
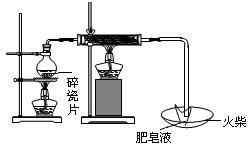
 ��#��#��#Դ��ͼ�е�E��F��������װ�ã������ⶨO2�������
��#��#��#Դ��ͼ�е�E��F��������װ�ã������ⶨO2������� (2��)
(2��)

 ��ʱ�ƿ�������ϰϵ�д�
��ʱ�ƿ�������ϰϵ�д� һ��һ��һ��ͨϵ�д�
һ��һ��һ��ͨϵ�д� �㽭֮��ѧҵˮƽ����ϵ�д�
�㽭֮��ѧҵˮƽ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
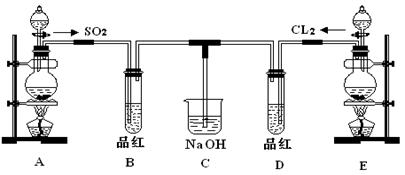
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
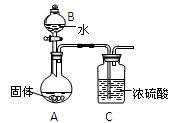
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
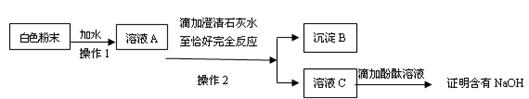
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
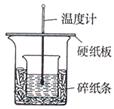
 Ȧ�����ձ�����ֽ���������⣬���貹��IJ��������� _______��
Ȧ�����ձ�����ֽ���������⣬���貹��IJ��������� _______�� ʵ�飬ʵ�������С���һ�¡��ɴ�ȷ�����������ȷ��
ʵ�飬ʵ�������С���һ�¡��ɴ�ȷ�����������ȷ�� ��С�ոĽ��ķ�����_______ ��
��С�ոĽ��ķ�����_______ ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
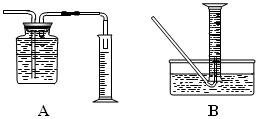
| | ���� | ��������� |
| ���ܢ� | | |
| ���ܢ� | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| ��� | ʵ������ | ʵ��Ŀ�� |
| A | ��SO2ͨ������KMnO4��Һ�� | ֤��SO2����Ư���� |
| B | ��Cl2ͨ��NaBr��Һ�У�Ȼ�����CCl4�������� | �Ƚ��������������ǿ�� |
| C | ��ͭƬ�ֱ���Ũ��ϡ���ᷴӦ | ̽��Ũ��ϡ���������Ե����ǿ�� |
| D | ��ʢ��20g���ǵ��ձ��м��뼸��ˮ��������ȡ��ټ�������Ũ���ᣬѸ�ٽ��衣 | ̽��Ũ�������ˮ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A������ƽ��ȡ4.0gNaOH���壬��100mL��Ͳ����1.00mol/L��NaOH��Һ |
| B����25mL�ĵζ��ܣ���װ�б�NaOH��Һ���ζ�δ֪Ũ�ȵ�������ȥNaOH��Һ22.32mL |
| C����pH��ֽ���������ˮ��pHΪ4 |
| D�������£��������ռ�500mLNO2���壬���NO2��������ʵ���Ϊ��0.5/22.4��mol |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com