(2)����H2O2����ʱ��������Ӧ�Ļ�ѧ����ʽΪ ��
(3)����2�ijɷ��� (�ѧʽ)��
 (4)���չ��̴������·�Ӧ��
(4)���չ��̴������·�Ӧ��
2MgSO4��C 2MgO��2SO2����CO2��
2MgO��2SO2����CO2��
MgSO4��C MgO��SO2����CO��
MgO��SO2����CO��
MgSO4��3C MgO��S����3CO��
MgO��S����3CO��
������ͼװ�ö����ղ�����������зֲ����ջ��ռ���
��D���ռ������������ (�ѧʽ)��
��B��ʢ�ŵ���Һ������ (����ĸ)��
a.NaOH ��Һ b.Na2CO3��Һ c.ϡ���� d.KMnO4��Һ
��A�еõ��ĵ���ɫ�������ȵ�NaOH��Һ��Ӧ��������Ԫ�����̬Ϊ��4��д���÷�Ӧ�����ӷ���ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��֪�� ���£�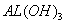 ��K�����ܽ��Զ����
��K�����ܽ��Զ����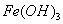 ����Ũ�Ⱦ�Ϊ0.1
����Ũ�Ⱦ�Ϊ0.1

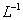 ��
��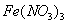 ��
��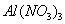 �����Һ�У���μ���NaOH ��Һ������ʾ��ͼ��ʾ����
�����Һ�У���μ���NaOH ��Һ������ʾ��ͼ��ʾ����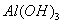 �����ʵ��������NaOH��Һ������Ĺ�ϵ ����������
�����ʵ��������NaOH��Һ������Ĺ�ϵ ����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�ڳ�ѹ��500�������£������ʵ�����Ag2O,Fe(OH)3 ,NH4HCO3 ,NaHCO3��ȫ�ֽ⣬�����������������V1\V2\V3\V4.�����С˳����ȷ����
A.V3��V2��V4��V1 B. V3��V4��V2��V1
C.V3��V2��V1��V4 D.V2��V3��V1��V4
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
LiPF6������ӵ���й㷺Ӧ�õĵ���ʡ�ij������LiF��PCl5Ϊԭ�ϣ����·�Ӧ�Ʊ�LiPF6�����������£�
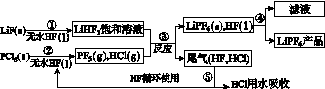
��֪��HCl�ķе��ǣ�85.0 �棬HF�ķе���19.5 �档
(1)�ڢٲ���Ӧ����ˮHF��������________________��________________����Ӧ�豸�����ò������ʵ�ԭ����______________________________________________(�û�ѧ����ʽ��ʾ)����ˮHF�и�ʴ�ԺͶ��ԣ�������ȫ�ֲ���ʾ�������С�Ľ�HFմ��Ƥ���ϣ���������2%��________��Һ��ϴ��
(2)������������ˮ�����½��У��ڢ۲���Ӧ��PF5����ˮ�⣬�����Ϊ�����ᣬд��PF5ˮ��Ļ�ѧ����ʽ��____________________________________��
(3)�ڢܲ�������õķ�����________���ڢݲ�����β����HF��HCl���õķ�����________��
(4)LiPF6��Ʒ��ͨ����������LiF��ȡ��Ʒw g�����Li�����ʵ���Ϊn mol�������Ʒ��LiPF6�����ʵ���Ϊ________mol(�ú�w��n�Ĵ���ʽ��ʾ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
38.4 mgͭ��������Ũ���ᷴӦ��ͭȫ����Ӧ���ռ�������22.4 mL����״��������Ӧ���ĵ�HNO3�����ʵ��������ǣ� ��
A��1.0��10-3 mol������B��1.6��10-3 mol����C��2.2��10-3 mol������D��2.4��10-3 mol
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com