
(14��) �����������������Լ�����Ӱ���ȣ����Ʊ��������£�

������(NH4)2Fe(SO4)2 6H2O��Һʱ���������ϡ���ᣬĿ����
��
6H2O��Һʱ���������ϡ���ᣬĿ����
��
�ƽ��ƵõIJ�Ʒ����������н������ط������������ͼ��TG%��ʾ������������ռԭ��Ʒ�������İٷ�������

����C��ʱ������Ļ�ѧʽΪ ��
�����о�ѧ����ʵ�������������ɫ�����H2�����ղ� ����Ҳ�����Ĵ����������ɣ����������һ������ʽ����������ʵ�� ��
����ȡ�������146����ˮ���FeC2O41.44g����ij��յ��ܱ������У��ٳ���0.04molCO��������1100�棬����FeO(s)+CO(g) Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣� ��
Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣� ��
��3������þ�ڹ�������������Ҫ���ã�����MgCl2Ϊԭ�ϻ�ȡ���¶Ⱥ�ѹǿP(HCl)g��MgCl2��6H2O�����ȷֽ�����Ӱ����ͼ��ʾ�������ͼ��ش��������⣺

��д��P(HCl)g = 0.25MPa���¶ȴ�300�����ߵ�550��ʱ��Ӧ�Ļ�ѧ����ʽ ��
��ʵ�������У���MgCl2��6H2O������ȵ�600��Ĺ����м����ò�����ˮMgCl2����ԭ���� ����Ҫ�õ���ˮMgCl2���ȡ�Ĵ�ʩ�� ��
������Fe2��ˮ�� �Ƣ�FeO
��3FeO(s)+H2O(g) Fe3O4(s)+H2(g)
��71.4%
Fe3O4(s)+H2(g)
��71.4%
��3����Mg(OH)Cl == MgO + HCl��
��P(HCl)С������ʱMg2+ �ᷢ��ˮ�⣬��Ҫ������MgO
ͨ��HCl���� ������P(HCl)����֤���ѹ����0.75������Mg2+ ˮ�⡣
����������1�����������ˮ�лᷢ��ˮ���ʹ��Һ�����ԣ����Լ���ϡ����ɷ�ֹˮ�⡣��������ȶ�������������������ͼ���֪��C��ʣ�������������ٷ����仯���������յĹ�����FeO�����������й���Fe3O4������Ϊ�����������ɣ����Է���ʽΪ3FeO(s)+H2O(g) Fe3O4(s)+H2(g)��FeC2O4������зֽ�ķ���ʽΪFeC2O4��FeO��CO����CO2������1.44g����������������CO2��CO����0.01mol��
Fe3O4(s)+H2(g)��FeC2O4������зֽ�ķ���ʽΪFeC2O4��FeO��CO����CO2������1.44g����������������CO2��CO����0.01mol��
FeO(s)+CO(g) Fe(s)+CO2(g)
Fe(s)+CO2(g)
��ʼ����mol�� 0.01 0.05 0 0.01
ת������mol�� x x x x
ƽ������mol�� 0.01��x 0.05��x x x��0.01
����ƽ�ⳣ���ı���ʽ��֪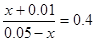 �����x��0.00714��������ת������71.4����
�����x��0.00714��������ת������71.4����
��2����ͼ���֪����300��ʱMg(OH)Cl�������ȵ�550��ʱ��������������þ���Ȼ��⣬���Է���ʽΪMg(OH)Cl == MgO + HCl�����ڳ�ѹ��þ������ˮ������������þ�����ռ��õ�����þ��Ҫ�õ���ˮMgCl2���ֹˮ�⣬����Ҫͨ���Ȼ���������Ҫѹǿ����0.75Mpa��


 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(14��) �����������������Լ�����Ӱ���ȣ����Ʊ��������£�
������(NH4)2Fe(SO4)26H2O��Һʱ���������ϡ���ᣬĿ���� ��
�ƽ��ƵõIJ�Ʒ����������н������ط������������ͼ��TG%��ʾ������������ռԭ��Ʒ�������İٷ�������
����C��ʱ������Ļ�ѧʽΪ ��
�����о�ѧ����ʵ�������������ɫ�����H2�����ղ� ����Ҳ�����Ĵ����������ɣ����������һ������ʽ����������ʵ�� ��
����ȡ�������146����ˮ���FeC2O41.44g����ij��յ��ܱ������У��ٳ���0.04molCO��������1100�棬����FeO(s)+CO(g)Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣� ��
��3������þ�ڹ�������������Ҫ���ã�����MgCl2Ϊԭ�ϻ�ȡ���¶Ⱥ�ѹǿP(HCl)g��MgCl2��6H2O�����ȷֽ�����Ӱ����ͼ��ʾ�������ͼ��ش��������⣺
��д��P(HCl)g = 0.25MPa���¶ȴ�300�����ߵ�550��ʱ��Ӧ�Ļ�ѧ����ʽ ��
��ʵ�������У���MgCl2��6H2O������ȵ�600��Ĺ����м����ò�����ˮMgCl2����ԭ���� ����Ҫ�õ���ˮMgCl2���ȡ�Ĵ�ʩ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(14��) �����������������Լ�����Ӱ���ȣ����Ʊ��������£�
������(NH4)2Fe(SO4)26H2O��Һʱ���������ϡ���ᣬĿ���� ��
�ƽ��ƵõIJ�Ʒ����������н������ط������������ͼ��TG%��ʾ������������ռԭ��Ʒ�������İٷ�������
����C��ʱ������Ļ�ѧʽΪ ��
�����о�ѧ����ʵ�������������ɫ�����H2�����ղ� ����Ҳ�����Ĵ����������ɣ����������һ������ʽ����������ʵ�� ��
����ȡ�������146����ˮ���FeC2O41.44g����ij��յ��ܱ������У��ٳ���0.04molCO��������1100�棬����FeO(s)+CO(g)Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣� ��
��3������þ�ڹ�������������Ҫ���ã�����MgCl2Ϊԭ�ϻ�ȡ���¶Ⱥ�ѹǿP(HCl)g��MgCl2��6H2O�����ȷֽ�����Ӱ����ͼ��ʾ�������ͼ��ش��������⣺
��д��P(HCl)g = 0.25MPa���¶ȴ�300�����ߵ�550��ʱ��Ӧ�Ļ�ѧ����ʽ ��
��ʵ�������У���MgCl2��6H2O������ȵ�600��Ĺ����м����ò�����ˮMgCl2����ԭ���� ����Ҫ�õ���ˮMgCl2���ȡ�Ĵ�ʩ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�콭��ʡ������ѧ����4��˫����ϰ��ѧ�Ծ����������� ���ͣ������
(14��) �����������������Լ�����Ӱ���ȣ����Ʊ��������£�
������(NH4)2Fe(SO4)2 6H2O��Һʱ���������ϡ���ᣬĿ���� ��
6H2O��Һʱ���������ϡ���ᣬĿ���� ��
�ƽ��ƵõIJ�Ʒ����������н������ط������������ͼ��TG%��ʾ������������ռԭ��Ʒ�������İٷ�������
����C��ʱ������Ļ�ѧʽΪ ��
�����о�ѧ����ʵ�������������ɫ�����H2�����ղ�����Ҳ�����Ĵ����������ɣ����������һ������ʽ����������ʵ�� ��
����ȡ�������146����ˮ���FeC2O41.44g����ij��յ��ܱ������У��ٳ���0.04molCO��������1100�棬����FeO(s)+CO(g) Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣� ��
Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣� ��
��3������þ�ڹ�������������Ҫ���ã�����MgCl2Ϊԭ�ϻ�ȡ���¶Ⱥ�ѹǿP(HCl)g��MgCl2��6H2O�����ȷֽ�����Ӱ����ͼ��ʾ�������ͼ��ش��������⣺
��д��P(HCl)g = 0.25MPa���¶ȴ�300�����ߵ�550��ʱ��Ӧ�Ļ�ѧ����ʽ ��
��ʵ�������У���MgCl2��6H2O������ȵ�600��Ĺ����м����ò�����ˮMgCl2����ԭ���� ����Ҫ�õ���ˮMgCl2���ȡ�Ĵ�ʩ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�꽭��ʡ������ѧ�ڿ�ѧ������⻯ѧ�Ծ� ���ͣ������
(14��) �����������������Լ�����Ӱ���ȣ����Ʊ��������£�

������(NH4)2Fe(SO4)2 6H2O��Һʱ���������ϡ���ᣬĿ����
��
6H2O��Һʱ���������ϡ���ᣬĿ����
��
�ƽ��ƵõIJ�Ʒ����������н������ط������������ͼ��TG%��ʾ������������ռԭ��Ʒ�������İٷ�������

����C��ʱ������Ļ�ѧʽΪ ��
�����о�ѧ����ʵ�������������ɫ�����H2�����ղ� ����Ҳ�����Ĵ����������ɣ����������һ������ʽ����������ʵ�� ��
����ȡ�������146����ˮ���FeC2O41.44g����ij��յ��ܱ������У��ٳ���0.04molCO��������1100�棬����FeO(s)+CO(g) Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣� ��
Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣� ��
��3������þ�ڹ�������������Ҫ���ã�����MgCl2Ϊԭ�ϻ�ȡ���¶Ⱥ�ѹǿP(HCl)g��MgCl2��6H2O�����ȷֽ�����Ӱ����ͼ��ʾ�������ͼ��ش��������⣺

��д��P(HCl)g = 0.25MPa���¶ȴ�300�����ߵ�550��ʱ��Ӧ�Ļ�ѧ����ʽ ��
��ʵ�������У���MgCl2��6H2O������ȵ�600��Ĺ����м����ò�����ˮMgCl2����ԭ���� ����Ҫ�õ���ˮMgCl2���ȡ�Ĵ�ʩ�� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com