
ʵ�����������᳧����(��Ҫ�ɷ�Ϊ���������P����FeS��SiO2��)�Ʊ�����(��ʽ�������ľۺ���)���̷�(FeSO4��7H2O)���������£�
(1)�����̢��в���������ͨ��������Һ�У���Һ����ɫ����________��
A��Ʒ����Һ������ B����ɫʯ����Һ
C������KMnO4��Һ������ D����ˮ
(2)���̢��У�FeS��O2��H2SO4��Ӧ�Ļ�ѧ����ʽΪ______________________________
(3)���̢��У�������������________��
(4)���̢��У������ᾧ��Ҫʹ�þƾ��ơ����żܡ������ǣ�����Ҫ��������________________��
(5)���̢ݵ���pH��ѡ�������Լ��е�________(��ѡ�����)��
A��ϡ���ᡡ��B��CaCO3����C��NaOH��Һ
(6)���̢��У�����ҺZ���ȵ�70��80 �棬Ŀ����______________________��
 ��ĩ���䵥Ԫ�����ิϰ��ϵ�д�
��ĩ���䵥Ԫ�����ิϰ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(14��)�����ֽ�������A��B��C������A����ɫ��ӦΪ��ɫ��B��C�dz������������ֽ�������A��B��C��������ס��ҡ���������D��E��F��G��H֮�䷢������ת����ϵ��ͼ����Щ��Ӧ�IJ���ͷ�Ӧ������û�б������
�����������Ϣ�ش��������⣺
��1��д���������ʵĻ�ѧʽ��
A_____________��H ______________�� G___________����____________��
��2��д�����з�Ӧ�Ļ�ѧ����ʽ��
��Ӧ��________________________________����Ӧ��_________________________________��
��3�� ��Ӧ���е���ת�Ƶ���ĿΪ6.02��1023��ʱ���ĵ������ҵ����ʵ���Ϊ mol
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ʵ����(14��)����HCOOH��һ����ɫ���ӷ���Һ�壬ijѧϰС����գ� ԭ�������������������ȼ��̶�װ��ʡ�ԣ��Ʊ����������CO������CO��ԭCuO��ĩ��
ԭ�������������������ȼ��̶�װ��ʡ�ԣ��Ʊ����������CO������CO��ԭCuO��ĩ��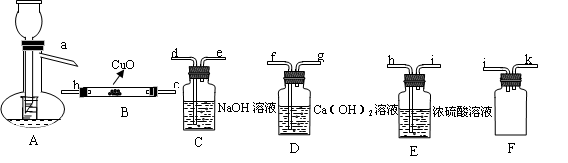
��1�����������������������ʱ��������������Ϊ�� A��( ) ��( )��( )��( ) ��( )
��2��Aװ����С�Թܵ����ã����ٴ���������
�� ���� ��
��3��֤��HCOOH���Ȳ�������CO������Ϊ�� ��
��4����ʵ����3���õ��ƾ��ƣ���A��B���⣬��ȱһ���ƾ��ƣ�Ӧ���� ��.
��ѧϰС���������֪��
Cu����ɫΪ��ɫ���Ϻ�ɫ����Cu2O����ɫҲΪ��ɫ��ש��ɫ����4CuO 2 Cu2O + O2������ Cu2O +2H+
2 Cu2O + O2������ Cu2O +2H+
= Cu+Cu2+ + H2O ����˶�CO��ֻ�ԭCuO�����ú�ɫ�����Ƿ���Cu2O������������о������������Ʒ�����
�����٣�ȡ�ú�ɫ������������ϡ�����У��۲���Һ��ɫ�ı仯��
�����ڣ�ȡ�ú�ɫ������������ϡ�����У��۲���Һ��ɫ�ı仯��
��5��д��Cu2O��ϡ���ᷴӦ�Ļ�ѧ����ʽ�� ��
��6���������۷����ڵĺ����ԣ����������ɣ������ڣ� �����ɣ� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�п��Ľ���Na��¶�ڿ����У���仯�������£�
��1����Ӧ��ķ�Ӧ�����������仯�Ĺ�ϵ���£�
�� ��Ӧ�� ���� ����Ӧ������ȡ������ȡ������ж���������������
�� 1 mol Na(s)ȫ��������Na2O(s)���Ȼ�ѧ����ʽ����������
��2����Ӧ����Na2O��ˮ�ķ�Ӧ�������ĵ���ʽ�� ��
��3����ɫ��ĩΪNa2CO3����������ˮ����Ϊ0.1 mol/L Na2CO3��Һ������˵����ȷ���� ������ĸ����
| A�������¶ȣ���Һ��pH���� |
| B��c(OH��)��c (H��)��c (HCO3��)��2 c (H2CO3) |
| C����������NaOH���壬c (CO32�D)��c (Na��)������ |
| D��c (Na��) > c (CO32�D) > c (HCO3�D) > c(OH�D) > c (H��) |
 Ni + 2NaCl����������Ӧʽ��_____��
Ni + 2NaCl����������Ӧʽ��_____���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����ѧʽΪNa2CO3����һ����Ҫ�Ļ���ԭ�ϡ��ִ��������������ֹ��գ�
һ�������Ƽ����ʳ��Ϊԭ���Ƽ�÷���������
�����Ȼ��������ᷴӦ�������ƣ�2NaCl+H2SO4=Na2SO4+2HCl��
���ý�̿��ԭ�����Ƶ����ƣ�Na2SO4+4C=Na2S+4CO��
����������ʯ��ʯ��Ӧ��̼���ƣ�Na2S+CaCO3=Na2CO3+CaS
�������������ά�Ƽ����ʳ�Ρ�����������̼Ϊԭ�ϣ��䷴ӦҲ���������У�
��NH3+CO2+H2O=NH4HCO3
��NH4HCO3+NaCl=NaHCO3+NH4Cl
��2NaHCO3=Na2CO3+CO2��+H2O
���������Ƽ������������ʳ��ˮ��ͨ�백������ͨ�������̼������̼�����ƣ��ټ���ϸ��ĩ����ͬ����ЧӦ�������Ȼ���ܽ��ͻȻ���ͣ���ʳ�ε��ܽ�ȱ仯���������Ȼ��������ʳ�β����������ð����ͺ�ͨ������̼�������������NaHCO3��NH4Cl���÷������Ĵ�������������������ѩ��
��1��ͨ�����ַ����ıȽϣ������Ƽ���յ�ȱ���� (д����)��
��2��������յ���ѭ�����õ������� (�ѧʽ)����Ʒ�ĸ�����NH4Cl�ȿ����������ֿ����������ɰ�����д��NH4Cl����ʯ�ҷ�Ӧ�Ļ�ѧ����ʽ ��
��3�������Ƽ��Ӧ�ķ���ʽΪ ��
��4��Ϊʲô�����Ƽ����������ʳ��ˮ��ͨ�백������ͨ�������̼�������� (д����)��
��5�������Ƽ��Ʒ�����к���̼�����ơ�����ü��ȷֽ�ķ����ⶨ������̼�����Ƶ�������������m1��ʾ����ǰ������Ʒ��������m2��ʾ���Ⱥ���������������̼�����Ƶ����������ɱ�ʾΪ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������ͭ�Ƚ������仯�������ճ�������Ӧ�ù㷺�����������ʵ��ش����⡣
(1)�����к���һ����̼������X(Fe3C)��X�������Ŀ����и������գ������д��ԵĹ���Y����Y���ڹ����������Һ�д������ڵ��������� ��Y�����Ũ���ᷴӦ����Һ�к��е��εĻ�ѧʽΪ ��
(2)ij��Һ����Mg2����Fe2����Al3����Cu2�������ӣ������м��������NaOH��Һ���ˣ��������������գ��������պ�Ĺ���Ͷ�������ϡ�����У�������Һ��ԭ��Һ��ȣ���Һ�д������ٵ��������� ��
A��Mg2�� B��Fe2�� C��Al3�� D��Cu2��
(3)����������Ҫ�Ĺ�ҵ���ϣ��÷���м�Ʊ������������£�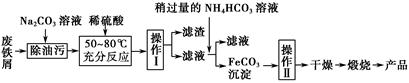
�ش��������⣺
�ٲ������������ ��������������� ��������ķ���Ϊ ��
����д������FeCO3���������ӷ���ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������Ļ�����Ӧ�ù㷺����FeCl3������������ӡˢ��·ͭ�帯ʴ��������ֹѪ���ȡ�
(1)д��FeCl3��Һ��ʴӡˢ��·ͭ������ӷ���ʽ_______________________
(2)����(1)�еķ�Ӧ��Ƴ�ԭ��أ��뻭��ԭ��ص�װ��ͼ�����������������д���缫��Ӧʽ��
������Ӧ_______________________________
������Ӧ_______________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��1�����µ�⼼���ܸ�Чʵ��CO2(g) + H2O(g) ="CO(g)" + H2(g) +O2(g) ������ԭ��ʾ��ͼ���£�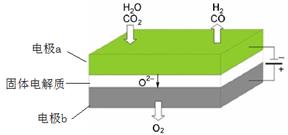
�ٵ缫b���� �����������ԭ������Ӧ��
��CO2�ڵ缫a�ŵ�ķ�Ӧʽ�� ��
��2����ҵ����ij����������Cu2O��Al2O3��Fe2O3��SiO2����ȡͭ�IJ����������£�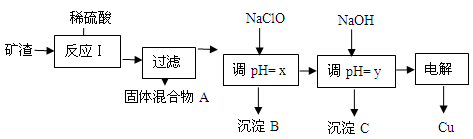
��֪�� Cu2O + 2H+ =" Cu" + Cu2+ + H2O
| ������ | Cu(OH)2 | Al(OH)3 | Fe(OH)3 | Fe(OH)2 |
| ��ʼ����pH | 5.4 | 4.0 | 1.1 | 5.8 |
| ������ȫpH | 6.7 | 5.2 | 3.2 | 8.8 |
 HClO��ClO������H+���Ӷ��ﵽ����pH��Ŀ��
HClO��ClO������H+���Ӷ��ﵽ����pH��Ŀ�� HClO+OH����OH������H+ ���Ӷ��ﵽ����pH��Ŀ��
HClO+OH����OH������H+ ���Ӷ��ﵽ����pH��Ŀ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������Ļ�����Ӧ�ù㷺����FeCl3������������ӡˢ��·ͭ�帯ʴ��������ֹѪ���ȡ�
��1��д��FeCl3��Һ��ʴӡˢ��·ͭ������ӷ���ʽ�� ��
��2��������1���еķ�Ӧ��Ƴ�ԭ��أ��뻭��ԭ��ص�װ��ͼ���������
��������д���缫��Ӧʽ��
������Ӧ�� ��
������Ӧ�� ��
��3����ʴͭ���Ļ����Һ�У���Cu2����Fe3����Fe2����Ũ�Ⱦ�Ϊ0.10 mol��L��1��������±����������ݺ�ҩƷ��������ȥCuCl2��Һ��Fe3����Fe2����ʵ�鲽�� ��
| | �������↑ʼ����ʱ��pH | ����������� ��ȫʱ��pH |
| Fe3�� Fe2�� | 1.9 7.0 | 3.2 9.0 |
| Cu2�� | 4.7 | 6.7 |
| �ṩ��ҩƷ��Cl2��ŨH2SO4��NaOH��Һ��CuO��Cu | ||
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com