
��10�֣���֪A��B�� C�� D�������ʶ�����һ�ֹ�ͬ��Ԫ�أ��ת���Ĺ�ϵ��ͼ��ʾ��
���У�A��һ�ֵ���ɫ�Ĺ�̬�ǽ������ʣ�B��H2S��C��һ����ɫ�д̼�����ζ�����壬����ʹƷ����Һ��ɫ��D��H2SO4��
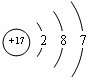
��1������������Ϣ�ƶϣ�A�� C�� ���ѧʽ��
��2����A�� B�� C�� D������������ѡһ�֣�������������ͬԪ�صļ�̬��Ԥ������ʾ��������Ի�ԭ�ԣ������ʵ����֤��
�ҵ�Ԥ�⣺
��Ҫ������
���ܵ�����
��ѧ����ʽ��
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
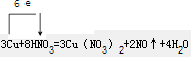

| �� |
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
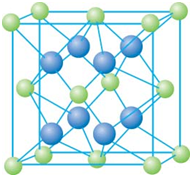 ��֪A��B��C��D��E����Ԫ�����ڱ���ǰ36�ŵ�Ԫ�أ����ǵ�ԭ��������������A������4��Ԫ�ؼȲ���ͬһ�����ֲ���ͬһ���壮B��C��ͬһ���壬D��E��ͬһ���ڣ���֪E�����ڱ���1-18���еĵ�7��Ԫ�أ�D��ԭ��������EС5��D��B�γɵľ����侧���ṹ��ͼ��ͼ��С�����D���������B����ش�
��֪A��B��C��D��E����Ԫ�����ڱ���ǰ36�ŵ�Ԫ�أ����ǵ�ԭ��������������A������4��Ԫ�ؼȲ���ͬһ�����ֲ���ͬһ���壮B��C��ͬһ���壬D��E��ͬһ���ڣ���֪E�����ڱ���1-18���еĵ�7��Ԫ�أ�D��ԭ��������EС5��D��B�γɵľ����侧���ṹ��ͼ��ͼ��С�����D���������B����ش�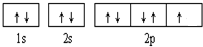


| 4��78g/mol |
| ag/cm3��6.02��1023/mol |
| 4��78g/mol |
| ag/cm3��6.02��1023/mol |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 A��I�ֱ��ʾ��ѧ��ѧ�г�����һ�����ʣ�����֮���ת����ϵ��ͼ��ʾ�����ַ�Ӧ�������û���г���������֪A��B��C��D��E��F���������о���ͬһ��Ԫ�أ�A��I�����ֳ����Ľ������ʣ�H�ڳ�������һ�ֳ����ķǽ�����̬���ʣ�
A��I�ֱ��ʾ��ѧ��ѧ�г�����һ�����ʣ�����֮���ת����ϵ��ͼ��ʾ�����ַ�Ӧ�������û���г���������֪A��B��C��D��E��F���������о���ͬһ��Ԫ�أ�A��I�����ֳ����Ľ������ʣ�H�ڳ�������һ�ֳ����ķǽ�����̬���ʣ�
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
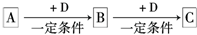
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com