
����Ŀ����ͨ�������£�����ͼ��ʾװ�����Ҷ�ȩ��OHC-CHO���Ʊ��Ҷ��ᣨH00C-COOH�������Ʊ���ӦΪ��OHC-CHO+2Cl2+2H2O��HOOC-COOH+4HCl������˵����ȷ����

A. ÿ����0.1mol�Ҷ�ȩ��Pt1���ų�2.24L���壨��״����
B. Pt1�ĵ缫��ӦΪ��4OH--4e-=2H2O+O2��
C. ÿ�õ�lmol�Ҷ��Ὣ��2molH+������Ǩ�Ƶ�����
D. ���������ṩCl-����ǿ�����Ե�����
���𰸡�D
��������A. ����ͼ��Pt1��Ϊ������������ԭ��Ӧ����Һ�е������ӷŵ�����������ÿ����0.1mol�Ҷ�ȩ��ת��0.4mol���ӣ�����0.2mol��������״���µ����Ϊ4.48L����A����B. ����A�ķ�����Pt1�ĵ缫�Ϸ��������ӵķŵ練Ӧ����B����C. ����A ������ÿ�õ�lmol�Ҷ��Ὣ��4molH+������Ǩ�Ƶ����ң���C����D. �������������ӵķŵ練Ӧ�����ɵ��������Ҷ�ȩ����Ϊ�Ҷ��ᣬ�������ṩCl-����ǿ�����Ե����ã���D��ȷ����ѡD��


 �п�������㾫��ϵ�д�
�п�������㾫��ϵ�д� ������ĩ��ϰ��ѵ��ϵ�д�
������ĩ��ϰ��ѵ��ϵ�д� С��ʿ��ĩ����100��ϵ�д�
С��ʿ��ĩ����100��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������ȷ��ʾ���з�Ӧ�����ӷ���ʽ����(����)
A. Fe2O3���ڹ����������Һ�У�Fe2O3��6H����2I��===2Fe2����I2��3H2O
B. 0.1 mol��L��1 NH4Al(SO4)2��Һ��0.2 mol��L��1 Ba(OH)2��Һ�������ϣ�Al3����2SO![]() ��2Ba2����4OH��===2BaSO4����AlO
��2Ba2����4OH��===2BaSO4����AlO![]() ��2H2O
��2H2O
C. ��0.1 mol��L��1��pH��1��NaHA��Һ�м���NaOH��Һ��HA����OH��===A2����H2O
D. �����������Һ��ͨ������SO2���壺ClO����SO2��H2O===HClO��HSO![]()
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����֪����ʮ�����ʣ�
��H2O����Cu����NO����SiO2����ϡ���ᡡ�������������߱���FeCl3��Һ���ఱˮ����ϡ���ᡡ��������
���������ṩ�����ʣ��ش��������⣺
��1�����ڴ��������__________�����ڵ���ʵ���__________��(���������)
��2�������кͷ�Ӧ�����ӷ���ʽΪH����OH��===H2O�������ӷ�Ӧ��Ӧ�Ļ�ѧ����ʽ��__________________________________��
��3��ʵ�����Ʊ�����Fe(OH)3�������õ���������________(���������)��������Ӧ�����ӷ���ʽΪ_________________________��
��4��ʵ��������0.5 mol��L��1 245 mL�����Һ����Ҫ�õ��IJ��������в��������ձ�����ͷ�ιܡ�________����Ҫ��������ƽ��ȡ���ʵ�����Ϊ________g�������ƺõ���Һ��ȡ��100 mL�����к��е�SO42����ĿΪ________(��NAΪ�����ӵ�������ֵ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������Ǽ���ԭ�ӵĻ�̬�����Ų����縺������ԭ����
A. 1s22s22p63s23p2 B. 1s22s22p63s23p3 C. 1s22s22p63s23p64s2 D. 1s22s22p4
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������ͼ�ǻ�ѧʵ���г��õļ���װ�á�


��ش��������⣺
��1��ָ���б�ŵ��������ƣ���________________����________________��
��2����������A��Bװ�õ���Ͽ�����ȡ��һ�ֳ������壬д��ʵ������ȡ������Ļ�ѧ��Ӧ����ʽ��______________________________________��
��3��Fװ�ÿ������������ɵ�CO2����������������ˮ���Ϸ�һ��ֲ���͵�Ŀ����____________________________��ֲ�����Ϸ�ԭ�еĿ�����ʵ��Ľ��__________��������������û����������Ӱ�졣
��4��д����ʯ��ʯ��ϡ������ȡ������̼�Ļ�ѧ����ʽ��___________________________________����ѡ��ͼ��________��________����������װһ����ȡ������̼��װ�á����������̼�����ķ�����_______________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���������Һ�ĵ絼��Խ��������Խǿ����0.100 mol��L��1��NaOH��Һ�ζ�10.00 mLŨ�Ⱦ�Ϊ0.100 mol��L��1 �������CH3COOH��Һ�����ô�������õζ���������Һ�ĵ絼����ͼ��ʾ������˵����ȷ����(����)
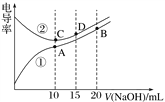
A. ���ߢٴ����ζ�HCl��Һ������
B. A����Һ�У�c(CH3COO��)��c(OH��)��c(H��)��0.1 mol��L��1
C. ����ͬ�¶��£�A��B��C������Һ��ˮ�ĵ���̶ȣ�C��B��A
D. D����Һ�У�c(Cl��)��2c(OH��)��2c(H��)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ʽ�Ȼ�ͭ����Ҫ����ɱ������
��1����ʽ�Ȼ�ͭ�ж����Ʊ�����
�ٷ���1: 45 ~ 50��ʱ����CuCl����Һ�г���ͨ������õ�Cu2 (OH)2 Cl2��3H2O�� �÷�Ӧ�Ļ�ѧ����ʽΪ_________________________��
�ڷ���2: ���Ƶ�CuCl2������ʯ���鷴Ӧ���ɼ�ʽ�Ȼ�ͭ��Cu��ϡ�����ڳ���ͨ������������·�Ӧ����CuCl2��Fe3+�Ը÷�Ӧ�д����ã����ԭ����ͼ��ʾ�� M'�Ļ�ѧʽΪ______��
��2����ʽ�Ȼ�ͭ�ж�����ɣ��ɱ�ʾΪCua(OH)bClc��xH2O�� Ϊ�ⶨij��ʽ�Ȼ�ͭ����ɣ� ��������ʵ��:
�ٳ�ȡ��Ʒ1.1160 g��������ϡHNO3�ܽ�����100.00 mL��ҺA��
��ȡ25. 00 mL��ҺA����������AgNO3��Һ����AgCl 0. 1722 g������ȡ25. 00 mL��ҺA������pH 4 ~ 5����Ũ��Ϊ0.08000 mol��L-1��EDTA(Na2H2Y��2H2O)����Һ�ζ�Cu2+ (���ӷ���ʽΪCu2++ H2Y2-=CuY2-+2H+)���ζ����յ㣬���ı���Һ30.00 mL��ͨ������ȷ������Ʒ�Ļ�ѧʽ(д���������)__________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ҵ����һ�ַ�������CO2������ȼ�ϼ״���Ϊ̽����Ӧԭ�����ֽ�������ʵ�飬�����Ϊl L���ܱ������У�����lmol CO2��3mol H2��һ�������·�����Ӧ��CO2(g)+3H2(g)![]() CH3OH(g)+H2O(g) ��H = ��49.0 kJ��mol-1��
CH3OH(g)+H2O(g) ��H = ��49.0 kJ��mol-1��
���CO2��CH3OH(g)��Ũ����ʱ��仯��ͼ��ʾ��
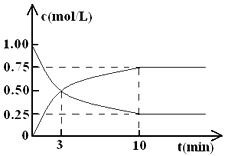
��1���ӷ�Ӧ��ʼ��ƽ�⣬������ƽ����Ӧ����v( H2 )= ________ ��CO2��ת����=________��
��2���÷�Ӧ��ƽ�ⳣ������ʽk=__________ ��
��3�����д�ʩ����ʹn(CH3OH)��n(CO2)�������_________________ (����)��
A����H2O(g)����ϵ�з���B������He(g)��ʹ��ϵѹǿ����
C�������¶� D���ٳ���lmolCO2��3molH2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��2016��IUPAC����117��Ԫ��ΪTS��������![]() ��ti��n����TS��ԭ�Ӻ���������������7.����˵������ȷ���ǣ� ��
��ti��n����TS��ԭ�Ӻ���������������7.����˵������ȷ���ǣ� ��
A. TS�ǵ������ڵڢ�A��Ԫ�� B. TS��ͬλ��ԭ�Ӿ�����ͬ�ĵ�����
C. TS��ͬ��Ԫ���зǽ��������� D. ������Ϊ176��TS���ط�����![]()
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com