
| O | 2- 3 |
| O | 2- 4 |
| ||


 ������ÿ�ʱ��ҵϵ�д�
������ÿ�ʱ��ҵϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A������ƽ����ֻ��������������������Ѭ�ƵĹ����в�����SO2���� |
| B����ɫ��ѧ�ĺ����Ǵ�Դͷ��������ҵ�����Ի�������Ⱦ |
| C�����ڳ�ʪ�Ŀ����з��ã�������ѧ��ʴ������ |
| D�����̿�ѧ�Ҹ����ڹ��˴�����Ϣ������ȡ��ͻ���Գɾͣ����˵���Ҫ�ɷ��Ǹߴ��ȵĶ������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ʱ��/s | 0 | 1 | 2 | 3 | 4 | 5 |
| c��NO�� ����10-4mol?L-1�� | 10.0 | 4.50 | 2.50 | 1.50 | 1.00 | 1.00 |
| c��CO�� ����10-3mol?L-1�� | 3.60 | 3.05 | 2.85 | 2.75 | 2.70 | 2.70 |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
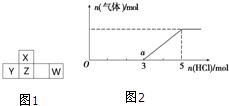 ������Ԫ��X��Y��Z��W�����ڱ��е�λ����ͼ1��ʾ������W���������������γɵ���Ҫ���ʣ�
������Ԫ��X��Y��Z��W�����ڱ��е�λ����ͼ1��ʾ������W���������������γɵ���Ҫ���ʣ�| 1 |
| 2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
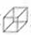 �����и߶ȶԳ��ԡ��������ԡ��������ܼ����ȶ��Ե��ص㣬��˺ϳ������鼰���������Ϊ��ѧ���ע���ȵ㣮������������������I��һ�ֺϳ�·�ߣ�
�����и߶ȶԳ��ԡ��������ԡ��������ܼ����ȶ��Ե��ص㣬��˺ϳ������鼰���������Ϊ��ѧ���ע���ȵ㣮������������������I��һ�ֺϳ�·�ߣ�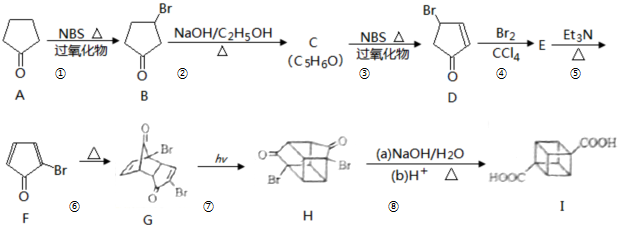

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
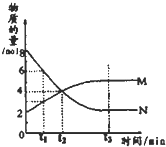 ��һ���¶��£�������ijһ��Ӧ��M��N�����ʵ����淴Ӧʱ��仯��������ͼ�����б�������ȷ���ǣ�������
��һ���¶��£�������ijһ��Ӧ��M��N�����ʵ����淴Ӧʱ��仯��������ͼ�����б�������ȷ���ǣ�������| A����Ӧ�Ļ�ѧ����ʽΪ��2M?N |
| B��t2ʱ�����淴Ӧ������ȣ��ﵽƽ�� |
| C��t3ʱ������Ӧ���ʴ����淴Ӧ���� |
| D��t3���������¶���Ӧ�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A��Na | B��Mg | C��Al | D��Zn |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com