
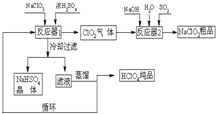
��ҵ������������(�е㣺90��)ʱ��ͬʱ�������������ƣ��乤���������£�

(1)��ȴ���˵�Ŀ��������NaHSO4��________�������NaHSO4������
(2)��Ӧ��2�з�����Ӧ�����ӷ���ʽΪ________��SO2����������________����
(3)ѭ��ʹ�õ�������________��
(4)����ͨ��������Һ�ķ����õ��������ԭ�������________��
(5)��ҵ���ò���������ͭ�����������������Ҳ���Ƶø����ᣬ���������ɵõ�20���ĸ����ᣮд�������ĵ缫��Ӧʽ(����������������Ի�ѧʽ����)________��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��ҵ�����������ᣨ�е㣺90��C��ʱ��ͬʱ�������������ƣ��乤���������£�
��ҵ�����������ᣨ�е㣺90��C��ʱ��ͬʱ�������������ƣ��乤���������£��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ������ѧ2012�������ѧ�ڵڶ����¿���ѧ���� ���ͣ�022
��ҵ������������(�е㣺90��)ʱ��ͬʱ�������������ƣ��乤���������£�

(1)��ȴ���˵�Ŀ��������NaHSO4��________�������NaHSO4������
(2)��Ӧ��2�з�����Ӧ�����ӷ���ʽΪ________��SO2����������________����
(3)ѭ��ʹ�õ�������________��
(4)����ͨ��������Һ�ķ����õ��������ԭ�������________��
(5)��ҵ���ò���������ͭ�����������������Ҳ���Ƶø����ᣬ���������ɵõ�20���ĸ����ᣮд�������ĵ缫��Ӧʽ(����������������Ի�ѧʽ����)________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��13�֣���ҵ������������ʱ����ͬʱ������һ�ֳ�������Ҫ������������Ư��
�������ƣ�NaClO2�����乤���������£�
��֪��
1��NaHSO4�ܽ�����¶ȵ����߶������ʵ������¿ɽᾧ������
2��������������Ϊֹ������֪���е���ǿ�ᣬ�е�90�档
��ش���������
��1����Ӧ�����з�����Ӧ�Ļ�ѧ����ʽΪ ����ȴ��Ŀ���� ��������������������ԭ���� ��
��2����Ӧ�����з�����Ӧ�����ӷ���ʽΪ ��
��3��ͨ�뷴Ӧ�����е�SO2 ����һ����H2O2����ͬ�������� NaClO2�����Ҫ˵��˫��ˮ�ڷ�Ӧ���ܴ���SO2 ��ԭ����
��4��Ca(ClO)2��ClO2��NaClO��NaClO2 �Ⱥ��Ȼ����ﶼ�dz��õ���������Ư������Ϊ���Ƕ����� ��д����ҵ����������NaOH��Һ������һ�������� NaClO�����ӷ���ʽ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010-2011ѧ��㶫ʡ��У������ѧ����ĩ���������ۣ���ѧ���� ���ͣ������
��ҵ�����������ᣨ�е㣺90oC��ʱ��ͬʱ�������������ƣ��乤���������£�
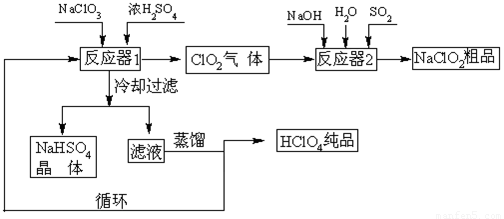
��1����ȴ���˵�Ŀ�� ��
��2��ͨ�뷴Ӧ��2��SO2������ ����Ӧ��2�з�����Ӧ�����ӷ���ʽΪ ��
��3��ѭ��ʹ�õ������� ��
��4������ͨ��������Һ�ķ����õ��������ԭ������� ��
��5��ͨ�����NaClO3ˮ��Һ�ķ���Ҳ�����Ʊ�NaClO4�����������Ʊ�HClO4��д�������ĵ缫��Ӧʽ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com